Summary
Cardiac allograft vasculopathy remains one of the most important factors leading to chronic cardiac allograft rejection. When revascularization is needed percutaneous coronary interventions with drug-eluting stents are the method of choice. Everolimus-eluting second generation stents are characterized by the lowest restenosis rate. Data regarding other rapamycin analogues are scarce. In the current analysis the 6 months restenosis rates were low and comparable for everolimus- and sirolimus-eluting stents, indicating safety and efficacy of both substances in the treatment of cardiac allograft vasculopathy.
Introduction
Cardiac allograft vasculopathy (CAV) remains one of the most important factors leading to chronic cardiac allograft rejection. According to the ISHLT report of 2017, CAV is responsible for 32.5% of deaths in the period between 5 and 10 years after heart transplantation. With time, CAV affects most patients after orthotropic heart transplantation (OHT). Its incidence is 29.3% and 47.4 at 5 and 10 years after the transplantation, respectively [1]. The pathomechanism of CAV involves numerous factors related to the immune system, the clinical profile of the donor and recipient, and to the surgery technique [2]. CAV differs from classic coronary atherosclerosis in that the stenoses are diffuse and concentric in nature, they do not break the internal lamina, and rarely include calcifications. Importantly, patients after heart transplantation may experience both forms of coronary artery disease at the same time [3].
The only effective method of CAV treatment is re-transplantation of the heart, which involves, however, a much higher risk of complications and death than the first heart transplantation [4]. In view of the above, and due to low organ availability, the basic treatment in clinical practice is based on continuous modifications of risk factors, the use of statins, and modifications of the immunosuppressive therapy [3]. Nevertheless, these methods have limited efficacy, and some patients require revascularization. Surgical treatment is rarely used, since it is technically challenging and has poor prognosis [5]. Therefore, the majority of patients with significant stenosis of coronary arteries secondary to CAV are qualified for percutaneous coronary intervention (PCI) [5]. With development of interventional cardiology, subsequent generations of devices were used in CAV patients, achieving a gradual improvement in long-term outcomes of the interventions performed [6–9]. Nevertheless, data on the efficacy of second generation stents eluting rapamycin analogues are limited.
Aim
To compare the short- and long-term outcomes of CAV patients treated with everolimus- (EES) or sirolimus-eluting stents (SES).
Material and methods
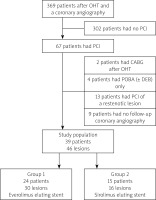
Between December 2012 and December 2020, we analysed 369 post-heart transplantation patients subject to coronary angiography at our facility. In the study period, 67 patients underwent PCI. Patients with balloon angioplasty of a restenotic lesion or a previously treated vessel, those with implanted stents other than second generation EES or SES, patients who underwent coronary artery bypass graft (CABG) after heart transplantation and patients with no angiography control were excluded from further analysis. Finally, 39 patients were enrolled in the study. The study design is shown in Figure 1.
The PCI procedures were conducted in accordance with standard local practice. The patients routinely received unfractionated heparin with target time of coagulation activation between 250 and 300 s. Intracoronary nitroglycerine (100 to 200 mg) was used during coronary angiography and before angioplasty. Baseline and follow-up angiography was performed using the same views. Quantitative coronary angiography (QCA) was performed by two independent specialists in all the patients enrolled. QCA calibration was performed with a guiding catheter. The following parameters were assessed in the study: minimum lumen diameter, reference vessel diameter, percent diameter stenosis and late lumen loss.
The primary endpoint of the study was the occurrence of angiographically significant restenosis assessed with QCA. Secondary endpoints included: binary restenosis, target lesion revascularisation (TLR) and cardiac death during a 6-month follow-up period. Standard definitions of endpoints were used in the study. Binary restenosis was defined as late lumen loss by at least 50%. TLR was defined as planned or urgent PCI of a previously treated lesion, covering the area of 5 mm after and before the implanted stent. All deaths were considered cardiac unless a definitive non-cardiac cause could be established. Clinical, angiographic, procedural, and mortality data were obtained retrospectively using the online reporting system. The patients were followed for 6 months after the first procedure. Follow-up coronary angiography in patients undergoing PCI was performed after 6 months of the initial procedure, in accordance with the protocol adopted at our site.
Additional information was obtained by telephone contact or, if necessary, from medical records. The study was approved by the Bioethics Committee of the Medical University of Silesia and was conducted in accordance with the principles set forth in the Declaration of Helsinki. All the patients gave their informed consent.
Results
The final number of patients enrolled in the study was 39, including 24 patients treated with EES and 15 treated with SES. The clinical characteristics of the analysed groups are presented in Table I. No significant differences were observed regarding the rate of risk factors of cardiovascular diseases and comorbidities. In most patients, OHT was caused by ischaemic cardiomyopathy. The patients treated with EES were younger (55.8 ±11.8 vs. 60.1 ±12.2) and less frequently male (79% vs. 93%). The incidence of hypertension, hypercholesterolaemia and type 2 diabetes was high in both groups. The SES group showed a slightly higher blood pressure (123 ±10.6 vs. 132 ±39) and a slightly lower left ventricle ejection fraction (53.3 ±2 vs. 51.7 ±6.7). The patients treated with SES had a higher level of blood everolimus (6.9 ±2.8 vs. 12.3 ±2.0 ng/ml).
Table I
Baseline characteristics of the study population
[i] BMI – body mass index, CABG – coronary artery bypass grafting, DBP – diastolic blood pressure, LVEF – left ventricular ejection fraction, MI – myocardial infarction, OHT – orthotopic heart transplant, PAD – peripheral artery disease, PCI – percutaneous coronary intervention, SBP – systolic blood pressure.
Angiographic and perioperative characteristics are illustrated in Table II. Most patients had ad hoc PCI (92% and 93%). In the EES group 30 stenoses were treated with 31 stents, and in the SES group 16 stenoses were treated using 25 stents. The majority of patients were diagnosed with single vessel disease with LAD involvement (62% and 86% in the EES group, and 47% and 56% in the SES group). Balloon predilation was more common in the patients treated with EES (57% vs. 12%). A higher number of stents was used in the group treated with SES (1.1 ±0.3 vs. 1.4 ±0.7). The frequency of perioperative complications was low in both groups.
Table II
Procedural characteristics of study population
The results of the QCA analysis are presented in Table III. Mean length of stenoses was 18.7 ±7.11 vs. 15 ±5.66 for EES and SES groups, respectively. Minimal lumen diameter (MLD) obtained after the surgery was comparable in both groups (2.44 ±0.47 vs. 2.47 ±0.37) although % stenosis of lumen area (%MLA) was slightly higher in the group treated with EES (12.2 ±8.6 vs. 1.57 ±0.78), as was the acute lumen gain (1.82 ±0.53 vs. 1.57 ±0.78). In 6 months follow-up, late lumen loss was comparable in both groups, 0.19 ±0.15 vs. 0.14 ±0.15, and binary restenosis was 4% and 0% for EES and SES groups, respectively.
Table III
Quantitative coronary analysis
No significant differences in the administered pharmacotherapy were found with regard to both cardiological and immunosuppressive pharmacotherapy (Table IV). During the 6-month follow-up period, no death, including cardiovascular death, was registered. 1 TLF was observed in the EES group.
Table IV
Pharmacotherapy on discharge
Discussion
The analysis showed that the use of second generation DES eluting rapamycin analogues is associated with high direct efficacy of surgery and low incidence of restenosis, late lumen loss and serious cardiac events in long-term follow-up. No differences were observed in relation to the type of antimitotic substance eluted (everolimus vs. sirolimus).
The treatment of CAV remains highly challenging, since there are no standards regarding the management of the disease. The only effective method, heart re-transplantation, is virtually unavailable, and surgical treatment is associated with high risk of complications, including death [1]. Percutaneous coronary interventions remain an attractive option due to low invasiveness and high direct efficacy of the procedures [5]. However, they must be treated as palliative treatment, since the interventions do not involve a reduced risk of progressive organ insufficiency and death [10, 11]. We do not have prospective studies in this area, and most data come from observational, retrospective studies. In a non-randomised study comparing CAV patients who were treated with PCI and patients in whom PCI was not possible, a lower mortality rate was observed in the PCI group, i.e. 20% vs. 43.5%, respectively, p = 0.03. Nevertheless, mean survival time after intervention was 4 years, and 32% of patients required a repeated intervention [12].
As in the case of PCI due to native atherosclerosis, the treatment of CAV initially involved balloon angioplasty, followed by metal stents, 1st generation DES, and second generation DES. The use of balloon angioplasty and BMS was associated with a high incidence of restenosis, 72% and 39%, respectively, in an 8-month follow-up [13]. The data comparing BMS with DES are ambiguous. A study analysing 45 patients with CAV subject to PCI with BMS or DES showed no significant differences in the rate of restenosis or clinical events [14]. Similar results were obtained in a study by Reddy et al., in which 42 patients treated with BMS or first generation DES were compared. The rate of restenosis was 22.6% and 22.7%, respectively. No differences in the incidence of adverse events were observed, either [15]. Nevertheless, an analysis of 6 studies including 312 patients with CAV, in which BMS were compared with 1st generation DES eluting sirolimus or paclitaxel, showed a significant reduction in restenosis when DES were used. As in the above-mentioned studies, this was not associated with a reduced incidence of serious cardiac events [16].
Available data on second generation DES indicate further improvement in PCI outcomes in patients with CAV. An analysis of 48 patients who underwent PCI regarding 113 stenoses showed that the incidence of binary restenosis increased by 3% in a 1-year follow-up [17]. In an analysis of 21 patients with CAV treated with EES, the rate of restenosis and TLR was 5.9% [18]. These results are similar to those achieved in the treatment of native coronary arteries [8, 9]. It is emphasised that everolimus may show additional benefits in the population of CAV patients in that it stops migration and proliferation of smooth muscle cells, and inhibits endothelial progenitor cells [19]. Moreover, systemic use of everolimus may be associated with inhibition of CAV progression [20].
In the present study, we found high direct efficacy of PCI procedures using EES and SES. QCA showed similar results in both groups, both directly after the procedure and in a long-term follow-up. The incidence of binary restenosis was low and comparable with the incidence observed in the above quoted studies. Of note is a higher number of stents used in the SES group; however, it did not affect the results of long-term angiographic follow-up. The obtained results show similar efficacy of both the rapamycin analogues in preventing restenosis in patients with CAV. Also, good results achieved with second generation stents may be associated not only with the type of the antimitotic substance released, but also with the construction of the stent platform. Nevertheless, small group sizes in the presented analysis prevent the formulation of final conclusions.
Advances in interventional cardiology, first of all the introduction of stents eluting antimitotic substances, are associated with improvement in direct efficacy of PCI procedures and with significant reduction in the restenosis rate. The efficacy of DES may result from the nature of CAV, which is characterised by excessive proliferation of the internal lamina. By reducing neointimal proliferation, DES reduces the incidence of restenosis [21]. On the other hand, DES do not reduce an increased lymphoproliferative response in the tunica intima, media and externa, and the mechanism is responsible for the progressive and diffuse nature of CAV beyond the stented lesion. This phenomenon may partially explain the lack of benefits from using DES in the reduction of adverse cardiac events in the published studies [10].
The presented analysis is a single-centre, observational, retrospective study, limited to CAV patients requiring revascularisation. In addition, the analysed groups are small, which affects the reliability of the analysis and the possibility of forming conclusions. Angiographic assessment was performed using QCA, and the sensitivity of angiography is known to be lower than that of intracoronary imaging using IVUS or OCT. Intracoronary imaging was not used in this study, so it was not possible to perform a detailed assessment of the mechanisms of late lumen loss, except for tunica intima hyperplasia. Since there are no standards of CAV management, a comparison of the conducted analysis with the results of other studies may involve a high number of confounding factors resulting from differences in the treatment administered, including immunosuppression, and modification of risk factors.