Introduction
The Fontan procedure remains the treatment of choice for congenital cardiac malformations categorized as single-ventricle defects. While this surgical intervention enables survival, it is associated with a range of multiorgan complications that significantly impair quality of life and long-term prognosis [1]. Among these, Fontan-associated liver disease (FALD) is a notable condition, intricately tied to the pathophysiology of Fontan circulation [1–5].
Liver fibrosis in patients who have undergone Fontan palliation was first reported in 1981 [6]. However, knowledge about this complication remains limited. It is widely accepted that the unique hemodynamics of Fontan circulation – where venous blood from the superior and inferior vena cava flows directly into the pulmonary arteries without the support of a subpulmonary ventricle – plays a central role in liver damage [7–10]. FALD is thus considered a distinct form of congestive hepatopathy. Its pathogenesis appears multifactorial, influenced by factors such as elevated central venous pressure, nonpulsatile blood flow, and reduced cardiac output [7–10].
Although some degree of FALD appears to affect all Fontan patients, the proportion that will progress to clinically significant advanced liver disease remains unclear [7–10]. For example, a study by Munsterman et al. revealed that all Fontan patients had biopsy-confirmed liver fibrosis, with severe fibrosis (stages 3–4 based on modified METAVIR criteria) identified in 68% of cases [11]. Advanced liver disease further complicates the clinical management of Fontan patients, particularly in terms of transplant eligibility and decision-making [12].
Despite its critical importance, there is no universally accepted approach to diagnosing and grading liver damage in this population [1]. While liver biopsy remains the gold standard, it is fraught with challenges in Fontan patients due to factors such as anticoagulant therapy, vascular malformations, and anatomical anomalies, including heterotaxy syndrome [12–14]. Consequently, non-invasive diagnostic methods, including transient elastography (FibroScan) and sonoelastography, are commonly used for routine monitoring [2, 4, 7, 10, 15]. However, the accuracy of these methods depends on specialized equipment and the operator’s expertise, particularly in accommodating the unique characteristics of Fontan circulation and its effects on liver stiffness (LS).
As seen in other chronic liver diseases, such as chronic hepatitis B (HB), laboratory-based indices have also demonstrated utility in clinical assessments [16–19]. Commonly used indices include the aspartate aminotransferase (AST)-to-platelet ratio (APRI) and the FIB-4 index. Additionally, a simpler metric – the red cell distribution width (RDW)-to-platelet ratio (RPR) – has been proposed for similar evaluation. The RPR index has shown reliability and comparable efficacy to APRI and FIB-4 in conditions such as HB, chronic hepatitis C (HC), and primary biliary cirrhosis [20, 21]. However, to date, no studies have evaluated the use of the RPR index in adults with Fontan circulation.
Material and methods
This retrospective, cross-sectional study included adult Fontan patients recruited from a large center specializing in adult congenital heart disease. The study was conducted from January 2022 to December 2024.
The inclusion criteria were as follows: age ≥ 18 years, a history of operations involving the creation of Fontan circulation, ability to provide informed consent, stable clinical condition during evaluation, liver sonoelastography performed in conjunction with a panel of laboratory tests within the last 2 years, and invasive hemodynamic diagnostics conducted within the last 5 years.
Exclusion criteria were: acute clinical deterioration during evaluation, conditions requiring immediate surgical or interventional treatment, pregnancy, a positive test result for hepatitis B surface antigen (HBsAg), a positive test result for hepatitis C virus antibodies, a past diagnosis of hepatitis B or C, autoimmune diseases causing chronic liver damage, severe ascites preventing reliable LS assessment on ultrasound, diagnosed cancer, alcohol abuse, and pregnancy.
The study involved a retrospective analysis of data obtained from routine diagnostics of Fontan patients at our center. Variables collected from medical records included: patient demographics, age at the time of surgical repair, type of systemic ventricle, the most recent liver sonoelastography results with corresponding laboratory tests, cardiopulmonary exercise test (CPET) results, transthoracic echocardiography (TTE) findings from the same hospitalization, and the latest results from invasive hemodynamic diagnostics.
Abdominal ultrasound with liver sonoelastography was performed in all cases by a single highly experienced researcher using the Philips iU22 XMatrix Ultrasound System (Koninklijke Philips N.V., Amsterdam, Netherlands). For each patient, 10 measurements were taken to estimate LS. The severity of LS was assessed using the METAVIR scoring system (F1–F4), with F3 and F4 categorized as severe Fontan-associated liver disease (FALD).
Exercise tolerance was assessed via CPET on a treadmill using the modified Bruce protocol (Reynolds Medical System, ZAN-600 [Lode BV, Groningen, Netherlands]). Maximum oxygen uptake (VO2 max) was defined as the highest value at peak workload, expressed in ml/kg/min and as a percentage of the predicted value. The test was considered valid if the respiratory exchange ratio was ≥ 1.0.
Hemodynamic diagnostics were performed according to clinical indications and adhered to a standardized protocol, including assessments of venous and arterial pressures, measurement of oxygen saturation at various circulatory levels, and angiographic evaluation to rule out significant anatomical abnormalities such as vascular fistulas.
In addition, non-invasive liver state indices were calculated using laboratory test results and published formulas for parameters such as AST/ALT ratio, APRI, FIB-4, and GGT/ALT. MELD and other parameters requiring international normalized ratio (INR) were excluded due to the frequent use of vitamin K antagonists, which could significantly affect the results. Instead, we assessed the Model for End-Stage Liver Disease excluding INR (MELD-XI)
Statistical analysis
IBM SPSS Statistics, version 29.0, was used for data analysis. Continuous variables were presented as mean (standard deviation) or median (interquartile range [IQR]), while qualitative data were summarized as counts and percentages. The c2 test or Fisher’s exact test was used for comparisons of categorical variables. For continuous variables, normal distribution was assessed using the Shapiro-Wilk test. For normally distributed data, the t-test was applied, while the Mann-Whitney test was used for non-normally distributed data. Data with normal distribution were presented as mean ± SD, and non-normally distributed data as median (IQR). Receiver operating characteristic (ROC) curves and the area under the curve (AUC) were calculated using IBM SPSS Statistics software. Logistic regression was applied to account for both quantitative and qualitative parameters. A p-value of less than 0.05 was considered statistically significant. The study power for average LS was estimated at 0.98.
Results
Fifty-six patients (24 females, 43%; median age 24 years, interquartile range: 21–27) were enrolled. The number of patients in individual LS categories according to METAVIR was as follows: F0-1 (0), F2 (22 patients – 39%), F3 (31 patients – 55%), F4 (3 patients – 5%). Patients were grouped by LS: Group 1 (METAVIR ≤ F2) and Group 2 (> F2). The basic characteristics of the study population and comparison between groups are presented in Table I. In terms of body weight, the difference between the groups was borderline statistically significant. However, for the average BMI, Group 2 shows a significantly lower value by 12%. Interestingly, the groups did not differ significantly in age and time since surgery.
Table I
Baseline characteristics of study population
[i] AFP – α-fetoprotein, ALP – alkaline phosphatase, ALT – alanine aminotransferase, AST – aspartate aminotransferase, BMI – body mass index, FO – Fontan operation, GGT – γ-glutamyl transferase, HCT – hematocrit, Hgb – hemoglobin, hsCRP – high-sensitivity C-reactive protein, IVC – inferior vena cava, MCH – mean corpuscular hemoglobin, MCHC – mean corpuscular hemoglobin concentration, MCV – mean corpuscular volume, MPV – mean platelet volume, NT-proBNP – N-terminal pro-brain natriuretic peptide, PDW – platelet distribution width, PLT – platelet count, RBC – red blood cell count, RDW – red cell distribution width, RPR – red blood cell distribution width-to-platelet ratio, SpO2 – peripheral oxygen saturation, SV – single ventricle, TTE – transthoracic echocardiography, VO2max – maximal oxygen consumption, WBC – white blood cell count. Values presented as mean ± standard deviation or median [interquartile range].
Turning to the complete blood count results, we found no differences between the groups in terms of the number of leukocytes and red blood cell parameters, including RDW. However, Group 2 was characterized by a 34% significantly lower median platelet count, with similar platelet distribution width (PDW) and mean platelet volume (MPV) results. It is also worth noting the lack of significant differences in the basic enzymes (ALT, AST, GGT, ALP) used to assess liver function. However, in Group 2, the median total bilirubin and direct bilirubin concentrations were significantly higher (+135% and +305%, respectively).
Patients with higher LS also exhibited higher pressures in the Fontan conduit (connection of inferior vena cava and pulmonary arteries). Surprisingly, we did not observe significant differences between the groups in other parameters that we used to assess the circulatory system, including: NT-proBNP, VO2max, and end-diastolic pressure for the systemic ventricle. It is also worth noting the higher concentration of cystatin C in Group 2 (+115%), with no significant differences in creatinine.
Table II summarizes the noninvasive liver markers calculated from laboratory results. The analysis confirmed that RPR achieved significantly higher values (+150%) in the group of patients whose LS was rated > F2 in sonoelastography. This is consistent with the results for other parameters (AST/ALT, APRI, FIB-4, MELD-XI) assessing the severity of liver dysfunction.
Table II
Comparison of laboratory markers used in assessment of chronic liver disease in patients with Fontan circulation
[i] ALT – alanine aminotransferase, APRI – aspartate aminotransferase to platelet ratio index, AST – aspartate aminotransferase, FIB-4 – Fibrosis-4 index, GGT/ALT – γ-glutamyl transferase to alanine aminotransferase ratio, MELD-XI – Model for End-Stage Liver Disease excluding INR, RPR – red blood cell distribution width-to-platelet ratio. Values presented as mean ± standard deviation or median [interquartile range].
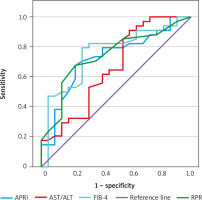
Additionally, we estimated ROC curves in predicting LS > F2 assessed by sonoelastography for RPR and laboratory markers typically used to estimate stage liver fibrosis. A graphical representation of these curves is shown in Figure 1. Analyzing Table III, which contains the AUC characterizing individual curves, it can be seen that RPR shows similar effectiveness in predicting severity of LS > F2.
Table III
Comparison of ROC curve parameters for selected markers used in assessment of chronic liver damage
| Parameter | ROC AUC | 95% CI | Proposed cut-off point |
|---|---|---|---|
| AST/ALT | 0.66 | 0.51–0.81 | 0.8 |
| APRI | 0.73 | 0.59–0.86 | 0.5 |
| FIB-4 | 0.76 | 0.62–0.89 | 0.8 |
| RPR | 0.74 | 0.61–0.87 | 0.1 |
Figure 1
Graphical presentation of ROC curves for individual laboratory markers of liver damage in predicting liver stiffness > F2 METAVIR assessed by elastography in adult patients with Fontan circulation
ALT – alanine aminotransferase, APRI – aspartate aminotransferase to platelet ratio index, AST – aspartate aminotransferase, FIB-4 – Fibrosis-4 index, RPR – red blood cell distribution width-to-platelet ratio.
Discussion
To the best of our knowledge, this study is the first to demonstrate that the RPR ratio can serve as a useful marker for assessing FALD adult Fontan patients. Our findings are consistent with previous studies conducted in other types of chronic liver diseases, such as HB or HC [16–21]. The straightforward availability of this marker underscores the justification for its validation in this unique population. This observation gains significance given the increasing number of physicians encountering Fontan patients in daily practice, often without access to specialized equipment [1].
As previously noted, liver damage is a universal finding in Fontan patients [7–10]. However, the accurate assessment of FALD is particularly challenging due to factors such as numerous comorbidities, prolonged asymptomatic periods, and the overlap of various complications (e.g., ascites resulting from heart failure or protein loss due to protein-losing enteropathy) [7–10]. Moreover, the use of liver enzymes (such as ALT, AST, GGT) in the circulation of the adult Fontan population may not always help in assessing the liver condition, as demonstrated in our study. Focusing on FALD is important because, as research shows, it is associated with higher mortality rates (12.60% vs. 3.70% in a 5-year follow-up) [3]. Furthermore, severe liver damage is recognized as a major risk factor for hepatocellular carcinoma (HCC) in the Fontan population, which, according to current data, occurs in this group in 1–2% of patients but is associated with a 1-year survival rate of only 50% [22, 23].
As is commonly known, liver biopsy – although the gold standard – is very problematic in the evaluation of FALD [3, 7–10]. Due to concerns about complications, the most frequently reported of which is hemorrhage (incidence of 4–18.5%), this procedure is performed quite rarely [13]. A Canadian study on care models revealed that hepatologists in 8 out of 12 centers perform liver biopsies in FALD patients, but often only under specific circumstances (e.g., during evaluation for heart transplant or when liver disease diagnosis is unclear) [24]. Even when performed, liver biopsies have limitations, including difficulties in obtaining diagnostic samples and the non-uniform distribution of fibrosis, which complicates histopathological evaluation [13]. Consequently, liver biopsies should be conducted exclusively in centers with appropriate expertise and equipment to minimize complications. This limitation highlights the importance of managing Fontan patients within multidisciplinary teams. Unfortunately, such comprehensive centers which meet the standards outlined in current guidelines remain scarce worldwide [1].
Thus, in normal daily practice, non-invasive techniques often serve as the primary tools for FALD assessment, as was the case in our study. Unfortunately, there is still no uniform care model for liver diseases in the Fontan population [1]. Referring again to the Canadian data, abdominal ultrasound is the most common (12/12 centers) imaging modality used to monitor FALD progression [24]. Moreover, most guidelines recommend ultrasound plus AFP every 6 months as the primary method of screening and monitoring HCC [7–10, 22, 23]. Our center follows the same protocol, supplementing ultrasound with sonoelastography. With many years of experience in using sonoelastography, we decided to employ it in this study.
Basic tests including CBC are probably ordered by all specialists caring for Fontan patients for routine check-ups. These tests allow for the estimation of non-invasive liver fibrosis markers, which, although not specifically validated for the FALD population, may complement imaging results and clinical assessments. Most of them, including APRI, FIB-4, and MELD-IX, are already widely used in studies among the Fontan population [2, 4–10, 23–25]. In our analysis, ROC curves showed that the RPR index demonstrated similar efficacy in assessing LS compared to FIB-4 and was slightly more effective than APRI and AST/ALT ratios. It is noteworthy that this simple and accessible index has not yet been extensively studied in the Fontan population, despite promising findings in other chronic liver diseases.
Interestingly, in our study, Groups 1 and 2 did not differ significantly in terms of age or time since Fontan surgery. This may be partially explained by the fact that in a recent meta-analysis, liver biopsy results showed only a weak positive correlation between the duration of Fontan disease and the severity of fibrosis [25]. This supports the idea that unfavorable hemodynamics in Fontan circulation, rather than time alone, is the primary driver of FALD progression. Our data also suggest a relationship between high pressure in Fontan circulation and higher LS. However, the lack of significant differences in NT-proBNP concentration and exercise tolerance additionally indicates that non-invasive cardiac diagnostics in this case may provide too little information about this complex pathophysiology.
This study has several limitations. First of all, it was a single-center, retrospective study conducted on a relatively small number of participants. Secondly, we used sonoelastography as a reference method in assessing the severity of FALD, when liver biopsy is the gold standard. However, as we have already emphasized in the text, this is an invasive method associated with a risk of complications. There are many other recognized reports where non-invasive techniques are considered sufficient. We are convinced that we have extensive experience in the use of sonoelastography, which allows us to obtain valuable preliminary data. However, we are aware of the fact that a large prospective study evaluating liver biopsy samples is needed to definitively confirm our findings. Additionally, the cross-sectional design did not allow for longitudinal observations to better understand FALD dynamics. Given the high heterogeneity of the Fontan population, repeated assessments of the same patients are particularly valuable for evaluating the progression of multi-organ complications over time.
Conclusions
Our study is the first to demonstrate that the RPR ratio can be a useful marker for higher LS in adult Fontan patients. This simple and accessible indicator is especially valuable in situations where specialized equipment is unavailable. Although further validation is required, RPR may assist in identifying patients who require more detailed diagnostics.






