Introduction
Primary hepatic tumors are rarely reported in children and account for 0.5% to 2.0% of pediatric neoplasms. Malignant tumors represent the majority of cases, while 30-40% are benign [1, 2]. Focal nodular hyperplasia (FNH) represents 2% to 7% of liver tumors and 0.02% of all pediatric solid tumors [3]. The most common benign hepatic lesions are hemangiomas and hemangioendotheliomas (15%), FNH (5%), and mesenchymal hamartomas (7%) [4]. Pediatric cases of FNH account for only 15% of all reported cases of FNH [2, 3]. The term FNH was introduced by Edmondson in 1956 and describes a well-circumscribed, non-neoplastic lesion characterized by pseudo-lobular hepatocellular hyperplasia, arranged around a centrally located fibrous scar, with accompanying abnormal vessels and ductal proliferation [1–3]. It occurs more frequently in women. The pathogenesis of FNH remains unknown. FNH may be associated with previous history of malignancy, chemotherapy, radiotherapy, and some vascular malformations [1–3]. Malignant transformation of FNH has not been reported [2, 5]. Development of the tumor is usually asymptomatic; thus, it is usually found incidentally as a result of widespread use of imaging [1–4]. Currently, there is no specific management algorithm for FNH in children, but due to its benign nature, a wait-and-see approach is recommended with rare indications for surgery limited to rapidly growing, symptomatic cases.
In this article we provide a review of the literature of the main characteristics, physiopathology, natural course, and therapeutic procedures of FNH in pediatric patients.
Pathophysiology and risk factors
The exact pathophysiology of FNH is still unknown. The most popular hypothesis suggests that FNH is a hyperplastic response of the hepatic parenchyma to preexisting aberrant blood flow [6, 7]. This abnormal perfusion may be related to presence of vascular malformations or local angiogenesis. Vascular malformation leads to increased blood flow and high pressure in sinusoids, resulting in hyperplasia [6]. Local angiogenesis may be triggered by local factors such as local venous thrombosis, post-thrombotic arteriovenous shunts, and tumor production of angiogenic factors, and its growth may be enhanced by systemic factors, such as oral contraceptives, female sex, and systemic elevation of tumor-associated growth factors [6]. Kumagai et al. considered that thrombosis of the hepatic artery and/or portal vein may cause hepatic ischemia/necrosis. Hepatic artery recanalization and tissue reperfusion also may lead to nodular hyperplasia [8]. The vascular theory of the pathogenesis of FNH is supported by several observations, such as a higher incidence of hemangioma among those with FNH in comparison with those without FNH lesions (20% vs. 9%) [9] and more frequent coexistence of FNH with hereditary hemorrhagic telangiectasia [10]. FNH and FNH-like nodules have also been described in patients with hypoplasia or agenesis of the portal vein, congenital or surgical portosystemic shunts, Budd-Chiari syndrome, and cirrhosis [1, 6].
Due to the predominance of FNH in young women, an association between female hormones and FNH was suggested [11]. Scalori et al. reported finding no association between FNH and age at menarche, menopausal status, or history of giving birth [12]. The potential association between oral contraceptives and FNH is controversial and debatable [7].
An increased incidence of FNH has been described in patients with a history of malignancies treated with chemotherapy and radiotherapy or who have undergone hematopoietic stem cell transplantation (HSCT) [13, 14]. Occurrence of FNH after chemotherapy was suggested to be related to different chemotherapeutic agents rather than the specific tumor type [14–16]. The use of high-dose alkylating regimens during myeloablative conditioning before HSCT, total body irradiation, and complications of HSCT leading to endothelial cell destruction such as graft-versus-host disease, veno-occlusive disease, and thrombotic microangiopathy are considered to be risk factors of FNH. They may lead to vessel injury, followed by a regenerative process resulting in development of FNH [17]. Icher-De Bouyn et al. suggested that polychemotherapy (especially busulfan and melphalan) and hepatic veno-occlusive disease could be the main risk factors for developing FNH [14]. Masetti et al. reported that the only independent risk factor associated with FNH was HSCT [18]. In another study, only age at time of HSCT (< 18 years vs. > 18 years) or age (< 12 years) and abdominal radiotherapy were significantly associated with a higher risk of FNH [19].
Molecular analysis showed polyclonal nature of liver cells in FNH in 50-100% of cases [20–22]. Recent studies demonstrated that the mRNA expression levels of the angiopoietin genes (ANGPT1 and ANGPT2), which are involved in vessel maturation, are altered. The ANGPT1/ANGPT2 ratio was elevated in all samples analyzed [22, 23]. The molecular study showed overexpression of genes encoding proteins of extracellular matrix, for example genes associated with activation of the transforming growth factor β (TGF-β) signaling pathway, and other genes such as platelet-derived growth factor subunit A (PDGFA) and platelet-derived growth factor receptor B (PDGFRB) involved in fibrogenesis. Furthermore, the expression of 19 zonated genes was altered in FNH. Most of these genes are regulated by β-catenin, including the gene encoding glutamine synthetase (GS), GLUL. GS is overexpressed in a “map-like” pattern, typical for FNH, visible in immunohistochemical staining [11, 23]. Central scar formation may be associated with the activation of hepatic stellate cells, the expression of 8-hydroxy-2’deoxyguanosine and inducible nitric oxide synthase, and the overexpression of vascular endothelial growth factor [24].
Clinical presentation
Focal nodular hyperplasia is rare among children and is often found incidentally [15, 25, 26]. It can be diagnosed at any age, from birth to adulthood. According to different studies, the median age is about 8 to 14 years [3, 15, 16, 25, 27]. Some prenatal cases of FNH are also reported [28]. In the group of children with isolated FNH, female preponderance is noticeable, which is the same as in adults [1, 2, 4, 15]. Among patients with a history of malignancy, the incidence of FNH is higher than in the general population (5% vs. 0.5%) and also higher among males than females [2, 15, 16, 19].
Children are more often symptomatic than adults. Symptoms are recorded in 25-51% of patients [1, 3, 4, 15, 27, 29]. The most common symptom is abdominal pain, while vomiting, anorexia, weakness, cholecystitis, and palpable abdominal masses occur more rarely [1, 3, 15, 30]. Abdominal pain is more common in pedunculated lesions and may be present in 80% of cases [31]. Liver function tests are usually within the normal range. The tumor marker α-fetoprotein (AFP) is also usually not elevated [2–4]. According to Zarfati et al., symptoms are more likely to occur if the FNH dimension is 5.5 cm or greater and in cases when the right liver is involved [3]. Patients with FNH with a maximal diameter of at least 2.5 cm, and with involvement of the fourth segment of the liver, are more likely to have an abnormal liver function test [3]. If there is a history of malignancy, lesions at diagnosis are more likely to be smaller and asymptomatic [1, 4, 15, 16]. Those patients also more often present multifocal FNH and FNH without a central scar [2, 4, 16].
Differential diagnosis
Diagnosis of FNH in the pediatric age group is more challenging than in adults. Confident diagnosis is difficult because of the higher frequency of atypical and multifocal lesions in children [1, 4].
Malignant tumors should be excluded in the first place as they are responsible for 70% of liver lesions in children [1, 4]. The differential diagnosis of hepatic lesions mainly depends on age at presentation [4]. In newborns, hemangiomas are reported as the most common, followed by mesenchymal hamartomas and hepatoblastoma [4]. In infants and toddlers, hepatoblastoma, rhabdoid tumor, and malignant germ cell tumor must be considered. In this group, the most common benign lesions are hemangiomas, mesenchymal hamartomas, and teratomas [4]. Children at school age and adolescents are more likely to be diagnosed with hepatocellular carcinoma, fibrolamellar hepatocellular carcinoma, embryonal sarcoma, angiosarcoma, or adenoma [4]. In incidental liver lesions, the risk of malignancy correlates with the tumor diameter greater than 5 cm [32].
Precise radiological assessment is important to avoid more invasive procedures, such as liver biopsy or surgical management, in benign liver lesions.
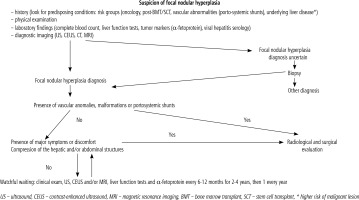
On ultrasonography (US), FNH is a well-delimitated, homogeneous, iso-, hypo- or hyperechoic mass with a central hyperechoic stellate scar [1, 2]. On color Doppler examination, a large artery with branches may be seen as a spoke-wheel pattern [1, 2]. The newest Doppler US technique – superb microvascular imaging (SMI) – allows the operator to assess vascularization of the lesion without a specific temporal window [32, 33]. A stellate scar with typical blood flow is visualized in 50% of cases [2] (Fig. 1).
Fig. 1
Well-marginated solid isoechoic mass off the right lobe of the liver, hypervascular on color and superb microvascular imaging (SMI). B mode – a solid mass without stellate scar. Color Doppler – a large artery with branches in a spoke-wheel pattern. Superb microvascular imaging (SMI) – a hypervascular solid lesion>
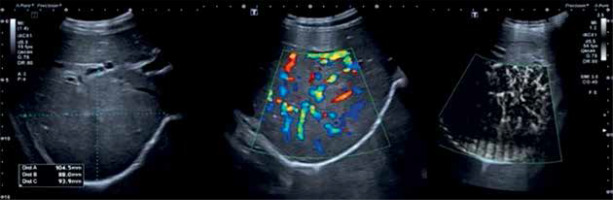
Contrast-enhanced ultrasound (CEUS) presents similar adequacy in detection rate as contrast-enhanced magnetic resonance and contrast-enhanced computed tomography [34]. The advantages of CEUS include performing the examination in real time and lack of exposure to ionizing radiation or nephrotoxic contrast agents. Furthermore, in comparison to magnetic resonance imaging (MRI) there is no need for sedation in children. As a result, it may be safely used for routine surveillance of liver lesions [34]. Typical features of FNH observed in CEUS are a centrifugal filling pattern, homogeneous hyperenhancement in the arterial and early portal venous phase, and iso- or hyperenhancement in the late phase [2]. Additional features are presence of spoke-wheel arteries and a central scar (Figs. 2, 3). CEUS enables differentiation of FNH from malignant tumors, for which hypoenhancement in the portal and/or late venous phase is characteristic, e.g. hepatoblastoma, metastasis of neuroblastoma [2, 34]. The main limitation of CEUS is its time restriction, which may result in reduced visualization of the spoke-wheel sign.
Fig. 2
Focal nodular hyperplasia. Contrast-enhanced ultrasound (CEUS). Arterial and early portal venous phase – centrifugal filling pattern and hyperenhance- ment of focal lesion in the liver
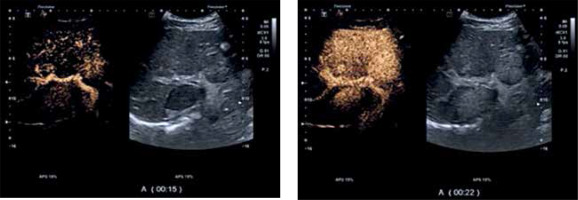
Fig. 3
Focal nodular hyperplasia. Contrast-enhanced ultrasound (CEUS). Late phase – isoenhancement of the lesion
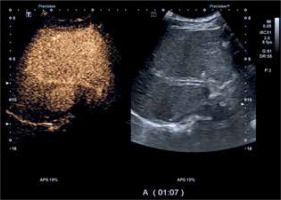
On an unenhanced computed tomography (CT) scan, FNH appears as a well-circumscribed, homogeneous mass, slightly hypo- or isodense to the surrounding liver parenchyma. A hypoattenuating central scar can be seen in 42-50% of the lesions [2, 35]. After contrast administration, the lesion enhances homogeneously and become hyperattenuating in the arterial phase. It does not include a central scar, which enhances in the late phase [1, 2, 5, 35]. In the portal vein phase, the lesion is hyper- or isodense and then isodense in the late phase [1, 2, 35]. In cases of atypical features, such as lack of a central scar, rapid washout of contrast agent in the portal venous phase, absence of enhancement of the central scar in the late phase, early draining veins, and partial peripheral rim-like enhancement on delayed images, further investigation is necessary to establish the correct diagnosis [2, 35] (Figs. 4-6).
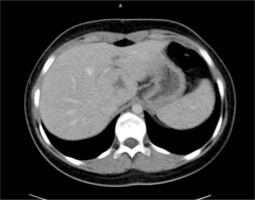
Fig. 4
Focal nodular hyperplasia – axial CT images. Non-contrast phase: lesion is isodense with surrounding parenchyma with slightly hypodense central scar
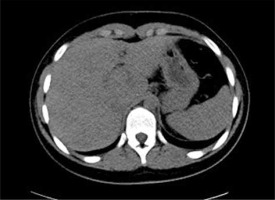
Fig. 5
Focal nodular hyperplasia – axial CT images. Post-contrast arterial phase: early contrast enhancement – hyperdense lesion with hypoattenuating central scar
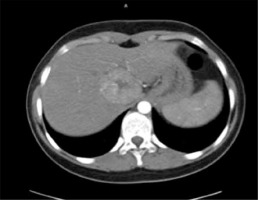
Fig. 6
Focal nodular hyperplasia – axial CT images. Post-contrast portal venous phase: the lesion became isoattenuating to liver parenchyma, the central scar remains hypodense
On MRI FNH is typically homogeneous and iso- or slightly hypointense on T1-weighted images and iso- or slightly hyperintense on T2-weighted images. The central scar appears hypointense on T1-weighted images and hyperintense on T2-weighted images and is present in about 75-80% of cases [2, 32]. After administration of hepatobiliary-specific agents, FNH enhanced homogeneously and became hyperintense to liver parenchyma in the arterial phase. In the portal venous phase, FNH may remain slightly hyperintense or become isointense. The central scar enhances in the delayed phase [1, 2, 5] (Figs. 7-9).
Fig. 7
Multiple focal nodular hyperplasia. MRI image. Axial T1 – iso- to moderately hypointense lesions with hypointense central scar
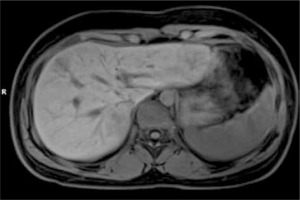
Fig. 8
Multiple focal nodular hyperplasia. MRI images. Axial T1 + contrast – early intense arterial phase enhancement, similar to CT
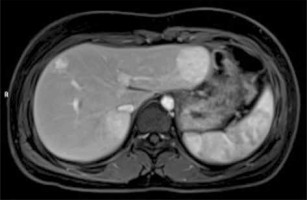
Fig. 9
Multiple focal nodular hyperplasia. MRI images. Axial T2 – iso- to somewhat hyperintense lesions with hyperintense central scar
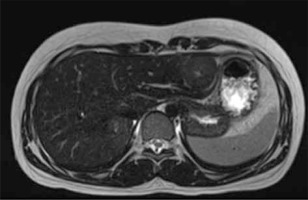
Focal nodular hyperplasia is characterized by an extended period of enhancement after gadolinium injection in comparison to other liver lesions. This is due to the presence of normal hepatocytes, which readily take up hepatobiliary-specific contrast agents, and the presence of malformed bile ducts, which prolong its excretion from hepatocytes [2, 36].
Fibrolamellar hepatocellular carcinoma (fHCC) is an important differential diagnosis due to presence of the central scar. Contrary to FNH, this scar is hypointense on all pre- and post- contrast sequences on CT images and hypointense on T2 images on MRI study [37, 38]. Diagnosis of fHCC is more probable when intratumoural calcifications, large size of the central scar, heterogeneity, concomitant lymphadenopathy and/or other metastases are present [1, 38]. Hepatic adenomas are associated with less vivid enhancement in the arterial phase and hypointensity in the delayed hepato-specific phase [36, 38]. Inflammatory and β-catenin subtypes of adenomas may be iso/hyperintense during hepatocyte phases and can be difficult to distinguish from FNH [36, 38].
Other imaging techniques include scintigraphy with 99mTc sulfur colloid. Sulfur colloid is taken up by Kupffer cells which are present in FNH. FNH shows normal (60-75% of cases) or increased (7-30% of cases) uptake of radiolabeled colloid [2, 39].
If the diagnosis of FNH in radiological imaging tests is uncertain, biopsy should be performed to exclude other lesions, most importantly malignant tumors [2, 3, 15].
Typical histopathological features of focal nodular hyperplasia include a well-circumscribed lesion without a capsule. The lesions may be singular or multiple, with the diameter ranging from a few millimeters to more than 20 cm [4]. FNH contains hyperplastic hepatocyte nodules, which are divided by fibrous bands. A central fibrous scar with fibrous septae radiating from it (stellate scar) contains malformed vessels of various size and thickness. Portal veins are not observed. Proliferating bile ductules are present at the junction of the fibrous regions with hepatocytes [1, 4, 22] (Figs. 10, 11).
Fig. 10
Needle biopsy of focal nodular hyperplasia. Part of a central scar with portal fibrosis and thin fibrotic septa, abnormal arterioles and mild distortion of the nodular architecture. H&E
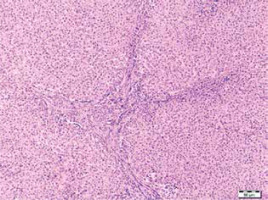
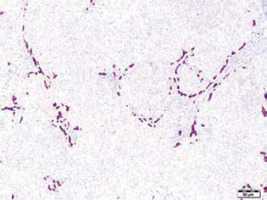
Fig. 11
Needle biopsy of focal nodular hyperplasia. The appearance of bile duct-like structures helps to distinguish FNH from adenoma. Immunohistochemical stain for cytokeratin 7 (CK7)
Nonclassical cases usually lack a stellate scar, especially if the diameter is < 3 cm [32]. In all forms of FNH, atypical hepatocytes or atypical mitotic figures are not found [2, 16]. Glutamine synthetase (GS) is positive in a “map-like” pattern, which is different than in β-catenin-mutated adenomas or FNH-like nodules [22, 23]. The decision to perform biopsy must be considered carefully as there is some risk of bleeding and dissemination of neoplastic cells if the lesion is not benign [15, 39]. Furthermore, according to some reports the percentage of confident diagnosis of FNH after needle biopsy is insufficient [15, 39]. It is related to difficulties in obtaining an adequate specimen for histopathology investigation, especially in the case of large and multifocal changes [15, 27]. It was reported that FNH may coexist with hepatocellular adenoma, hepatocellular carcinoma, and hepatoblastoma [7, 13, 15, 27, 40, 41].
Natural course
All kinds of FNH evolution have been described, from spontaneous involution to growth [1, 27]. The tumor diameter remains stable in about 2/3 cases, while about 1/3 to 1/4 of cases may gradually diminish [2]. Spontaneous regression may occur [2, 29]. Progressive increase of size of FNH is sporadic [3, 15, 42]. Serious complications are very rare and include hemorrhage, rupture, gastric outlet, hepatic vein and biliary obstruction, portal hypertension, and ascites [2, 3, 7, 15, 42]. No recurrence or neoplastic transformation had been described in children [2, 3, 42].
Management
There are no specific management guidelines for FNH in children. Over the years, the strategy has changed from a surgical approach to watchful waiting, which is currently recommended [3, 11, 15, 27, 42].
Management depends on the certainty of clinical and radiological diagnosis, comorbidities, presence of symptoms, and growth rate of the lesion [3, 4]. Zarfati et al. provided an algorithm useful for physicians, radiologists, and surgeons to deal with this uncommon benign tumor [3]. When typical features are present in imaging, the patient is asymptomatic, and there are no vascular abnormalities, biopsy may not be performed, and regular routine surveillance is indicated [1, 3]. Routine surveillance should be based on regular clinical examination, liver tests and AFP, imaging with US or MRI every 6 months for 2-4 years after diagnosis and then once a year. For children previously treated for tumors or after HSCT, if FNH is diagnosed with maximal certainty, a conservative “wait-and-see” strategy is also recommended [14, 18, 19].
When required, a surgical or radiological procedure should be performed in an experienced center [3].
Surgery is indicated in cases of diagnostic difficulties, pedunculated lesions, presence of complications, lesion progression, or for symptomatic patients [3, 11]. Lautz et al. analyzed 172 patients with FNH. Among them, 62% required resection, 21% underwent biopsy followed by observation, and 18% were managed nonoperatively. The main indications for surgery were presence of symptoms and suspicion of malignancy [27]. Ji et al. reported that 47 of 79 (59%) patients underwent primary surgical resection and another 2 after initial observation. Surgical management was recommended in the case of symptomatic patients, uncertain diagnosis, abnormal liver function, large diameter of the lesion (≥ 5 cm), or lesion growth. Liver biopsy was performed in 14 patients [15]. Zarfati et al. reported 50 patients; 13 (26%) had surgery as first-line management and 10 patients required second-line surgery. Indications for first-line surgery included: symptoms, portosystemic shunt, and diagnostic uncertainty; for second-line surgery: lesion growth, persistent symptoms, and patent portosystemic shunts. One patient underwent embolization of the FNH as third-line treatment [3]. There is no need for anatomical resection of the segment with FNH, and tumorectomy alone may be performed [3]. In cases where FNH is secondary to presence of congenital porto-systemic shunt, closure of the shunt with restoration of intrahepatic portal flow may result in tumor shrinkage and should always be performed when possible [1, 43, 44]. Also, closure of surgical porto-systemic shunt should be considered [1, 43].
In cases when the lesion is unresectable due to central location or proximity to large vessels, transarterial vascular embolization (TAE) may be performed as a minimally invasive treatment option [45]. Embolization may lead to reduction of the mass size and make surgical resection amenable or ablate it such that no further therapy is needed [42, 45]. Various agents, including polyvinyl alcohol, tris-acryl gelatin microspheres, and gel foams, have been used in the management of FNH [38, 46]. Yan et al. reported on 17 patients who underwent TEA with a lipiodol-bleomycin emulsion and N-butyl cyanoacrylate, achieving a positive response in all cases, with an average reduction in lesion volume of 96.9% (ranging from 86% to 100%) [46]. Alaoui et al. presented two pediatric patients, who were managed with transarterial coil embolization followed by surgery [47]. Radiofrequency ablation may be an option in patients with small but anatomically difficult lesions [42]. There are few reports of liver transplant due to FNH [48, 49].
A proposed management algorithm is provided in Figure 12 (modified from sources [3, 50]).
Conclusions
Focal nodular hyperplasia is a benign lesion rarely reported in children. Diagnosis can be made by radiological imaging with rare indications for diagnostic biopsy or resection. In most cases, a watchful waiting strategy
is appropriate. Invasive intervention is indicated for symptomatic patients, or in the case of diagnostic difficulties, large diameter of the lesion, and its rapid growth.