Venous thromboembolism (VTE), defined as deep vein thrombosis (DVT) and/or pulmonary embolism (PE), is a well-recognized complication in patients with traumatic brain injury (TBI) due to increased venous stasis from paralysis, prolonged coma [1–3] and systemic hypercoagulability [4, 5]. Approximately 5% of all hospitalized patients develop VTE during their hospital stay. However, for patients with TBI, the risk of VTE has been reported to vary from 30 to 60% [6–9] and to be associated with significant morbidity and mortality [4, 10].
The radiological prevalence of intracranial hemorrhage (ICH) among patients with TBI is approximately 45% [11]. The risk for progression of ICH is high, particularly early after injury, above all due to a trauma-induced coagulopathy, which affects approximately one third of all patients with TBI [12] and which is associated with increased mortality risk [13]. Furthermore, an estimated 3 to 4% of patients show ICH progression following pharmacological VTE prophylaxis (PTP) [14, 15]. Therefore, it remains challenging to balance the initiation of PTP with the potential risk of worsening ICH in this patient population.
Currently no standard exists on the optimal timing and agent of choice for PTP in patients with TBI and wide variability in the practice patterns has been reported [16].
Accordingly, studies attempting to identify patients with potential benefit from PTP reported conflicting results [17–22]. However, as patients with different grades of severity of TBI and thus various risk profiles were included, interpretation of these findings remains difficult. In particular, patients with moderate and severe TBI have longer periods of immobilization than patients with mild TBI and hence are at higher risk of VTE. Only a few studies have analyzed PTP in patients with moderate and severe TBI [3, 4, 23, 24], presenting conflicting results concerning the influence of PTP agents on development of VTE.
The present study investigates the timing of PTP exposure with a continuous infusion of unfractionated heparin (UFH) on VTE and ICH progression in patients with moderate to severe TBI during the hospitalization and aims to identify risk factors associated with development of VTE.
METHODS
This non-concurrent cohort study was conduct-ed in the surgical ICU of the University Hospital of Zurich, a level I Trauma Center in Switzerland, in compliance with the current version of the Declaration of Helsinki and the national legal and regulatory requirements, and was approved by the local Ethics Committee (KEK-ZH-Nr. 2016–00332). The ethics committee waived the requirement for written informed consent.
All subjects admitted to the surgical ICU between 1 January 2012 and 30 June 2015 with a diagnosis of TBI (ICD Code S00–S09) were identified in the hospital’s electronic database (KISIM, Cistec Zurich, Switzerland) containing all relevant information about the patients.
Inclusion criteria for this study were: age > 16 years; diagnosis of blunt TBI classified as moderate or severe TBI before sedation and intubation. Patients with GCS 12–9 were classified as having moderate TBI, and patients with GCS < 8 as having s evere TBI. A minimum length of stay (LOS) in the ICU of 48 hours was set arbitrarily to exclude patients with initially over-estimated severity and no need of an intensified treatment, e.g. due to alcohol or drug influence at the time of trauma, and patients who died before an intensified treatment was implemented.
Baseline patients’ demographic data, pre-hospital data, and severity scores were collected. Baseline demographic data included age, sex, and treatment with anticoagulants or antiplatelet agents before trauma. Pre-hospital data after TBI referred to hypotension (defined as systolic blood pressure < 90 mm Hg), hypoxemia (defined as SpO2 < 90% without supplemental oxygen administration), and pupillary reactivity during the rescue phase. Pupillary reactivity was considered pathological if one or both pupils presented no response to light. Severity scores included GCS before sedation and intubation and in particular its motor component (GCS m), which better predicts 6-month outcome after TBI [25], Injury Severity Score (ISS) [26], and the Abbreviated Injury Scale of the head region (AIS-head) [27].
Data collected during the ICU stay included timing of initiation of PTP (expressed in days after trauma), the number of days on mechanical ventilation (MV-days, expressed in days), the ICU length of stay (LOS, expressed in days), and ICU mortality (ICU-mortality).
The Marshall classification [28] of head injury was used based on the first computed tomography (CT) after trauma. Particularly, the percentages of patients with epidural hematoma (EDH) and traumatic subarachnoid hemorrhage (tSAH) were recorded. We decided to use the Marshall classification and to collect data about frequency of EDH and tSAH because these parameters are needed to calculate the IMPACT score to predict 6-month outcome [29].
Patients were sedated with propofol and remifentanil, or midazolam and fentanyl. In the case of elevated intracranial pressure, after a control head CT scan to exclude the need of surgery, the sedation was deepened and, in some cases, barbiturates were administered. Furthermore, moderate hyperventilation (PaCO2 4.8–5.2 kPa) was allowed.
Screening for VTE was not routinely performed at our ICU. By clinical symptoms and/or signs suggestive of VTE (for intubated and sedated patients, redness, pain, warmth and/or swelling in one extremity), or diagnosis of vein thrombosis by ultrasound-guided insertion of central lines, further investigations were performed. VTE was defined as an event of DVT and/or PE, verified by either Doppler ultrasonography of the extremities, and/or CT pulmonary angiography depending on the patient’s clinical stability and the attending physician’s choice. The observation period was the hospitalization length of stay.
According to our institutional policy, sequential pneumatic compressive devices (SCD) (Kendall SCD, Covidien) were universally applied to lower extremities, whenever not contraindicated, until initiation of PTP. The decision and timing for initiation of PTP were at the discretion of the attending intensivist in accordance with the neuro- and trauma surgeons. At our institution, a continuous infusion of UFH of 10,000 IE/day is used for PTP in all patients with TBI, based on the possibility of a rapid reversal effect with protamine in case of need. Any progression of ICH after the initiation of PTP was identified through the review of head CTs both before and after initiation of PTP by two experienced intensivists in accordance with the neuroradiologist. A CT scan after initiation of PTP was performed in the case of neurological deterioration, defined as a decline in GCS ≥ 2 or onset of a new motor deficit, or by absent neurological improvement. If the GCS assessment was not possible, such as in the case of sedated and intubated patients, a CT scan was usually performed. In the case of neurological improvement, a CT scan is usually planned in an out-patient clinic.
Statistical analysis
For descriptive statistics, categorical variables were expressed as absolute numbers with percentages, normally distributed quantitative variables as mean ± standard deviation (SD) and non-normally distributed variables as median with interquartile range (IQR).
In order to identify potential risk factors associated with VTE, we first performed a univariate logistic regression analysis considering VTE as the outcome variable (dependent variable). Odds ratios (ORs) were calculated and expressed with the corresponding 95% confidence intervals (95% CI). With only 23 clinically relevant episodes of VTE (events), we decided to insert no more than two independent variables into a multivariable logistic regression model, in order to keep the risk of overfitting low [30]. By more than two statistically significant independent variables identified by univariate analysis, we constructed all possible multivariable models as a combination of 2 independent variables per model. Statistical significance was set at P value < 0.05 for all analyses. Statistical analysis was performed using Stata Version 12.1 (StataCorp LP, College Station, TX, USA).
RESULTS
One hundred seventy-seven patients were included in this study (Figure 1). Demographic and baseline characteristics of the study population are shown in Table 1. At the time of admission, 32 patients (18.1%) were being treated with anticoagulants or antiplatelet agents. Patients were stratified by the Marshall classification and numbers of patients per strata are provided in Table 2. Median time between TBI and initiation of PTP was 8 days (IQR 3–13).
FIGURE 1
Flowchart of the study
TBI – traumatic brain injury, ICU – intensive care unit, GCS – Glasgow Coma Scale
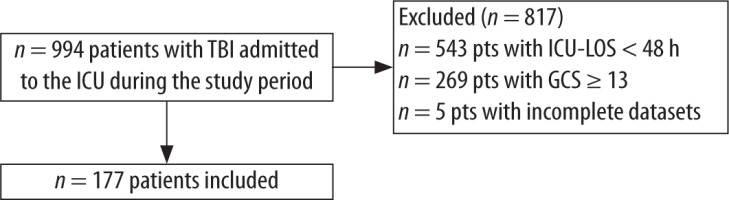
TABLE 1
Baseline demographic data and severity scores of the study population
[i] GCS m – motor component of Glasgow Coma Scale, AIS-head – abbreviated injury scale for head region, ISS – Injury Severity Score, SAPS II – Simplified Acute Physiology Score, ICU-LOS – length of stay at the intensive care unit, MV-days – number of days on mechanical ventilation, ICU-mortality – mortality in the intensive care unit, VTE – venous thromboembolism, IVC filters – inferior
TABLE 2
Radiological findings on first brain computed tomography scan and Marshall classification
A total of 23 clinically relevant episodes of VTE occurred (13%) during the hospitalization. On average, VTE was diagnosed on day 16.4 (± 11.2) after trauma. Demographic and baseline characteristics of patients who developed VTE (n = 23) did not differ from patients who did not, as presented in Table 1. However, patients who developed VTE had significantly more MV-days and longer ICU-LOS than patients who did not, as shown in Table 1.
In the univariate logistic regression analysis (Table 3), hypotension in the pre-hospital setting, ICU-LOS, MV-days and delay of initiation of PTP were associated with the development of VTE. With four statistically significant independent variables, we could construct six different multivariable models. The different models (no more than 2 independent variables per model) are presented in Table 4. The multivariable model containing MV-days and ICU-LOS is not displayed due to overfitting. PTP initiation is the only independent variable which remains statistically significant in the different multivariable logistic regression models.
TABLE 3
Univariate logistic regression analysis considering venous thromboembolism as the outcome variable
TABLE 4
Multivariable models constructed with two independent variables per model
Follow-up head CT scans after initiation of PTP were obtained from 65 (35.6%) patients. ICH progression was diagnosed in 7 (4%) patients and in 3 of the them it led to a therapeutic change: intracranial surgery (n = 2) and interruption of life-sustaining therapy (n = 1).
Major bleeding complications after PTP initiation other than ICH occurred in 5 patients (2.8%). Three of them (1.7%) had gastrointestinal bleeding (two patients needed administration of blood products due to bleeding stomach ulcers, and one patient had retroperitoneal bleeding and needed surgery), and 2 (1.1%) patients bled from the respiratory system (one patient had a hemothorax after cardiopulmonary resuscitation, and one patient presented massive hemoptysis after airway trauma due to malposition of a feeding tube).
DISCUSSION
The present study suggests that a delayed onset of PTP in patients with moderate to severe TBI is associated with an increased risk of developing VTE. This finding is of interest as the timing of PTP is one of the factors directly influenced by the attending physician in the absence of internationally recognized recommendations. Furthermore, ICH progression after initiation of PTP in the study population was a rare event (4%) and only in very few patients (1.7%) did it initiate a therapeutic change, such as an operative intervention or interruption of life-sustaining therapies.
Because many events of VTE occur early after injury [31], any delay of onset of PTP could expose the patient to a major cause of secondary injury after trauma. On the other hand, due to the high risk of ICH progression in patients with TBI-induced coagulopathy, the time of initiation of PTP should be carefully evaluated. Currently, the time of initiation of PTP and the agent of choice for patients with TBI are widely variable due to insufficient evidence to support any recommendations. Therefore, timing, agent of choice, and dose of PTP are based on the physicians’ perceived risk for ICH progression.
The Brain Trauma Foundation suggests either UFH or low-molecular-weight heparin (LMWH) because both are efficacious [32]. A large, multicenter, retrospective study on trauma patients showed that LMWH was associated with a significantly lower rate of PEs compared to UFH [23]. On the other hand, UFH is more easily reversible and thus may be the preferred agent in the case of increased risk of hemorrhagic progression and if anesthesiological intervention or surgery is at stake. For this reason, a continuous infusion of UFH is the preferred approach for patients with TBI at our institution. This approach, however, needs to be discussed critically after this analysis. The percentage of VTE observed in the study population was higher than in previous reports, with a delayed initiation of PTP being the only significant risk factor for VTE identified. Previous studies, in fact, described a lower rate of VTE after earlier administration of PTP [22, 33]. In these studies, however, also patients with mild TBI were included, while we considered patients with moderate to severe TBI only. In a similar population of patients with severe TBI, an even higher rate of VTE of 19% was reported [3]. In that study, however, and in contrast to our study, routine VTE screening was performed. Consequently, comparison of VTE prevalence data among studies is difficult due to the different severity of TBI and the different diagnostic VTE procedure.
Overall, the frequency of relevant ICH progression after initiation of PTP was low. We are not able to distinguish whether this progression was a direct consequence of the initiation of PTP or independent of the onset of PTP. In any case, a therapeutic impact, such as an operative intervention or interruption of life-sustaining therapy, was initiated in a very low proportion of patients, so the use of UFH seems to be safe in this context. In addition, Kwiatt found that ICH progression occurred with a similar frequency in patients who received PTP within 48 hours after trauma, between 48 hours and seven days after trauma, and more than a week after trauma, suggesting that the administration of PTP does not seem to change the natural course of TBI [19].
Our findings correspond to and complement the few previous studies in patients with moderate and severe head injury. In accordance with the report of Kim et al. [34], we support that UFH-based PTP is safe. However, we also confirm that efficiency of UFH-based PTP remains a concern, as we could not demonstrate a reduced incidence of VTE, compared to previous studies with different agents.
Preventing secondary brain injury in patients who are already at a high risk of death and disability due to the primary injury should be the therapy goal of TBI management. So far, a standardized approach for PTP in patients with TBI does not exist.
Our study has several limitations. Firstly, the retrospective nature of our single-center experience limits the generalization of our results. Secondly, the initiation of PTP was an interdisciplinary consensus of neurosurgeons, trauma surgeons and ICU physicians – but not based on a specific protocol. In fact, our results highlight the need for a critical discussion of our practice. It seems, in the decision-making process regarding the initiation of PTP, that we tend to overestimate the risk of ICH progression and to underestimate the risk of VTE in patients with moderate and severe TBI. This leads to delayed PTP. Thirdly, the true incidence of VTE may be underestimated in this study, because extremity ultrasonography and CT scans were only performed in the case of clinical suspicion of VTE and not routinely. With only 23 events, we are not able to insert all relevant independent variables into the same multivariable logistic regression model. The small number of VTE events, in fact, precluded the inclusion in our final multivariable regression model of potentially interesting additional variables. Furthermore, with only seven ICH progressions we were unable to build any predictive model of their occurrence. Fourthly, head CT scans to detect ICH progression after initiation of PTP were not performed systematically for all patients, but rather when clinically indicated in the case of neurological deterioration or if a neurological improvement was not observed. Consequently, the rate of ICH progression may be underestimated. Finally, patients with moderate and severe TBI were analyzed together due to the small sample size, although the risk of a VTE event and of TBI-related coagulopathy increases with the severity of TBI.
As strengths of this study, we included all consecutive patients with moderate to severe TBI admitted to the ICU for at least 48 hours, thus protecting against selection bias. In addition, differently from several previous studies, we considered only patients with moderate to severe TBI to better characterize this specific population, which has a higher risk of VTE and ICH progression due to the severity of the injury, the immobilization, and the worse prognosis. Furthermore, despite the small number of VTE events reported, we constructed all possible “solid” multivariable models and we found in all of them that a delayed PTP initiation remains statistically significant. This finding encourages considering critically in the decision-making process regarding the initiation of PTP the increased risk of VTE associated with a delayed initiation.
A delayed onset of PTP exposes the patients with TBI to increased risk of VTE. The risk of ICH progression seems to be a major concern for initiation of PTP. Based on our data, it seems to be less threatening than the risk of VTE. Due to the relevance of the topic and the lack of evidence to support any strategy for PTP, larger studies are required to address the timing, efficacy, safety, dose, and agent of choice for PTP in patients with TBI, ideally as randomized controlled trials.




