Introduction
Amiodarone is a type 3 anti-arrhythmic agent that is widely used and has many toxic effects on different tissue functions, including hepatic toxicity, steatosis, gastrointestinal upset, hepatitis, bradycardia, liver and pancreas fibrosis. It was shown that amiodarone has cytotoxic effects on different cell types in cell culture studies. It is most extensively prescribed for the benefits as a drug used in atrial fibrillation (AF). Amiodarone can cause many adverse effects especially on the liver [1]. Many studies have concluded that amiodarone generates free radicals in vitro and in vivo which are the main causes of oxidative stress [2, 3]. Oxidative stress manifests by producing reactive oxygen species (ROS) in the cell [4]. Superoxide anion is a reactive and superoxide dismutase that converts superoxide to hydrogen peroxide, which is less reactive than oxygen anion. Glutathione peroxidase detoxifies hydrogen peroxide to water [4]. Reduced glutathione (GSH) regulates the activity of glutathione peroxidase by giving hydrogen. Therefore, GSH is considered as a detoxifying molecule. Glutathione is present mostly in reduced form (GSH) in the cell. It can be oxidized to GSSG (oxidized glutathione), which is approximately 1% of total glutathione in the cell in normal conditions [5]. GSH has an important role in detoxification and it can help regenerating some antioxidants such as vita-min C and E, which are also capable of detoxifying hydrogen peroxide [4]. In addition to them, N-acetyl cysteine (NAC) is another antioxidant molecule. It is a cys residue with acetylation and in this way it becomes a part of the cysteine pool in the cell to maintain the biosynthesis of GSH [6]. Deficiency of GSH or the ratio of GSH/GSSG could be a marker for several pathological problems [5]. As a result of AF, in myocardial tissues, high levels of superoxide and hydrogen peroxide are observed [7–10]. Also, as an oxidative stress marker, the ratio of GSSG to GSH (reduced glutathione) was found increased in blood samples of patients with AF [11]. Both AF and using amiodarone as a drug for AF can be obvious causes of increased ROS and consequently change of total concentration of GSH in cells and related tissues. Many studies have shown that using antioxidants such as NAC, vitamin C and vitamin E could be helpful to minimize the effects of oxidative stress in the cell [3]. With this information, using antioxidants could reduce oxidative stress, which is a result of amiodarone as a drug for AF.
Recently, especially amiodarone’s toxic effects on the liver have not been well studied and no clinical agents are available for prevention. Oxidative stress has been proved to be the major pathogenic mechanism of amiodarone’s tissue damage effects. From this perspective, the aim of this study is to investigate toxic effects of amiodarone and the prevention potential of NAC, vitamin E and vitamin C in a rat experimental model.
Aim
We studied the level of glutathione in rat liver tissue, following the oral administration of amiodarone and antioxidants.
Material and methods
Agents used
Amiodarone hydrochloride (Cordorone, 200 mg, Sanofi-Aventis, Istanbul, Turkey), vitamin E (Evicap, 400 I.U., İlaç ve Kimya Sanayi A.Ş., Çerkezköy, Tekirdağ, Turkey), ascorbic acid (Redoxon, 500 mg/tablet, Bayer Türk Kimya San Ltd. Şti., Istanbul, Tukey), and N-acetyl cysteine (NAC, 200 mg/tablet, Bilim İlaç San ve Tic. A.Ş., Gebze, Kocaeli, Turkey) were purchased from a pharmacy. Glutathione Assay Kit (CAS number: CS0260) was obtained from Sigma-Aldrich (St. Louis, MO).
Animals
Thirty Wistar albino male rats were used with the approval of the Animal Care and Use Committee of Hacettepe University (Permit no. 2013/5-10) and carried out in accordance with the National Guidelines for the Use and Care of Laboratory Animals. After all animals were purchased from Animal Laboratory Units of Hacettepe University, they were allowed to become adapted to the conditions of the laboratory for seven days. All animals were housed randomly selected as three animals per cages, using a 12 h light-dark cycle.
Design of the study
All rats were randomly assigned to five groups as follows: control group (untreated group, n = 6), amiodarone treated group (100 mg/kg/day, n = 6), amiodarone + Vit. E treated group (50 mg/kg/day, n = 6), amiodarone + Vit. C treated group (50 mg/kg/day, n = 6), amiodarone + NAC treated group (50 mg/kg/day, n = 6). Amiodarone and all other substance were given orally for 7 days. The amiodarone dose is higher compared to the dose used in human beings due to the fact that the metabolism of rats is higher compared to human beings [12]. All antioxidant agents were given 1 h after administration of amiodarone. All the animals were sacrificed under anesthesia on the 8th day. Liver tissues were taken from the animals, homogenized in 0.9% NaCl and 10% (w/v) homogenates were prepared. Total glutathione was determined in the liver homogenates. Experiments were carried out at the temperatures given in the procedures.
Glutathione assay
The level of total glutathione (GSH and GSSG) in the cell was measured using the Glutathione Assay Kit (Sigma-Aldrich, USA) according to the manufacturer’s instructions. Liver samples was firstly deproteinized with 5% 5-sulfosalicylic acid solution and then centrifuged to remove the precipitated protein. Supernatants of the samples were used for the determination of the glutathione according to the manufacturer’s protocol. Samples were read with kinetically at 1 min intervals for 5 min at 412 nm using an ELISA reader (EZ Read 400 Micro-plate Reader, Biochrom).
Statistical analysis
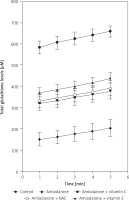
Data are presented as mean ± SD in Table I and mean ± SEM in Figure 1. Differences between groups were analyzed using analysis of variance (ANOVA) for triplicate measurements. The level of significance was taken as p < 0.05 in all instances. GraphPad Prism 5.01 for Windows (GraphPad Software) was used for statistical analysis. In cases of significance, Bonferroni adjusted Mann-Whitney U test was used to determine the efficient group.
Table I
Total glutathione data of all groups (μM)
| t [min] | Control | Amiodarone | P-value** | Amiodarone + Vit. C | Amiodarone + NAC | Amiodarone + Vit. E |
|---|---|---|---|---|---|---|
| 1 | 584.2 ±91.2 | 150.5 ±87.1 | < 0.0001 | 321.4 ±126.1 p*** < 0.0001 | 332.8 ±118.7 p*** < 0.0001 | 372.7 ±92.0 p*** < 0.0001 |
| 2 | 607.7 ±91.4 | 162.2 ±96.3 | < 0.0001 | 334.3 ±134.1 p*** < 0.0001 | 345.7 ±125.6 p*** < 0.0001 | 386.8 ±95.2 p*** < 0.0001 |
| 3 | 627.0 ±88.2 | 176.3 ±104.7 | < 0.0001 | 347.7 ±142.7 p*** < 0.0001 | 360.1 ±132.8 p*** < 0.0001 | 402.3 ±99.2 p*** < 0.0001 |
| 4 | 644.3 ±83.6 | 188.8 ±113.8 | < 0.0001 | 363.6 ±151.5 p*** < 0.0001 | 375.5 ±140.0 p*** < 0.0001 | 419.7 ±104.4 p*** < 0.0001 |
| 5 | 659.4 ±77.4 | 205.4 ±122.5 | < 0.0001 | 380.0 ±157.7 p*** < 0.0001 | 392.8 ±147.4 p*** < 0.0001 | 440.6 ±109.3 p*** < 0.0001 |
| P-value* | < 0.0001 | < 0.0001 | < 0.0001 | < 0.0001 | < 0.0001 |
Results
Total glutathione amount of the liver samples of rats was measured spectrophotometrically with kinetic read at 1-minute intervals for 5 minutes. When the groups were examined individually there was a significant increase in glutathione levels with time (p < 0.0001) (Table I). When separate time intervals were compared, the amiodarone treated group had the lowest levels compared to control and each antioxidant co-administered group in all time intervals (p < 0.0001), which may demonstrate the protective potential of antioxidants (Fig. 1). When the antioxidants were compared, there was no difference between amiodarone + NAC, amiodarone + Vit. C and amiodarone + Vit. E (p > 0.05 for each).
Discussion
Hepatic injury is a common manifestation of drug toxicity and may be reflected as different clinical features [13]. Mitochondrial dysfunction is one of these features and clinical studies scrutinizing mitochondrial dysfunction came a long way for decades [14]. Mitochondrial dysfunction manifests itself in different ways such as membrane permeabilization, oxidative phosphorylation impairment, inhibition of fatty acid oxidation and mitochondrial DNA depletion [14]. Within the cell, mitochondria are the source of ROS production through the mitochondrial respiratory chain. In this respiratory chain, electrons enter the system and directly react with oxygen to generate superoxide anion radical. Then this radical is converted to hydrogen peroxide (H2O2) by superoxide dismutase. Hydrogen peroxide is detoxified to water by glutathione peroxidase which uses reduced glutathione (GSH). In healthy tissues, all reactive oxygen species are detoxified properly by mitochondrial systems. However, in different pathophysiological situations, GSH depletion was seen in liver mitochondria, which reduce the capacity of H2O2 detoxification. Hydrogen peroxide accumulation causes mitochondrial dysfunction. Amiodarone can also induce hepatotoxicity due to the mitochondrial dysfunction [15–18]. Also, many studies have documented that it has long-term effects on different tissues. After discontinuation of the drug, the effects of amiodarone were still detected in patients’ different tissues including liver, lung and adipose tissue [19]. In our study, we measured total glutathione level (total GSSG and GSH in μM) of rat livers which were administered with amiodarone. Compared to the control group (which has high levels of glutathione), the amiodarone administered group showed dramatically reduced glutathione levels at each time interval, which shows the reduced detoxification capacity of mitochondria. On the other hand, it was shown that treatment with antioxidants such as vitamin C, vitamin E and NAC has preventive effects on reduced detoxification capacity of mitochondria [20–23]. Both cell culture studies and animal model experiments on amiodarone in relation to several antioxidants show similar beneficial effects of antioxidants to reduce toxicity of amiodarone [24–27]. These antioxidants have similar mechanisms in antioxidant action via scavenging ROS [28, 29]. Also, NAC has importance for production of glutathione in the cell because its cysteine part together with glutamate and glycine is a precursor of glutathione [30]. Glutathione is already an antioxidant in the cell; therefore, NAC administration has a kind of backup. To analyze the antioxidant effects of vitamin C, vitamin E and NAC on amiodarone toxicity, we treated rats with both amiodarone and these antioxidants separately as described above. All antioxidants that we used showed increased glutathione levels when they were coadministered with amiodarone compared to the reduced glutathione level of the amiodarone group. They could not produce glutathione like in the control group but when compared to the effect of amiodarone only, we can say that antioxidant usage is successful in producing glutathione in the cell. The group receiving vitamin E coadministered with amiodarone had the highest level of glutathione, followed by the NAC + amiodarone group and Vit. C + amiodarone group. It is already known that separate treatments of NAC, vitamin C and vitamin E on cultured cells have antioxidant effects [3].
Conclusions
Our results are important in terms of understanding the toxic effects of amiodarone and its prevention via use of antioxidants. In our previous study, we demonstrated the decrease of amiodarone cytotoxicity using antioxidants in cell culture [3]. Now, we showed the toxicity reducing capacity of antioxidants in animal experiments. To make a comprehensive conclusion, further analysis such as separate measurement of GSSG and GSH amounts, glutathione peroxidase enzyme activity and measurement of lipid peroxidation products could be supportive. In addition, we believe that this study is reproducible, and clinical studies may be performed based on these findings.