Introduction
Cardiovascular disease is the leading global cause of death, according to the World Health Organization (WHO), accounting for more than 31% of deaths in 2016 (17.9 million) [1].
Over 43% of these deaths were due to coronary artery disease (CAD). Non-ST-segment elevation myocardial infarction (NSTEMI) accounts for up to 70% of acute coronary syndromes caused by CAD [2]. A substantial risk factor of CAD is diabetes mellitus, prevalence of which has nearly doubled in the last 30 years (from 4.6% to 9.3% of the global adult population according to the International Diabetes Federation), and the majority of patients have diabetes mellitus type 2 (T2DM) [3]. The latest data show that CAD affects up to 32% of patients with T2DM [4]. It has also been proven that T2DM worsens the in-hospital and long-term prognosis of patients with NSTEMI [2]. CAD has complex etiology, including metabolic disturbances in cardiomyocytes and endothelial dysfunction. Those are especially affected in T2DM, due to insulin resistance and deregulation of glucose and fatty acid metabolism [5].
The transcriptional factor forkhead box O1 (FOXO1) is an important regulator of cell metabolism in several tissues, including the heart and the endothelium, where it is involved in cardiomyocyte metabolism (glucose and lipid metabolic pathways) [6], cell survival [7], atherosclerosis [8], and progression of diabetic cardiomyopathy [9–11].
The role of FOXO1 in genetic predisposition to CAD is unclear. A study by Kedenko et al. linked the polymorphism of FOXO1 with carotid atherosclerosis [12]. In contrast, the study by Zhao et al. on a Han Chinese population (n = 1287) showed no association of FOXO1 and the prevalence of CAD [13]. Previous studies on animal models have shown that FOXO1 expression is increased in myocardial infarction and that FOXO1 protects the myocardium through upregulation of antioxidant genes, in consequence reducing the infarct size [14, 15]. However, the increased expression of FOXO1 in mice with insulin resistance or diabetes led to development of diabetic cardiomyopathy [11] and deletion of the FOXO1 gene in diabetic animal models prevented heart failure [9]. FOXO1 is also proven to be involved in diabetes-induced vascular remodeling in rats [16].
Vascular remodeling is also modulated by vascular growth factors among other angiopoietins, with two major ones being angiopoietin 1 (Ang-1) and angiopoietin 2 (Ang-2). One of particular interest is angiopoietin-2 (Ang-2), exclusively expressed in endothelium cells, possessing an opposite action to Ang-1, which exhibits endothelium protective properties [17]. Conditions such as chronic hyperglycemia or chronic inflammation lead to both FOXO1 transcriptional activation (PI3K/Akt signaling) [18] and Ang-2 gene expression induction in endothelial cells, binding the Tie-2 receptor, and therefore inhibiting the angiopoietin-1 because it binds to the same Tie-2 receptor, which cause destabilization of the endothelium and increase the arteriosclerosis process [19–22].
It is unclear how the FOXO1 and ANGPT2 gene expression changes during myocardial ischemia.
This study is a continuation of our previous research [23] which proved that Ang 2 serum concentration is elevated in patients with NSTEMI both with and without T2DM as well as among T2DM patients without a history of CAD.
Aim
Our objective was to evaluate ANGPT2 and FOXO1 gene expression in patients with NSTEMI, both with and without T2DM, and compare it to patients with T2DM but without CAD and to those without T2DM and without CAD. To our knowledge, no studies have evaluated ANGPT2 and FOXO1 gene expression in human patients with NSTEMI and diabetes.
Material and methods
This was a multi-center, prospective study that included patients consecutively hospitalized in cardiology or diabetology wards between January 2017 and July 2017 due to NSTEMI (emergency admission) or T2DM. The control group consisted of patients with arterial hypertension and suspected of cardiovascular diseases which were finally unconfirmed.
Inclusion and exclusion criteria are presented in Table I. In brief, 138 patients who fulfilled inclusion/exclusion criteria were enrolled in the study; 11 patients were not included for further analysis (due to incomplete data).
Table I
Inclusion and exclusion criteria
There were 127 patients divided into 4 groups: Group A: 24 patients with NSTEMI and T2DM; Group B: 43 patients with NSTEMI without T2DM; Group C: 30 patients with T2DM, without a history of CAD; Group D (control group): 30 patients without CAD and without T2DM.
On the day of hospital admission patients had 10 ml of peripheral blood drawn for the biochemical analysis with high-sensitivity C-reactive protein (hs-CRP) and troponin level (TnT), and anthropometric and demographic data, as well as past medical history, were collected. Electrocardiography and echocardiography with assessment of ejection fraction (EF%) were performed. The blood analysis included: FOXO1 and ANGPT2 gene expression (qPCR analysis) and serum level of Ang-2 and glycated hemoglobin (HbA1c). Patients from groups C and D had no history of CAD, presented no symptoms of CAD and had no ischemic changes in ECG or ECHO (preserved ejection fraction and no features of having undergone myocardial ischemia).
The study was approved by the Medical University of Silesia Ethics Committee and conformed to the Declaration of Helsinki (KNW/0022/ KB1/28/16; KNW/0022/KB1/28/I/16/17). Informed written consent was obtained from every patient enrolled in the study.
FOXO1 and ANGPT2 gene expression analysis
The buffy coat was obtained by centrifugation of peripheral blood and stored in aliquots at –80°C until further analysis.
RNA was extracted from the buffy coat using a Maxwell 16 MDX device (PROMEGA Wisconsin, USA) with Cell Lysis Solution (Kit A793A PROMEGA, Wisconsin, USA) and Lysis Buffer (Kit MC501C, PROMEGA Wisconsin, USA) according to the manufacturer’s protocol [24].
RNA concentration was determined spectrophotometrically by measuring the absorbance at 260 nm and the purity was estimated by the ratio A260/A280 nm and A260/A230 nm (Denovix DS-11, Denovix Inc., Wilmington, Delaware, USA).
The purified RNA was reverse transcribed to cDNA using a SuperScript VILO MasterMIX kit (Kit 11755-250, Thermo Fisher Scientific, California, USA) according to the manufacturer’s protocol [25].
ACTB and TBP genes were initially selected as candidates for the reference gene. Quantitative analysis of the FOXO1, ANGPT2, actin beta (ACTB) gene and TATA box binding protein (TBP) genes was carried out by two-step reverse transcription real-time quantitative PCR assay (qPCR) using a LightCycler 96 Instrument (ROCHE) with a thermal profile set as follows: 95°C/10 min then 55 cycles of 95°C/15 s, 60°C/60 s.
FOXO1, ANGPT2, TBP and ACTB gene expression levels were measured using TaqMan chemistry and the primer/probe pairs with the following assay IDs – Hs01054576 m1 (FOXO1), Hs01048042 (ANGPT2), Hs00427620 m1 (TBP), Hs99999903 (ACTB).
After validation using RefFinder [26] the TBP gene proved to be more stable and was used as a reference gene. The 2–ΔΔCq method was used to analyze FOXO1 and ANGPT2 expression levels relative to the TBP reference gene control [27].
If a PCI was performed, the same blood serum analysis was repeated at discharge to assess potential changes of FOXO-1 and ANGPT2 gene expressions after PCI.
Blood serum collection and analysis
The Ang-2 levels were measured using enzyme-linked immunosorbent assay (ELISA) (RayBio ANGPT2 ELISA Kit, RayBiotech Inc. Norcross USA, Catalog #: ELH-Angiopoietin2). HbA1c value was analyzed with a high-performance liquid chromatography method. Serum high-sensitivity troponin concentration was measured using the chemiluminescence method.
Angiographic complexity of CAD
Patients from groups A and B (NSTEMI with/without T2DM) underwent urgent (< 24 h) coronarography and the SYNTAX score grading system was used to determine the complexity of coronary artery disease and in order to qualify patients for PCI or CABG. Each patient with NSTEMI also had the GRACE 2.0 score calculated in order to estimate the in-hospital outcome. The SYNTAX score, GRACE 2.0 score, number of implanted stents (in the case of PCI) and type of intervention (PCI or CABG) were further analyzed and compared with FOXO1 and ANGPT2 gene expression.
Statistical analysis
All statistical calculations were performed using Microsoft Office Excel, Statistica 12.0 (StatSoft Inc., USA) software and R software (version 3.4.4). The Shapiro-Wilk normality test was used to establish the distribution of quantitative variables. Descriptive statistics for continuous parameters of normal distribution are presented as the arithmetic means (standard deviation (SD)). Median values with interquartile range are presented for continuous data that did not have normal distribution. Absolute values and percentages are given for categorical and qualitative variables. The Kruskal-Wallis test was used to determine the significance of differences between multiple groups, and the Wilcoxon test was employed for the cases with two groups. Correlations between pairs of continuous variables were fitted using the Spearman rho coefficient. Logistic regression analyses were performed to determine whether FOXO1 or ANGPT2 gene expression levels were independently associated with the complexity of CAD. The prognostic accuracy of NSTEMI of FOXO1 gene expression was assessed using an area under the receiver operating characteristic (ROC) curve. The threshold of statistical significance for all tests was set at p < 0.05.
Results
Baseline group characteristics and results are presented in Table II. There were 127 patients included in the final analysis (84 males – 66%). Age of patients in groups A, B, and C did not differ significantly between each group, though the control group was significantly younger in comparison to groups A, B and C (p < 0.05). T2DM patients (groups A and C) had higher BMI and HbA1c in comparison to patients without T2DM (groups B and D, p < 0.05). Among patients with NSTEMI, both groups included more active smokers than among patients without NSTEMI. The level of cholesterol was lower in patients with NSTEMI and T2DM (group A) than in patients from groups B, C, D, p < 0.05.
Table II
Baseline group characteristics and results. Group A – NSTEMI with T2DM; group B – NSTEMI without T2DM; group C – T2DM; group D – control subjects
Relative expression of ANGPT2 and FOXO1 genes
The ANGPT2 gene expression in buffy coat in the analyzed samples was very low and detectable only in 11 patients from all groups (8.66 % of all patients) (Table III).
Table III
Kruskal-Wallis test; comparison of FOXO-1 expression in study groups – median, 1st and 3rd quartile. Group A – NSTEMI and T2DM; group B – NSTEMI without T2DM; group C – T2DM; group D – control subjects
The FOXO1 gene expression in buffy coat was detectable in all analyzed samples.
Kruskal-Wallis test was used to determine significance of differences between multiple groups and showed no significant difference of FOXO1 relative expression (Table III).
However, the level of FOXO1 expression was significantly higher in patients with NSTEMI (groups A and B combined, median relative expression = 1.39) than in non-NSTEMI patients (groups C and D combined, median = 1.09) (W = 1578, p < 0.05) (Table III).
In the between-group comparison significantly higher FOXO1 expression was observed in patients with NSTEMI and T2DM (group A) in comparison to the control group (1.49 vs. 1.08, p < 0.05).
Angiographic complexity of CAD
We compared the complexity of coronary artery disease in patients with NSTEMI and with/without T2DM. Diabetic patients had a higher SYNTAX score (17 points vs. 10 points; p < 0.05) and were more often qualified for CABG (29.2% vs. 16.3%; p < 0.05). In both groups PCI was performed in a similar percentage of patients (54% vs. 60%; p = NS). After statistical analysis (logistic regression model, Spearman correlation) no link between FOXO1 expression and the severity of CAD (SYNTAX score), GRACE score, number of implanted stents or type of intervention (PCI vs. CABG) was found.
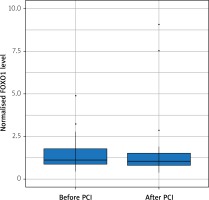
The FOXO1 relative expression was not correlated with GRACE 2.0 score or SYNTAX score of NSTEMI patients. Also, we did not observe any significant change in FOXO1 gene expression after successful angioplasty (Figure 1).
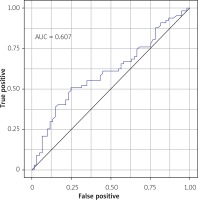
The prognostic accuracy of FOXO1 gene expression in detecting NSTEMI determined with the area under the curve (AUC = 0.607; Figure 2) was not a significant predictor of NSTEMI in the logistic regression model (p = 0.308).
Discussion
We observed a very low ANGPT2 relative gene expression in buffy coat (only in 8.66% of patients) – it was under the cut-off value (Ct > 40) of the qPCR reaction. These results are in line with a previous publication by Ehrlich et al. – ANGPT2 had the lowest expression in blood relative to other tissues [28].
We found no correlation of ANGPT2 relative gene expression with Ang-2 concentration, although the Ang-2 serum level in peripheral blood was elevated in patients with NSTEMI and T2DM [23].
The next step in our study was to evaluate the FOXO1 relative gene expression, given that the FOXO1 protein is involved in vascular remodeling and has been previously reported to be elevated in myocardial infarction in animal models and to promote cardiac dysfunction [9, 14, 15, 29–31]. The FOXO1 gene expression was under the Ct cut-off value in all study patients. The main finding of this study is that FOXO1 gene expression in blood peripheral cells is elevated in patients with NSTEMI compared to patients without NSTEMI (both with T2DM and without T2DM; Table III, relative expression – 1.39 vs. 1.09; p < 0.05). The percutaneous coronary intervention had no significant effect on FOXO1 gene expression and did not correlate with the angiographic complexity of CAD in patients with NSTEMI.
A recent study by Hueso et al. [32] reported downregulation of FOXO1 gene expression in patients with stable CAD. Subjects in this study (n = 10 with) were undergoing scheduled CABG surgery. This study combined with the results of ours can lead to the conclusion that FOXO1 gene expression is downregulated in stable CAD and upregulated in myocardial infarction.
Accumulating evidence has demonstrated that FOXO1 as a key angiogenic regulator also participates in the development and progression of diabetes. FOXO1 expression level has been found to be elevated in many tissues of diabetic mice and involved in diabetes-induced oxidative stress and cell apoptosis [7, 33]. In our study we found no significant differences between FOXO1 gene expression in patients with T2DM (group C) and the control subjects (group D) (Table III). Also, no correlation between HbA1c and FOXO1 gene expression was found.
Conclusions
On the basis of our results we can conclude that analyzing the ANGPT2 gene expression in peripheral blood has no role in assessment of CAD complexity among patients with and without T2DM. FOXO1 gene expression in blood peripheral cells is elevated in patients with NSTEMI regardless of the presence of T2DM. FOXO1 expression does not decrease after percutaneous coronary intervention and is not correlated with the severity of CAD in patients with NSTEMI.