Summary
In percutaneous coronary intervention (PCI) to atherosclerotic lesions in the right coronary artery (RCA), coronary artery dissection in the ostium of the RCA is a rare but fatal complication. We investigated factors associated with aorto-ostial stent coverage to mild to moderate ostial stenosis during the treatment of severely narrowed, non-ostial RCA lesions. The multivariate logistic regression analysis revealed that continuous ostial plaque detected by intravascular ultrasound (IVUS) was significantly associated with ostial stent coverage, and ischaemia-driven target vessel revascularization was less frequently observed in the ostial-coverage group than in the non-coverage group. The use of IVUS may enhance the safety but may increase the total stent length in PCI to RCA.
Introduction
In percutaneous coronary intervention (PCI) to atherosclerotic lesions in the right coronary artery (RCA), coronary artery dissection in the ostium of the RCA is a rare but fatal complication, because dissection can extend to the ascending aorta retrogradely [1–3]. Stent implantation to the ostium of the RCA may be selected not only for the treatment of atherosclerotic lesions, but also for the prevention of aorto-ostial dissection [1, 4, 5]. Indeed, it is not uncommon to implant stents to non-severely narrowed ostial lesions following stent implantation to severely narrowed, non-ostial RCA lesions in the contemporary drug-eluting stent era. However, references regarding aorto-ostial stent coverage have been sparse in PCI to RCA.
Aim
The purpose of this study was to find factors associated with aorto-ostial stent coverage to mild to moderate stenosis during intravascular ultrasound (IVUS)-guided PCI of severely narrowed, non-ostial RCA lesions.
Material and methods
Study lesions
We reviewed consecutive PCI cases in the Saitama Medical Center, Jichi Medical University from 1 January 2017 to 31 December 2019. Inclusion criteria were (1) cases that underwent PCI to severely narrowed, non-ostial RCA stenosis (≥ 75%), and (2) cases that had mild to moderate stenosis at the ostium of the RCA before PCI. Exclusion criteria were (1) cases that had severe stenosis (≥ 75%) at the ostium of the RCA before PCI, (2) cases that did not have any stenosis (i.e. 0% stenosis) at the ostium of the RCA before PCI, (3) cases that underwent stent-less PCI, (4) cases in which PCI was unsuccessful, (5) cases that did not use IVUS, (6) cases in which IVUS data was not available for analysis, and (7) cases in which quantitative coronary angiography (QCA) was not available before stent implantation. We defined the ostium of RCA as ≤ 3 mm from the aorto-ostial border (4). The ostial-stent coverage was defined as stenting in the area of the ostium of RCA (i.e. ≤ 3 mm from the aorto-ostial border). The final study population was divided into the ostial-coverage group, which underwent stent implantation to the ostium of the RCA, and the non-coverage group, which did not undergo stent implantation to the ostium of the RCA. Our primary interest was to find factors associated with ostial-stent coverage using multivariate logistic regression analysis. We also compared the clinical outcomes between the ostial-coverage group and the non-coverage group. In the follow-up analysis, the primary outcome was ischaemia-driven target vessel revascularization (TVR). We defined the follow-up duration ranged from the day of PCI until the date of ischaemia-driven TVR or the study end date (31 July 2020). This study was approved by the institutional review board (S20-076), and written informed consent was waived because of the retrospective study design.
Definition
Acute myocardial infarction (AMI) was defined based on the universal definition of myocardial infarction [6, 7]. ST-elevation myocardial infarction (STEMI) was diagnosed in the presence of new ST elevation at the J point in at least 2 contiguous leads ≥ 2 mm (0.2 mV) [8]. Hypertension was defined as medical treatment for hypertension or a history of hypertension before admission [9, 10]. Dyslipidaemia was defined as a total cholesterol level ≥ 220 mg/dl, low-density lipoprotein cholesterol level ≥ 140 mg/dl, medical treatment for dyslipidaemia, or a history of dyslipidaemia [11–13]. Diabetes mellitus was defined as a haemoglobin A1c level ≥ 6.5% (as NGSP value), medical treatment for diabetes mellitus, or a history of diabetes mellitus [11]. We calculated the estimated glomerular filtration rate (eGFR) from the serum creatinine level, age, weight, and gender using the following formula: eGFR = 194 × Cr – 1.094 × age – 0.287 (male), eGFR = 194 × Cr – 1.094 × age – 0.287 × 0.739 (female) [14].
Angiographical analysis
Quantitative coronary angiography (QCA) parameters were measured using a cardiovascular angiography analysis system (QAngio XA 7.3, MEDIS Imaging Systems, Leiden, Netherlands) [15–17]. The values of minimum lumen diameter, percentage diameter stenosis, lesion length, and reference diameter were obtained from pre-procedural angiography. In cases of total occlusion such as STEMI or chronic total occlusion, QCA was performed from the image after balloon dilatation. Cases that did not have an appropriate angiographic image for QCA were excluded. In our catheter laboratory, the use of IVUS was encouraged during the study period. Continuous ostial plaque was defined as continuous plaque from the culprit lesion to the guide catheter or RCA ostium.
Statistical analysis
Data are shown as the mean ± SD for normally distributed variables, median (interquartile range) for non-normally distributed variables, or percentage. The Kolmogorov-Smirnov test was performed to determine whether the continuous variables were normally distributed. Normally distributed continuous variables were compared between the groups using the unpaired Student’s t-test. Otherwise, the continuous variables were compared using the Mann-Whitney U test. Categorical variables were compared using a χ2 test. Event-free survival curves were constructed using the Kaplan-Meier method, and statistical differences between the curves were assessed by the log lank test. P < 0.05 was considered statistically significant. We performed a multivariate logistic regression analyses to find factors associated with ostial-stent coverage. In this model, the ostial-stent coverage was adopted as a dependent variable. Variables that had significant differences (p < 0.05) between the ostial-coverage group and the non-coverage group were adopted as independent variables. When there were 2 or more similar variables, only 1 variable was entered into the multivariate logistic regression analysis to avoid multi-collinearity. The odds ratio (OR) and the 95% confidence interval (CI) were also calculated. We analysed all data by SPSS ver. 24 for Windows (SPSS, Inc., Chicago, Illinois).
Results
During the study period, we had 810 patients who underwent PCI to the RCA lesions in our medical centre. We excluded 466 cases that had ≥ 75% stenosis or no stenosis at the RCA ostium by angiography. Of 344 cases that had mild to moderate stenosis at RCA ostium, 108 cases were excluded according to the exclusion criteria. The final study population was 236 patients, who were divided into the ostial-coverage group (n = 52) and the non-coverage group (n = 184). A study flowchart is shown in Figure 1. The median follow-up duration was 13 months.
Figure 1
Study flowchart
PCI – percutaneous coronary intervention, RCA – right coronary artery, IVUS – intravascular ultrasound, QCA – quantitative coronary angiography.
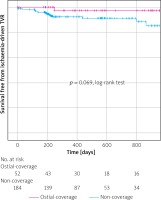
Figure 2
Kaplan-Meier curves describing survival free from ischaemia-driven target vessel revascularization
The comparison of clinical characteristics between the ostial-coverage and non-coverage groups is shown in Table I. Clinical characteristics except age were comparable between the 2 groups. A comparison of lesion and procedural characteristics between the 2 groups is shown in Table II. Lesion length was significantly longer in the ostial-coverage group (32.1 mm (13.1–42.7 mm)) than in the non-coverage group (17.2 mm (10.3–31.1 mm)) (p = 0.012). The prevalence of chronic total occlusion lesion was significantly greater in the ostial-coverage group (23.1%) than in the non-coverage group (8.7%) (p = 0.008). The prevalence of continuous ostial plaque detected by IVUS was significantly greater in the ostial-coverage group (84.6%) than in the non-coverage group (52.9%) (p < 0.001). Rotational atherectomy was more frequently used in the ostial-coverage group (23.1%) than in the non-coverage group (8.7%) (p = 0.008).
Table I
Comparison of clinical characteristics between the ostial-stent and non-stent groups
Table II
Comparison of lesions and procedural characteristics between ostial-stent and non-stent groups
[i] Data are expressed as the median (interquartile range) or number (percentage). Student’s t-test or Mann-Whitney U test was used for continuous variables, and χ2 test was used for categorical variables. PCI – percutaneous coronary intervention, STEMI – ST-segment elevation myocardial infarction, NSTEMI – non-ST-segment elevation myocardial infarction, RCA – right coronary artery.
The multivariate logistic regression analysis performed to find factors associated with ostial stent coverage is shown in Table III. Continuous ostial plaque detected by IVUS (OR = 5.398, 95% CI: 2.322–12.553, p < 0.001) was significantly associated with ostial stent coverage and the use of Amplatz left (AL) type guide catheter (vs. others: OR = 0.403, 95% CI: 0.187–0.868, p = 0.02) were inversely associated with ostial stent coverage. The Kaplan-Meier curves showed that ischaemia-driven TVR was less frequently observed in the ostial-coverage group than in the non-coverage group, without reaching statistical significance (p = 0.069).
Table III
Multivariate logistic regression analysis to find factors associated with ostial stent implantation
Discussion
The present study included 236 cases that underwent stent implantation to severely narrowed, non-ostial RCA lesions, divided into an ostial-coverage group (n = 52) and a non-coverage group (n = 184). Ostial stent coverage was significantly associated with continuous ostial plaque detected by IVUS and was inversely associated with the use of an AL-type guide catheter in the multivariate logistic regression analysis. Kaplan-Meier curves revealed a trend that the ostial stent coverage was inversely associated with the occurrence of ischaemia-driven TVR.
We should discuss why continuous ostial plaque detected by IVUS was significantly associated with ostial stent coverage. First, because expert consensus documents have recommended that plaque burden should be less than 50% at the stent edge without lipid pool [18], we might have added another stent when the stent proximal edge was on the plaque that continued until the ostium of RCA. In fact, because the retrograde coronary artery dissection is the most catastrophic complication in PCI [19], we might take a conservative strategy to avoid the retrograde coronary-aorta dissection. Moreover, complete full-metal jacket strategy is known to be better than incomplete full-metal jacket strategy in PCI for diffuse RCA stenosis [20]. Yano et al. reported that the length of stent was not associated with long-term adverse events in IVUS-guided PCI using everolimus-eluting stents [21]. Therefore, we might not hesitate to place another stent to cover mild to moderate ostial RCA lesions, while long stent coverage poses a greater risk of long-term adverse events such as stent thrombosis especially in first-generation, drug-eluting stents [22].
We should also discuss why the AL-type guide catheter was inversely associated with ostial stent coverage. In general, the risk of coronary injury is considered to be greater in the AL-type guide catheters than in the Judkins type guide catheters [23, 24]. However, our results were contradictory, which requires careful interpretation. When operators planned to perform aorto-ostial stenting before procedures, they might select Judkins-type guide catheters, because the Judkins type is technically easier for aorto-ostial stenting than the AL type. Furthermore, there were no cases that showed coronary injury caused by the guide catheter in our study population, partly because side holes on the guide catheter were available in all cases. Therefore, our results do not imply that the AL-type guide catheters have a protective effect to avoid aorto-ostial stenting.
The clinical implications of the present study should be noted. Because the mid-term, ischaemia-driven TVR tended to be less in the ostial-coverage group than in the non-coverage group, ostial stent coverage may be an option for mild to moderate stenosis in the RCA ostium, when PCI to a severely narrowed non-ostial RCA lesion is planned. Because a factor associated with aorto-ostial coverage was continuous ostial plaque detected by IVUS, the use of IVUS may increase the chance of aorto-ostial coverage as compared to PCI without IVUS. However, it is still unknown whether the use of IVUS might enhance the safety of PCI to RCA lesions or just increase the total length of stenting. Moreover, although IVUS had difficulty identifying vulnerable plaques such as thin-cap fibroatheroma due to the limitation of the image resolution [25], stent coverage only to the vulnerable plaques might be more reasonable. Because optical coherent tomography (OCT) would detect vulnerable plaques more precisely than IVUS, especially in the culprit lesions of AMI [26, 27], OCT-guided stent coverage only to the vulnerable plaques may be a better option if possible. A prospective study is warranted to confirm whether aorto-ostial coverage is beneficial to the mild to moderate ostial stenosis when PCI to severely narrowed, non-ostial RCA lesions is planned.
First, because this study was a single-centre, retrospective study, there is a risk of selection bias. Second, our study population did not include any cases that had coronary artery injury caused by the guide catheter, partly because our study sample size was not sufficiently large to have such cases. Third, there were no strict instructions regarding aorto-ostial stent coverage during the study period in our catheter laboratory. Thus, ostial stent coverage was influenced by the discretion of each interventional cardiologist. Although the use of rotational atherectomy is known to be associated with extended stenting, it was difficult to determine whether the procedure itself or rather the plaque composition (calcium) played the central role in the extended stenting in this retrospective study. When we checked continuous ostial plaque in IVUS, we tried to include minor plaques seen in IVUS. However, some minor plaques might be missed because of the limitation of the IVUS resolution.
Conclusions
Continuous ostial plaque detected by IVUS was significantly associated with ostial stent coverage to mild to moderate stenosis when PCI to severely narrowed non-ostial RCA lesions were performed. The use of IVUS may enhance the safety but may increase the total stent length in PCI to the RCA.







