Summary
The limitations of this FIM study are its non-randomized nature and small sample size. A distinct advantage was that follow-up tools included not only Doppler ultrasound but also angiography, which was in accordance with the study protocol. In conclusion, this is a first-in-man study reporting long-term follow-up after superficial femoral artery (SFA) chronic total occlusion (CTO) revascularization using the Ocelot catheter. The catheter proved to have a satisfactory safety profile and a high proportion of CTO crossings. A 5-year follow-up revealed high primary and primary-assisted patency rates.
Introduction
Chronic total occlusions (CTO) of the superficial femoral artery (SFA) are historically recommended by TASC II as a subgroup where surgical intervention is preferred over endovascular revascularization [1]; according to literature reports approximately 40% of patients with symptomatic peripheral arterial disease have CTO of SFA [1]. Therefore, long SFA occlusion is frequently referred to as an “Achilles heel” and represents the “last frontier” of lower extremity percutaneous revascularizations [2–4]. Nowadays, the market offers many devices dedicated for revascularization of SFA CTO – non-imaging-guides catheters such as Crosser, TruePath, Frontrunner, but only one of these has built-in optical coherence tomography (OCT) – the Ocelot catheter [5–7]. The OCT provides spatial resolution that is ten times greater compared to intravascular ultrasound (IVUS) currently applied in atherectomy devices [8]. Real time high-resolution visualization may facilitate navigation through long occlusions allowing recanalization in the true lumen, thus avoiding subintimal recanalization, and intraluminal crossing may have the benefit of reducing inflammatory reaction. The OCT give a 3-dimensional rendering of the artery wall and the operator is able to use this information for stent apposition, vulnerable plaque location and target vessel for endovascular treatment. Thanks to intraluminal recanalization physicians may have a wide array of endovascular strategies, including angioplasty, stenting and atherectomy.
Aim
Due to an increasing number of older, cardiologically high-risk patients as a result of comorbidities and the patients’ demand for less invasive procedures and faster recuperation we sought to evaluate early and long-term follow-up after first-in-man (FIM) revascularization of long SFA occlusion with the OCT Guided Ocelot Catheter.
Material and methods
The paper concerns a prospective single-center nonrandomized first-in-man registry of patients with CTO SFA and severe claudication treated with the Ocelot Catheter (before receiving the CE mark). The purpose of this study was to evaluate short- and long-term outcomes after CTO SFA revascularization using the Ocelot catheter. The periprocedural outcomes as well as vessel patency at 6 months and 5 years of the intervention were evaluated using angiography and Doppler ultrasound (according to study protocol – AVI-OCT-10003 v.0; PB/WKWM/21/2010), respectively.
Patient eligibility and study requirements
Patients were considered eligible for the study if they were diagnosed with chronic limb ischemia graded as Rutherford 3; target vessel diameter > 5 mm and target lesion total occlusion. The exclusion criterion was target limb necrosis, i.e., stage 6 according to the Rutherford classification. All patients were maintained on aspirin (75–150 mg), clopidogrel (75 mg) and statin 2 days prior the intervention. Other medications were prescribed at the physician’s discretion. An intra-arterial bolus of 5000 IU heparin was administered during the interventional procedure.
Definitions
The CTO was defined as complete occlusion of the artery. Vessel patency was defined as diameter stenosis ≤ 50%. Inability to cross the lesion, perforation, no reflow and bailout stenting were evaluated as periprocedural outcomes. Serious adverse events comprised cardiovascular death, myocardial infarction, stroke, non-target lower limb vessel revascularization and amputation.
Description of Atherectomy System
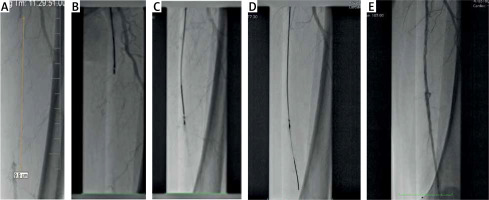
Ocelot is a unique CTO-dedicated atherectomy system with built-in OCT for intravascular imaging. This catheter utilizes spiral-fluted wedges to corkscrew the CTO cap while real time OCT provides visualizations to facilitate intravascular navigation. OCT is located at the tip of the catheter allowing high-precision navigation without contrast use. Ocelot has a length of 110 cm and a crossing profile of 2 mm, and is compatible with 6 Fr sheaths and 0.014-inch guidewires (Figure 1 A). The second part of the Ocelot system is a Lightbox console, which transmits a laser to the catheter through an optical fiber. The Lightbox creates an image, which can be presented in 2 modes: 1 – sector, 2 – waterfall view.
Results
Patient characteristics and procedure
Baseline demographic and clinical characteristics of 10 patients are shown in Table I. The cardiovascular risk factors were prevalent. One patient had a history of myocardial infarction. A history of stroke was present in one patient with minor disability. Mean SFA occlusion (measured in QVA) length was 74.5 ±32.5 mm.
Table I
Baseline demographics, comorbidities and lesion characteristics
Contralateral access was used in 50% of the patients due to iliac and common femoral artery anatomy. The success rate was 90%. In one patient the catheter could not cross the CTO due to massive calcification. Balloon angioplasty was performed in all cases; bailout stenting for vessel dissection was performed in 4 out of 9 patients (Figures 1 B–E). In all cases Astron Pulsar stents were used. No protection devices were used. Artery perforation and no-reflow syndrome did not occur. No patients developed access-site complications. The mean duration of the procedure and contrast agent volume were 100 ±48 min and 433 ±139 ml, respectively. In the authors’ opinion this fact is strongly connected with the type of this study, i.e. first in man, and is not representative for current Ocelot procedures. During the hospital stay, particular attention was paid to hydration of patients before and after the procedure. In the group of patients, no nephropathy requiring dialysis was observed during both 30-day and 5-year follow-up.
Outcomes and follow-up
30-day outcomes
All 9 patients after successful procedure were claudication-free, i.e., asymptomatic (Rutherford 0). In all patients color duplex scan was performed, without symptoms of restenosis in examinations. The patient after a failed procedure was put on medical treatment (ASA, cilostazol, statin); the claudication distance was approximately 100 m. All 9 patients were maintained on medical treatment – ASA 75 mg, clopidogrel 75 mg, statin 40 mg.
6-month angiography follow-up
Angiographic follow-up (according to the study protocol) performed at 6 months of the procedure revealed 89% patency of the target vessel in 8 out of 9 patients. Follow-up angiography revealed significant although non-occlusive restenosis and re-CTO of the target lesion in SFA in three and one patient, respectively. In all patients with restenotic lesions the primary treatment included CTO crossing and balloon angioplasty without stent placement. At 6 months, claudication distance decreased below 100 m and repeat balloon angioplasty was therefore performed. Percutaneous revascularization was also attempted in the re-CTO case but proved unsuccessful. Consequently, common and deep femoral artery endarterectomy with patch plasty was performed.
5-year Doppler ultrasound follow-up
Doppler ultrasound performed at 1, 2 and 5 years of the primary intervention did not reveal important target vessel restenosis above 50% and peak systolic velocity (PSV) > 1.5 m/s. The primary and primary-assisted patency was 89%. During a 5-year follow-up, four peripheral percutaneous interventions and one femoro-popliteal bypass surgery were performed in non-target limbs. There were no cardiovascular deaths, myocardial infarction or stroke and no amputation was required. Throughout the follow-up all patients were maintained on medical treatment – ASA 75 mg, clopidogrel 75 mg, statin 40 mg. The long-term double antiplatelet treatment in the H2 blocker cover was conducted in accordance with the authors’ belief that the prevention of cardiovascular events was more effective.
The target limb of eight patients remained asymptomatic (Rutherford 0) from month 6 and throughout year 5 of the primary intervention. Claudication distance in the patient after patch plasty repair remained unchanged (i.e., approximately 100 m).
The patient in whom CTO crossing failed due to massive calcification underwent femoro-popliteal bypass surgery 2 years after the primary intervention due to shortening of claudication distance below 50 m.
Discussion
The CTO SFA recanalizations are mostly performed using the subintimal approach which undoubtedly results from lesion type and operator experience. New tools specially designed for intraluminal interventions allow efficient and safe treatment of long and heavily calcified CTOs that would previously be qualified (TASC II) for surgical repair.
The current study is a direct observational first-in-man registry of patients with CTO revascularized with an optical coherence tomography (OCT) Guided Ocelot Catheter (Avinger). According to available research, the registry for the first time describes very long-term follow-up after OCT guided CTO of SFA revascularization. Moreover, this study was designed to obtain the CE (Conformité Européenne) mark for the OCT Guided Ocelot Catheter (Avinger). All subjects included in the study were high cardiovascular risk, symptomatic patients with long SFA CTO. Those factors determined that they were high-risk for open bypass surgery. Nonetheless, long SFA CTO was actually classified as TASC D, which should be treated with the open surgical procedure rather than endovascular [9]. The OCT Guided Ocelot Catheter is designed for chronic total SFA occlusion revascularization and 10 μm image resolution allows differentiation between various healthy arterial structures including media and adventitia, and diseased arterial walls including atherosclerotic plaque [10]. The high resolution visual guidance would minimize media injury which could yield better long-term outcomes and patency [11].
The first large trial designed to assess safety of the Ocelot Catheter was the single arm prospective multicenter CONNECT study which included 84 patients with femoropopliteal CTO. Efficiency defined as successful CTO crossing with the device was achieved in 89% of included patients. Perforations were reported in 5% of cases, resolved with prolonged balloon occlusion. The clinically significant perforations were not observed [12]. In the CONNECT II study outcomes were very similar with 72% successful crossing with Ocelot alone, whereas 97% were successful when combined with assist or re-entry devices [13]. In the prospective, single-arm, multicenter Vision study of the Ocelot Catheter combined with adjunctive therapy such as stent implantation or used alone outcomes were encouraging. One hundred fifty-eight patients were enrolled, and 198 lesions were treated. The safety profile was satisfactory with no clinically significant perforations, 0.5% dissections and 2% embolic events. The clinically driven repeat target lesion revascularization was 6.4% in the 6-month follow-up [14]. Stavroulakis et al. published long-term outcomes of endovascular revascularization with the Ocelot Catheter combined with local antimitotic drug delivery by drug-eluting balloons (DEB). The results are encouraging; in the 33 patients and 37 lesions only 5% had TLR in 12 months and low periprocedural complications [15]. Moreover, OCT Guided Ocelot Catheter is suitable even for very long, calcified CTOs involving the whole SFA [16].
Those studies confirm our observations on safety and feasibility outcomes. Moreover, the long-term follow-ups are also encouraging and comparable to our study. When comparing re-entry devices, the safety and feasibility are similar if they are used in short CTO SFA. When performed in longer CTO the efficiency ratio is more encouraging when orbital atherectomy with OCT is used [17].
Nowadays, when local drug delivery is more and more efficient this technology may by combined with orbital atherectomy in PAD revascularization. Early reports on the combination of plaque modification with atherectomy and subsequent drug-eluting balloons (DEB) seem to be promising [18, 19]. Novel technologies, including local drug delivery nano-technology, may soon become available for the follow-up treatment of plaque modifications after atherectomy.
Study limitations
The main drawbacks of this analysis are those inherent to any single-center observational study [20]. The first-in-man nature of this study could have a significant influence on long-term outcomes due to the learning curve. This is a hypothesis-generating study rather than conclusive.
Conclusions
This first-in-man study with long-term follow-up showed that the OCT Guided Ocelot Catheter has a high success rate of CTO recanalization and very promising long-term outcomes. Nevertheless, the long-term outcomes should be evaluated in comparison with re-entry devices and drug technology in a prospective randomized trial.