Summary
Little is known about factors that determine enrolment (vs. non-enrolment) in chronic ischaemic heart failure (CIHF) interventional randomized controlled trials (iRCT). In this study, out of 5,473 analysed hospitalized patients, 526 with CIHF were selected and clinical characteristics and medical therapy were compared in eligible-and-enrolled (E-E) vs. eligible-but-not-enrolled (E-NE) patients in CIHF myocardial regeneration iRCTs. CIHF medical management was similar in E-E and E-NE while ineligibility resulted mainly from recent/planned intervention outside iRCT. LV impairment was more severe in E-E patients, consistent with higher-risk patient enrolment in CIHF-iRCTs. This contrasts with typical lower-risk patient enrolment in other cardiovascular RCT types and populations.
Introduction
Ischaemic heart disease is a leading cause of heart failure [1]. With improved survival of myocardial infarction, increasing prevalence of obesity and diabetes, and ageing populations, the burden of cardiovascular diseases, including heart failure, will be increasing rather than decreasing over the next decades [2]. Management of chronic ischaemic heart failure (CIHF) remains a fundamental challenge in contemporary cardiology despite unquestionable progress in pharmacotherapy. Even with recent reduction in CIHF hospitalizations, in-hospital and post-discharge mortality remains high [3]. Five-year mortality of CIHF patients with reduced ejection fraction (EF) is the highest among all known aetiologies of heart failure [1].
Pharmacological therapies or implantable devices may improve the clinical course and prognosis but they do not affect the cause of CIHF – loss of viable myocardial tissue [3]. Myocardial regenerative therapy, by addressing this gap, is arising as a novel therapeutic strategy for CIHF [4, 5]. Myocardial regenerative therapy, using different types of cell-based products and different delivery methods, may counteract the cause of the disease [4, 5]. Randomized controlled clinical trials (RCTs) are mandatory to critically evaluate these effects [4, 5].
RCTs are central to evidence-based medicine. By providing data assumed to be of the highest level of evidence, RCTs have a critical impact on clinical management guidelines [3]. However, factors beyond intervention may contribute to the measured effect. In routine clinical practice, the RCT-derived treatment effect may be enhanced or reduced [6, 7]. A fundamental concern over RCTs’ impact on clinical decision-making today is their generalizability [8, 9]. One fundamental source of bias may arise from major differences between RCT populations and real-life patient cohorts [8, 9]. Another source of bias may result from performing the trial intervention in the cohort of patients with a low level of indication for the trial-tested intervention (maintaining the essential principle of randomization – principle of uncertainty) while other patients (for whom randomization might lead, in the opinion of the investigator, to harm) receive the trial-tested intervention outside the trial [7]. Such trials may lead to erroneous modifications of guidelines because of widespread belief in the “randomized” label [7].
RCT recruitment of patients representative of real-life clinical populations is fundamentally required for the trial data applicability to everyday practice [6]. Today, little is known about eligibility and enrolment in CIHF interventional RCTs (iRCTs) [4, 10].
Aim
The aim of the study was to compare clinical characteristics and medical therapy in eligible-and-enrolled (E-E) vs. eligible-but-not-enrolled (E-NE) patients in CIHF myocardial regeneration iRCTs.
Material and methods
We evaluated clinical characteristics and medical therapy in E-E vs. E-NE CIHF patients in randomized clinical trials of myocardial regeneration. Patient data were derived from consecutive participants of two cell therapy iRCTs that required patient hospitalization as well as from consecutive hospitalized CIHF patients outside the iRCT recruitment periods. The trials were conducted in the Department of Cardiac and Vascular Diseases, Jagiellonian University Medical College, John Paul II Hospital, Krakow, Poland, as per national regulations and Ethics Committee approvals.
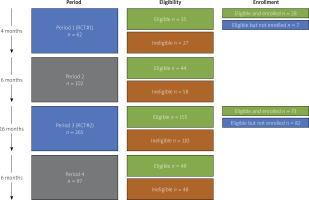
The recruitment period of the first trial (iRCT#1) was 4 months (Period P1) while for the second one (iRCT#2) it was 16 months (Period P3). For the purpose of the present analysis, two non-recruitment periods were established – one between the trials (Period P2) and the other one after the second trial (Period P4, Figures 1, 2). Both non-recruitment periods (P2 and P4) were 6 months long.
Figure 1
Distribution of patients hospitalized in RCT periods (Periods 1 and 3; A) and non-RCT (Periods 2 and 4; B)
CHF – congestive heart failure, RCT – randomized controlled trials.
The study was registered with the local Ethics Committee (OIL/KBL/110/2021). With pseudonymized patient records used in the setting of a retrospective analysis, no separate patient consent was required.
Our database of consecutive hospitalizations during periods P1-P4 was searched and patients with heart failure were identified. Detailed data were captured for those with heart failure defined as a persistent (≥ 3 months) left ventricle systolic dysfunction (EF ≤ 45% by echocardiography) in absence of potentially reversible causes of lowered EF such as acute myocarditis, toxic or tachycardia-induced cardiomyopathy. Of those, patients with an ischaemic cause were selected. CIHF definition was according to P1 and P3 iRCTs; it included history of myocardial infarction and/or presence of coronary artery stenosis greater than 50%, history of percutaneous coronary intervention (PCI) or coronary artery bypass grafting (CABG).
CIHF patient inclusion and exclusion criteria were similar for RCT#1 and RCT#2. Inclusion criteria were the following: patient age 18–80 years, NYHA Class II–IV, clinically stable HF for the past 3 months, more than 3 months since any intervention that could affect EF (cardiac resynchronization therapy (CRT), acute coronary syndrome (ACS), PCI, CABG), execution of complete (if feasible) myocardial revascularization.
Exclusion criteria involved planned cardiac surgery for co-existing severe valvular heart disease, left ventricle (LV) aneurysmectomy or other cardiac surgery, tumour or neoplasm history, undetermined coronary arterial status, planned non-cardiac major surgery, heart transplant waiting list, moderate to severe immunodeficiency or chronic immunosuppressive therapy, severe acute or chronic infections, current hyperthyroidism, coagulopathies, pregnancy or breast feeding, alcohol or drug abuse, substantial soft tissue disease, severe chronic kidney disease (eGFR < 20 ml/min), contrast allergy, body mass index (BMI) < 18 kg/m2 or > 45 kg/m2, non-compliance with study procedures (e.g. lack of effective arterial access) or life expectancy < 12 months.
Trial eligibility was individually assessed. Enrolment was captured for P1 and P3. Eligible patients in P1 and P3 were divided based on their actual enrolment in the trials: E-E and E-NE. Reasons for non-enrolment were recorded.
Clinical characteristics and medical therapy in E-E patients were compared to (1) E-NE patients from the iRCT period and to (2) eligible patients from the non-iRCT period (eligible-but-enrolment-not-applicable – E-NAE). Moreover, trial-eligible patients in iRCT periods (enrolment-applicable patients; E-NE plus E-E) were compared to (3) trial-eligible patients in non-iRCT recruitment periods (E-NAE). Furthermore, E-E patients were compared to (4) patients who refused participation in iRCTs.
Statistical analysis
The frequencies of qualitative variables were presented as percentages and compared using the χ2 test of proportions for categorical variables with Yates’ correction as applicable. Quantitative variables were evaluated for distribution using the Shapiro-Wilk test or Kolmogorov-Smirnov test and were compared with the T-test or Mann-Whitney U test as applicable. Multiple group comparisons were performed using analysis of variance (ANOVA) or the Kruskal-Wallis test as applicable. Statistical analysis was performed using StatSoft Statistica 13.3 software for Windows (StatSoft Inc., Tulsa, OK, USA). P-values of < 0.05 were considered statistically significant.
Results
Overall analysis
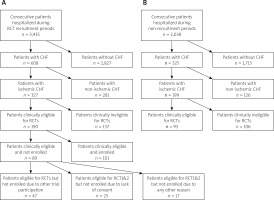
Out of total 5,473 hospitalized patients in Periods P1–P4, 933 (17.0%) had heart failure. CIHF was the dominant type of chronic heart failure (526 patients; 56.4% of the total chronic heart failure cohort undergoing hospitalization). Out of all patients with CIHF, 283 (53.8%) were eligible for iRCTs. Patient distribution in the study periods is given in Figure 1, with iRCT periods reflected in Figure 1 A and non-iRCT periods in Figure 1 B.
Eligibility/ineligibility analysis
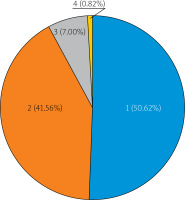
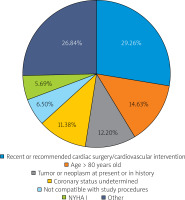
The iRCT eligibility rate was similar between the periods evaluated (P1 = 56.5%, P2 = 43.1%, P3 = 58.5%, P4 = 50.3%, p = 0.08, Figure 2). Analysis of reasons for ineligibility (including the number of reasons and specific cause(s)) is provided in Figures 3 and 4. One in every 2 (50.6%) ineligible patients had a single-only reason for ineligibility (Figure 3). Principal reasons for ineligibility (Figure 4) were recent/planned cardiac intervention outside iRCT (29.3%), age above threshold (14.6%), and coexisting disease as the exclusion criterion (12.2%). Figure 4 presents the distribution of specific reasons for ineligibility (single ineligibility reason patients).
Prevalence and reasons for non-enrolment
More than 1 in every 2 (53.2%) eligible CIHF patients in the recruitment periods were enrolled into cell therapy iRCTs. Reasons for non-enrolment of the eligible patients in the iRCT recruitment periods are shown in Figure 1 A. The most common reason for CIHF iRCT non-enrolment (52.8%) was other trial participation that included trial enrolment in a prior disease stage (43 out of 47 CIHF iRCT clinically eligible but non-enrolled other trial patients were AMI trial participants). A further 28.1% of iRCT-eligible patients were not enrolled due to lack of consent.
Comparison of clinical parameters in iRCT-eligible patient groups
Clinical characteristics and medical therapy of E-E patients, E-NE patients (both during iRCT recruitment periods, P1 and P3) as well as those of eligible (eligible, non-applicable enrolment; E-NAE) patients in non-iRCT periods (P2 and P4) are presented in Table I.
Table I
Clinical characteristics of patients E-E in the trials, and E-NE in the RCT periods (Periods P1 and P3), total eligible patients in RCT periods, total eligible patients in non-RCT periods (Period P2 and P4) and clinically eligible during RCT periods but not enrolled due to lack of consent
[i] RCT – randomized controlled trials, NYHA – New York Heart Association, AMI – acute myocardial infarction, PCI – percutaneous coronary intervention, CABG – coronary artery bypass graft, TIA – transient ischaemic attack, CRT – cardiac resynchronization therapy, ICD – implantable cardioverter-defibrillator, IFG – impaired fasting glucose, IGT – impaired glucose tolerance, PAD – peripheral artery disease, COPD – chronic obstructive lung disease, AF – atrial fibrillation, e-GFR – estimated glomerular filtration rate, EF – ejection fraction, EDV – end diastolic volume.
E-E and E-NE patients were similar with regard to sex (9.9% vs. 15.7% women, p = 0.32), age (64.1 vs. 63.6 years old, p = 0.85), prior myocardial infarction (MI) (80.2% vs. 83.1%, p = 0.6009) and prior CABG (16.8% vs. 12.4%, p = 0.38). Prevalence of ICD (33.7% vs. 7.9%, p < 0.0001), arterial hypertension (AH, 89.1% vs. 70.8%. p = 0.0015), and atrial fibrillation (AF, 31.7% vs. 15.7%, p = 0.0104) was significantly higher in enrolled patients, consistent with higher-risk patient enrolment. Indeed, the E-E patients had a significantly lower EF (31% vs. 34%, Simpson, p = 0.0393) and a higher end diastolic volume (EDV) (186.9 ml vs. 153.7 ml, Teichholz, p < 0.0001). There were no differences in angiotensin-converting-enzyme inhibitors (ACEI)/angiotensin receptor blocker (ARB), β-blocker, diuretic (thiazide, loop, K+ sparing), sacubitril/valsartan and statin therapy between the groups.
The E-E patients compared with iRCT-eligible patients in non-recruitment periods (non-iRCT periods; E-NAE) did not differ in sex (9.9% vs. 10.8%, p = 0.85), age (64.1 vs. 64.9 years old, p = 0.18), prior MI (80.2% vs. 82.8%, p = 0.64), or prior CABG (16.8% vs. 23.7%, p = 0.23), indicating that the differences identified for the recruitment periods were maintained during iRCT non-recruitment. Indeed, there was a difference in EF (31% vs. 35%, p = 0.036) and EDV (186.9 ml vs. 166.6 ml, p = 0.002) but not in prevalence of ACEI/ARB, β-blocker, diuretic (thiazide, loop, K+ sparing), sacubitril/valsartan or statin therapy between the groups (Table II). ICD prevalence (33.7% vs. 19.4%, p = 0.037) was higher in the E-E group than the E-NAE cohort.
Table II
Medical therapy of E-E patients in the trials, and E-NE patients in the RCT periods (Periods P1 and P3), total eligible patients in RCT periods, total eligible patients in non-RCT periods (Period P2 and P4) and clinically eligible during RCT periods but not enrolled due to lack of consent
iRCT-eligible patients who could not be enrolled due to lack of consent (LoC group in Tables I and II), compared to E-E patients had similar age (66.3 vs. 64.1 years old, p = 0.21), sex (16.0% vs. 9.9%, p = 0.61), incidence of prior MI (84.0% vs. 80.2%, p = 0.66) and CABG (28% vs. 16.8%, p = 0.32). However, patients who refused participation in iRCTs had a lower rate of prior PCI (68.0% vs. 87.1%, p = 0.046). No differences were noted in EF (34% vs. 31%, p = 0.367) and pharmacological HF treatment.
There were no differences in clinical characteristics and CIHF medical therapy between eligible patients in iRCT and non-iRCT periods (E-AE vs. E-NAE; Tables I and II).
Discussion
Principal findings from this work, regarding evaluation of eligibility and enrolment in CIHF iRCTs, are as follows:
Over 50% of CIHF patients were eligible for recruitment in iRCTs with trans-endocavitary or transcoronary delivery of cell-based myocardial regeneration therapies/placebo.
In iRCTs trial recruitment periods, the majority of eligible patients were effectively enrolled; exceptions resulted primarily from other (prior) trial participation.
The majority of iRCT-ineligible patients with CIHF had a single reason for non-eligibility. The leading reasons for non-eligibility were recent or planned cardiac/coronary intervention outside the RCT and age above the threshold of 80 years.
The eligible-but-not-enrolled patients had overall less severe left ventricular impairment (parameters – LVEF, EDV), and lower incidence of cardiologic comorbidities (such as AF), while the incidence of other comorbidities was similar to that in eligible-and-enrolled patients.
In this first (to our knowledge) study to evaluate in detail eligibility and enrolment in CIHF iRCTs, we (i) analysed whether clinical characteristics and medical therapy differed between eligible-and-enrolled patients and eligible-but-not-enrolled patients in CIHF iRCTs and (ii) identified fundamental reasons for non-enrolment. Moreover (iii) we evaluated whether the eligible CIHF patient profile differed between iRCT recruitment and non-recruitment periods. Also, (iv) we investigated whether the iRCT-enrolled population was reflective of the trial-designed target population. Finally, we identified several factors that may affect external validity of cell therapy iRCTs.
RCTs are considered the most reputable source of medical evidence [11]. The “evidence” is anticipated to come from well-designed trials that should be conducted according to protocols, with minimized patient selection bias and other bias types [11]. RCTs should exhibit internal and external validity. Internal validity refers to trial-tested intervention. It may be negatively affected by factors that influence the cause-and-effect relationship evaluated in the study, through means other than trial-tested intervention. External validity refers to the extent to which results obtained in a study population can be extrapolated to other populations [6]. To be clinically useful the trial results should be relevant to a definable external group of patients in a particular clinical setting.
Our present work concerns several factors important for external trial validity, including the ratio of randomized patients to eligible-but-non-randomized patients in participating centres, the proportion of patients who declined randomization, baseline clinical characteristics, uniformity of underlying pathology, severity of disease and the stage of its natural history, and comorbidities [6]. Some other factors of potential relevance, particularly those in relation to the trial-specific setting (e.g., healthcare system status, recruitment from primary vs secondary vs tertiary care, selection of participating centres) could not be presently evaluated due to a single centre/single healthcare system being outside the scope of our present analysis.
In cardiology RCTs, a low eligibility rate (which may affect external validity) has been identified as a major obstacle in the study results’ applicability to everyday clinical practice. In a recent overview of large studies, ineligibility varied from 33.6 to 90.6% [12]. In HF RCTs the eligibility rates were observed to be generally even lower. For instance, Constantino et al. [10] found that only 34% of everyday patients from the HF outpatient clinic were eligible to participate in at least one of 16 analysed HF RCTs. Out of 20,388 patients with HF registered in the US National Heart Failure Project, eligibility rates for three HF RCTs of interest ranged from 13% to 25% [13]. In the case of iRCTs under present evaluation a higher proportion, more than one in every 2 patients with CIHF, were eligible for trial participation (Figures 1, 2).
Lack of effective enrolment of eligible patients is an important argument in the discussion of data extrapolation from the RCT population to general clinical practice populations. On average, some leading RCTs in cardiology demonstrated enrolment rates at the level of ~50% (48 to 64% in all-comers designed trials [14, 15]), 58% in pharmacology RCTs [16] and 59% in NSTEMI RCTs [17]). Consistent results were obtained from iRCTs of a complex intervention using a novel cell-based agent in CIHF (53.2% of eligible patients enrolled, Figures 1, 2). Of note, this high enrolment rate in our study was already negatively impacted by the fact that many potential participants were already effectively recruited in other ongoing RCTs (Figure 1). In the present study, the most common reason for CIHF iRCT non-enrolment (52.8%) was other trial participation that included trial enrolment in a prior disease stage.
Reasons for non-enrolment of potentially eligible patients vary depending on specific study requirements. Consent-related reasons, present in 52.5% of non-participants, were the main cause of non-enrolment in several all-comers trials [14]. Similar findings were presented by Martin et al. [16], who reported that declining to participate was the main cause of non-enrolment of potentially eligible patients (29.5%) in a series of cardiovascular RCTs. Constantino et al. [10] examined HF RCTs and identified too low NYHA class, comorbidity, recent acute event, too high EF and age as the main reasons for exclusion of patients with HF. In the present study, 28.1% of iRCT-eligible patients were not enrolled due to lack of consent.
Kennedy-Martin et al. [12] found that patients encountered in everyday practice compared to patients enrolled in major cardiology RCTs were more likely to have higher risk characteristics as they were older, more likely to be female and to have clinical impairment and co-morbid disease, were treated less frequently with guideline-recommended therapy, and underwent fewer in-hospital procedures. Even after application of RCT inclusion/exclusion criteria to these real-world cardiology patients, the majority of the clinical differences were still present [12]. Moreover, all-comer cardiology RCTs also favour enrolling patients in less severe condition, younger, with a lower risk profile and less likely to have HF and a history of MI [14, 18]. Patients not enrolled in trials were reported to have two to six times higher long-term mortality compared to study-enrolled patients [14, 15, 17–21]. In HF patients, rarely, the trial-enrolled patients had more complex comorbidities [22] or lower EF or NYHA functional class [9]. In our present study we found that eligible-and-enrolled patients had greater severity disease as demonstrated by lower EF and higher EDV compared to eligible-but-not-enrolled patients (Table I). Moreover, our trial-enrolled patients had a higher incidence of cardiologic comorbidities and higher incidence of ICD presence. While our analysis could not reveal the mechanism of this bias, we can hypothesize that with the novel therapy and its interventional delivery in the RCTs in the subject of our analysis, the bias could arise on the side of both the patient and the physician (due to the tendency to “sicker” patient population recruitment). Overall, our present findings refute, for our study population, the commonly assumed enrolment bias towards lower risk patients’ recruitment.
The present study was retrospective. Nevertheless, due to the detailed analysis of hospital database no patient records were lost to analysis.
Conclusions
The enrolment rate was high. Ineligibility resulted mainly from recent or recommended cardiovascular intervention outside the RCT. Medical treatment of heart failure was similar between E-E and E-NE patients. Age (when within the inclusion criteria), gender and time frame were not factors of bias. However, the trial-enrolled patients had more severe left ventricle impairment. This argues, for CIHF RCTs, against the routinely assumed lower-risk patient enrolment bias as indicated in other trial types and populations.