Introduction
Polycystic ovary syndrome (PCOS) is the most common endocrinological disorder associated with infertility in women of reproductive age. Moreover, it can have an impact on immune, metabolic, and cardiovascular health. The psychological aspects of PCOS, which were underestimated for a long time, cover the occurrence of anxiety and depression [1]. Despite a large amount of research, the pathomechanism of PCOS is still unknown. The genetic, epigenetic, and environmental factors are considered to have an impact on PCOS pathophysiology. Guney et al. investigated the correlation between elevated endotrophin concentrations in women with polycystic ovary syndrome. In PCOS, adipose tissue exhibits dysregulated adipokine secretion, potentially impacting the endocrine and reproductive systems negatively. The authors propose that endotrophin contributes to an increase in proinflammatory cytokine levels, leading to insulin resistance, a recognised significant factor in the development of PCOS [2]. Additionally, research by Bahceci et al. highlights elevated levels of X-box binding protein-1 in PCOS women. X-box binding protein-1 plays a crucial role in glucose metabolism and lipid regulation, serving as a valuable marker for metabolic syndrome and diabetes [3]. Furthermore, multiple studies have confirmed associations between kisspeptin levels and PCOS [4]. Kisspeptin is identified as an effective stimulator of pulsatile gonadotropin-releasing hormone release, which predominantly influences luteinising hormone (LH) levels. Elevated LH pulsatility contributes to increased thecal androgen secretion, leading to anovulation, irregular menstrual cycles, and excessive androgen release.
In the recent available data, there are studies suggesting a significant correlation between ovarian sympathetic nervous system (SNS) hyperactivity and its association with PCOS pathogenesis. Increased serum androgen levels, anovulation, and irregular menstruation are all linked to SNS hyperactivity [5]. Moreover visceral adiposity is linked to chronic SNS hyperactivity [6, 7]. It is important to emphasise that anxiety and depression are now significant issues that PCOS-affected women have to deal with. As we mentioned, it is important to emphasise the connection between anxiety and SNS activation in PCOS women [8].
Lansdown et al. showed that SNS hyperactivity not only promotes PCOS pathogenesis but may also contribute to increasing the risk of developing cardiovascular diseases [9]. The very interesting results such as the improvement of sympathetic tone of the autonomic nervous system (ANS), diminished androgen levels, and the restoration of ovulation were confirmed by Linares et al. after cutting the superior ovarian nerve in an experimental model of rats [10]. The authors of this research highlight the role of the SNS in the pathomechanism and clinical manifestation of PCOS.
For infertile women with PCOS to receive effective treatment, a holistic, interdisciplinary approach is necessary. Pharmacological treatment can be efficient, but its application is frequently restricted because it may not be appropriate for some patients or result in side effects. Finding alternative, efficient, secure, and risk-free treatments for PCOS in women is currently the main goal of the therapy.
Among the alternative treatments available, it has been confirmed that osteopathic manipulative treatment (OMT) has a positive influence on a variety of diseases including back pain, pneumonia, stress, and depression. According to Davis et al., manipulating Chapman points and visceral reflexes improves sympathetic tone and relieves the signs and symptoms of sympathetic hyperactivity. Thus, the authors suggest that physical therapy may be an alternative treatment for PCOS women [11]. Additionally low-grade inflammation is a recognised factor causing metabolic disorders in PCOS. Osteopathic manipulative treatment through activation of the cholinergic anti-inflammatory pathway can inhibit the production of proinflammatory cytokines (TNF, IL-1β, TNF-β) [12, 13].
Moreover, it was confirmed that lifestyle modification and other nonpharmacological therapies have a positive influence on women’s health and can improve their quality of life. Given the prevalence of sympathetic hyperactivity and its metabolic effects in PCOS-affected women, OMT may be a beneficial treatment [14].
There are very few data analysing the influence of OMT on infertility management among PCOS women. In the presented study we assessed the impact of OMT on infertility treatment and pregnancy development among fertility failure PCOS women.
Material and methods
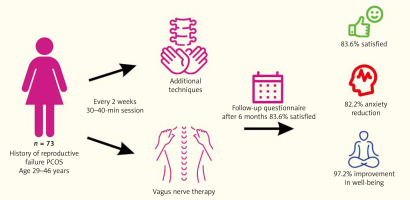
A retrospective analysis was performed at the Department of Gynaecology and Oncological Gynaecology of the Military Institute of Medicine and the m-Clinic Private Practice in Warsaw, Poland. The Military Ethics Committee in Warsaw approved the study. Each patient signed an informed consent form before entering the study and was made aware of the goal of the research. Procedures for physiotherapy were carried out at the Fimedica Clinic in Warsaw, Poland. Seventy-three of the women of reproductive age (29–46 years old) with PCOS, who had received physiotherapeutic care and had a history of fertility failures (recurrent pregnancy loss or recurrent implantation failures), were assessed. Polycystic ovary syndrome was diagnosed according to the Rotterdam criteria [15].
Based on the European Society of Human Reproduction guidelines, we diagnosed recurrent pregnancy loss and recurrent implantation failure [16]. According to the medical documentation, each of the women received personalised treatment depending on the presented abnormalities related to infertility. Patient demographics, physical examination, and test result data were obtained from the medical documentation. For our study we only included women currently trying to get pregnant. Therefore, patients undergoing contraception or menopausal therapy were excluded from the analysis.
Physiotherapy, which involved specific osteopathic techniques, was recommended as an additional therapeutic element, to improve the functioning of the ANS and to restore the balance between its sympathetic and parasympathetic parts. The duration of a single session was 30–40 minutes, and the frequency of visits was once every 2 weeks. Application of pressure at the tissue’s elastic limit for at least 30 seconds (typically 90–120 seconds) as part of musculofascial relaxation techniques led to a visibly felt decrease in soft tissue tension. The physiotherapist followed this relaxation technique by applying pressure at the newly achieved elastic limit. After a series of such compressions, the elasticity of the soft tissues increased [17].
The therapy was carried out according to the following scheme:
Vagus nerve therapy (fascial technique of the anterior aspect of the neck; CV4 technique, fascial release technique for the upper chest opening, mediastinal fascial release technique, the technique of musculofascial release of the diaphragm);
Additional techniques (rib lifting technique, craniosacral system balancing techniques, mobilisation techniques for the cervical, thoracic, and lumbar spine, techniques of musculofascial release of the shoulder girdle, musculofascial release techniques for the pelvic floor, and the muscles of the external rotator group of the hip joint).
Six months after the end of a physiotherapy session we performed a survey containing questions evaluating the course of physiotherapy treatment and its impact on the patients’ well-being, general condition, and the influence on infertility treatment.
Collected data were presented using descriptive methods. Absolute and relative frequency were reported for categorical variables. For continuous variables, at first, their distribution was tested for normality using the Shapiro-Wilk test, then the arithmetic mean with standard deviation and minimum–maximum range were reported for variables with normal distribution, and the median with the 25th and 75th percentiles (Q1 and Q3) and minimum-maximum range were provided otherwise. To compare the distribution of categorical variables between groups, the exact Fisher test or, if the expected size of each cell of the contingency table was larger than 5, the χ2 test was used. To compare the distribution of continuous variables between groups, for normally distributed variables Student’s t-test was used to compare 2 groups, and the ANOVA test was used to compare more than 2 groups, otherwise the Mann-Whitney U test and Kruskal-Wallis test was used, to compare 2 and more than 2 groups, respectively.
Univariable logistic regression models were used to assess the effect of specific parameters on the odds for positive results of an assessment of the effects of physiotherapy on general health and infertility treatment; odds ratio (OR) with 95% confidence intervals and Wald’s test p-values were reported. The level of statistical significance was set at 0.05. Two-sided tests were used.
Results
Population characteristics of the study
The clinical and demographic information about the study’s female subjects, as gathered from surveys, history interviews, medical records, and clinical examinations, are shown in Table 1.
Table 1
Descriptive characteristics of the polycystic ovary syndrome group
The analysis of the obtained results showed that the most common indication for physiotherapy in the study group was a pain in the area of the pelvis and the lumbar spine (60.3%); increased pelvic girdle muscle tone (13.7%), immunological abnormalities diagnosed during infertility diagnostics (20.5%), and infertility of unknown aetiology (5.5%). Physiotherapy procedures in 16 women (21.9%) were part of the treatment of infertility, while in the vast majority of procedures (n = 57) the women’s participation (78.1%) continued and also took place during pregnancy. In the group of women who underwent procedures during pregnancy, 32 (56.1%) pregnancies ended in caesarean section due to medical indications, while only n = 1 pregnancies ended in miscarriage. The other women gave birth by vaginal delivery. Analysis of the available data showed that each woman underwent on average 7 (IQR 4–10) therapy sessions during the entire treatment process.
The assessment of compliance during the physiotherapeutic session
The analysis showed that the vast majority of women were satisfied with the therapeutic process, and that the sessions met the patient’s expectations. As many as 68 (93.1%) women declared that they had been thoroughly informed about the course of therapeutic sessions, while 67 (93%) received very detailed information about the indications for the procedure and the essence of their own ailments. All women confirmed that they had the opportunity to ask questions. N = 43 women (68.2%) received information on potential risks and side effects resulting from the performed procedures. Most women (81%) confirmed that they received recommendations on how to prevent the recurrence of symptoms and how to deal with them if they reappeared.
Evaluation of the effectiveness of the therapy process: psychological and medical aspects
The positive effects of physiotherapy treatments on general health were confirmed by 61 (83.6%) of the study’s female participants. Lowering the degree of anxiety brought on by infertility treatment and pregnancy was declared by 60 (82.2%) women; improvement in well-being was declared by 72 (97.3%), and increasing awareness of one’s own body after physiotherapy sessions was declared by 70 (95.9%) women. In the aspect of infertility treatment, 59 (80.8%) women believed that physiotherapy had had a positive impact on the diagnostic and therapeutic process; the remaining women indicated no such relationship. Physiotherapy had no unfavourable effects, according all the women. When analysing the group of surveyed women, no statistically significant correlations were observed between the effectiveness of the therapy process, in the psychological and medical aspects, and the patient’s age. The effect of physiotherapy on the infertility treatment process is, however, perceived more favourably by younger patients. Logistic regression analysis is presented in Table 2.
Table 2
The physiotherapy influence on infertility treatment according to women’s age
| Parameters | OR |
|---|---|
| Age | 0.863 |
The results of the analysis also suggest no significant relationship between the number of obstetric failures in the history and the effectiveness of physiotherapy in terms of infertility treatment and positive impact on general health (Table 3).
Table 3
The influence of physiotherapy according to the number of pregnancy losses
| Parameters | OR | 95% CI | p-value |
|---|---|---|---|
| Infertility treatment | 1.07 | 0.693–1.73 | 0.773 |
| General health | 0.908 | 0.579–1.47 | 0.677 |
Analysing the indications of physiotherapy treatments, a statistically significant (p = 0.029) positive effect on general health and improvement of well-being was obtained in the group of women with pain in the area of the pelvis and the lumbar spine. In addition, a statistically significant improvement in well-being was also observed in women who had a greater number (10–30) of therapeutic sessions (p = 0.04). Figure 1 presents the results of our study.
Discussion
Polycystic ovary syndrome affects approximately 10% of women of reproductive age, predominantly leading to difficulties in conception and an elevated risk of miscarriages and implantation failures. The symptomatology of the syndrome is notably diverse, ranging from discrete symptoms to complex endocrine, metabolic, and immunological disorders in patients [18]. The causes of fertility disorders in this group of women are intricate. In addition to the mentioned abnormalities, there are observed significantly higher homocysteine levels, contributing to increased oxidative stress. This elevated homocysteine level has embryotoxic effects, induces hormonal disorders, and results in changes in DNA methylation patterns. Furthermore, it leads to vascular endothelial damage, thereby amplifying the susceptibility to cardiovascular diseases among these women [19]. Moreover, Rshoud et al. confirmed a higher incidence of endometrial pathologies, particularly uterine polyps, in this group of women. Uterine polyps are identified as a significant factor contributing to implantation disorders in individuals with PCOS [20].
Genetic predisposition, insulin resistance, hyperandrogenism, and recently also the presence of increased activity of the SNS are predisposing factors for PCOS. Heider et al. observed higher amounts of catecholaminergic fibres in ovaries among PCOS women [21]. Based on this research the authors hypothesised the influence of the ANS on polycystic ovaries morphology and PCOS pathophysiology [9]. Moreover, there are data that confirm the association of SNS with metabolic abnormalities such as insulin resistance and obesity [6, 22]. The influence of ANS on the immunological system was also confirmed. Lee et al. suggested that the synchronisation of cellular metabolic processes and the regulation of the local immune response are under the control of SNS [23]. It is well known that in PCOS excess production of pro-inflammatory mediators and subsequent chronic inflammation exists [24]. In addition, many studies confirmed the influence of chronic low-grade inflammation on hyperandrogenism, insulin resistance, and fertility failures in PCOS women [25, 26]. Numerous abnormalities observed in PCOS mean that the treatment of infertility or recurrent miscarriages in this group of women is usually multidirectional and requires a lot of experience. However, the treatment of disorders associated with PCOS that have been used so far are often associated with the occurrence of side effects and remain ineffective. New, alternative methods of treatment are sought after to avoid side effects and reduce the stress associated with infertility treatment.
In our study, we presented the possibility of including physiotherapy as an additional, complementary form of treatment, especially in women with pelvic pain, low back pain, and immune disorders that could be the cause of reproductive failure. It is also worth noting that in the analysed group, most women finally became pregnant and gave birth to a healthy child, which may suggest a measurable, positive effect of the applied treatment regimen. According to the literature, based on the experimental model, it was confirmed that OMT changes the parameters of the immune system. Mechanical stimulus exerted on tissue during an OMT session leads to immune modification, which reflects their mobilisation rather than synthesis during the time of treatment session. Crane et al. confirmed the positive influence of OMT on the immune system by reducing the level of pro-inflammatory cytokines, such as TNF-β (tumour necrosis factor-β) or IL-6 (interleukin-6). Based on the presented results, the authors of the study suggest a potential positive effect of OMT on the therapy of women with PCOS [27]. Similar results on immunological milieu were presented by Walkowski et al., who observed, after the OMT session, a lower level of dendritic cells, which are normally involved in inflammatory responses. Taking this data into account, incorporating OMT as an additional treatment option may bring great benefit for the patients and improve the infertility treatment results [28]. Moreover, this therapy has a positive influence on the immunological milieu especially in PCOS women, which seems to be very important during infertility treatment.
Our results also confirmed the positive impact of OMT therapy sessions on the overall process of infertility treatment in PCOS women. It was observed that increased SNS activity may contribute to a higher prevalence of anxiety and depression in PCOS women, which is commonly observed in this group [29]. The osteopathic manipulative treatment therapy sessions restore the balance between the sympathetic and parasympathetic systems. Concerning the above, it can be concluded that the infertility treatment process including OMT may have a positive impact on general well-being and significantly improve quality of life. The positive psychological aspect of improving well-being seems to be extremely important, especially in infertility treatment. In our study, most women observed a lower level of anxiety, improvement in well-being, and increased awareness of their bodies after physiotherapy sessions.
Moreover, it is worth knowing that in normal pregnant women, changes in the activity of the ANS and the association of these activities with pregnancy-related anxiety have been observed. It was confirmed that during a healthy pregnancy, the physiological decrease in ANS activity occurs normally in the second trimester of pregnancy [30, 31]. The very interesting data were presented by Mizuno et al., which showed that in the second trimester of pregnancy anxiety or depression was associated with increased SNS activity [32].
During normal pregnancy the adaptation of haemodynamic changes in the cardiovascular system also depends on the balance in ANS activity. Taitubayeva et al. describe disturbance in the ANS and their influence on the risk of pregnancy-induced hypertension [30]. Moreover, the autonomic dysfunction syndrome may result in various peripartum complications.
Improvement in sympathetic tone was an important issue in alternative medicine especially in PCOS women. It was confirmed that pregnant women who participated in meditation (mindfulness) sessions experienced a reduction in perceived levels of emotional stress before and after birth [33]. The data cited above suggest that mindfulness levels during pregnancy are associated with changes in ANS activity, which are likely to be adaptive. Similar findings come from a study by Muthukrishnan et al., who proved that training of awareness improves the parasympathetic function of ANS [34].
Effects of osteopathic techniques on the autonomic nervous system
From the available methods for ANS therapy, osteopathic techniques through various manual interventions are useful in improving the whole body function and restoring its homeostasis.
The most common osteopathic techniques are high-velocity low-amplitude (HVLA), soft tissue techniques (myofascial release), and visceral or craniosacral techniques. The impact of manipulative techniques performed in the cervical and thoracic spine on ANS activity was confirmed in the available literature [35–37].
Also Rechberger et al., after the analysis of 40 articles, confirmed a positive impact of osteopathic procedures on ANS activity [38]. Taking this data into account, ANS must be positively impacted especially by HVLA procedures used in the lumbar spine. According to the cranial methods, the data are still ambiguous. Only suboccipital soft tissue decompression techniques performed in the suboccipital region show a significant effect on the improvement of the function of the parasympathetic part of ANS and the vagus nerve [39]. It is clear from this review that while a positive effect of osteopathic techniques on ANS function can be assumed, the issue is still too rarely addressed, and studies lack uniform research standards, which affects their methodological quality. Studies on larger groups of subjects are recommended.
Several strengths characterised our study, enhancing its validity and reliability: well-defined inclusion and exclusion criteria; the retrospective qualitative design allows for an in-depth exploration of the experiences of women in fertility therapy and the specific issue of holistic infertility therapy in PCOS women. Despite its strengths, our study is not without limitations: a small study group; the survey nature of the study inherently introduces a subjective element, and as a result, the findings may be nuanced and challenging to interpret definitively.