Summary
In stroke survivors matched for stroke severity and for major health-related quality of life (HRQoL) determinants beyond stroke, carotid revascularization has a major positive impact on patient-reported HRQoL. The early post-revascularization improvement is greater with carotid artery stenting (CAS), due to less bodily pain and better physical functioning and role-physical as well as better role-emotional and higher general well-being scores. At 6 months the remaining carotid endarterectomy (CEA)-CAS difference is small – to become not statistically significant at 12 months. As surgeons are increasingly performing endovascular procedures, these findings may affect – on top of major adverse clinical events associated with CEA and CAS – the patient and physician preferences, though they require confirmation in larger cohorts and multi-centre settings. In essence, our results show that in survivors of CS-related stroke, carotid revascularization is associated with a significant improvement in patient-perceived HRQoL, supporting its use in stroke survivors who were not lucky enough to benefit from carotid revascularization to prevent the devastating event.
Introduction
Ischaemic stroke has a profound impact on health-related quality of life (HR-QoL) [1]. Despite the progress in pharmacotherapy and increasing penetration of atherosclerosis-modifying drugs such as statins and angiotensin-converting enzyme inhibitors [2], atherosclerotic carotid artery stenosis (CS) continues to be a major modifiable risk factor of ischaemic stroke [3]. With 15–25% of ischaemic strokes occurring in relation to CS [3, 4], the CS contributes importantly to the individual and social burden of stroke disability [3].
Carotid revascularization, either surgical (carotid endarterectomy – CEA) or endovascular (neuroprotected carotid artery stenting – CAS), has been demonstrated to reduce stroke risk in both symptomatic (i.e., after cerebral/retinal stroke or transient ischaemic attack) and asymptomatic subjects with CS [2, 4] but there is an ongoing debate on whether and which patient subsets should be revascularized [2, 4, 5]. Pharmacotherapy may reduce stroke risk but is not, on its own, sufficient to prevent strokes, because CS-related strokes continue to occur, in vascular clinic patients, at a yearly rate of 2.5–2.9% despite guideline-based pharmacotherapy [2, 5]. One surprising view is that despite the recent large-scale (n = 3120 patients) level-1 evidence from the Asymptomatic Carotid Surgery Trial (ACST-1) that demonstrated a profound (≈ 50%) stroke reduction with CS revascularization irrespective of lipid-lowering pharmacotherapy [6], revascularization “should” be limited, in principle, to patients after occurrence of symptoms of cerebral (or retinal) ischaemia such as transient ischaemic attack (TIA) or stroke [4]. According to this concept, effective treatment becomes provided, to many stroke survivors, already too late because the cerebral damage that has occurred is largely irreversible [1, 7–9]. Thus although CS revascularization does have a role in secondary stroke prevention, its role in reducing the post-stroke physical disability is limited once a major stroke-related disability has already occurred [1, 2, 7–10].
It is debated not only whether and which CS patients should be revascularized but also which of the two revascularization methods, CEA or CAS, should applied as a first-line interventional treatment modality. Outcomes of this debate are driven, in principal, by which clinical endpoints are (subjectively) considered to be more important than others. A recent meta-analysis of 6526 patients from 5 trials with a mean follow-up of 5.3 years showed that while the composite outcome of periprocedural death, stroke, myocardial infarction (MI), or non-periprocedural ipsilateral stroke was not significantly different between CAS or CEA (OR = 1.22; 95% CI: 0.94–1.59), the risk of any periprocedural stroke plus non-periprocedural ipsilateral stroke was higher with first-generation carotid artery stents (CAS) (OR = 1.50; 95% CI: 1.22–1.84) [11]. The higher stroke risk with CAS was mostly attributed to periprocedural minor stroke; OR = 2.43; 95% CI: 1.71–3.46) [11]. The CAS, however, was associated with a significantly lower risk of periprocedural MI (OR = 0.45; 95% CI: 0.27–0.75), cranial nerve palsy (OR = 0.07; 95% CI: 0.04–0.14) and the composite outcome of death, stroke, MI, or cranial nerve palsy during the periprocedural period (OR = 0.75; 95% CI: 0.60–0.93) [11]. Indeed, 10-year data from the Carotid Revascularization Endarterectomy Versus Stenting Trial (CREST-1) have recently shown that patients with periprocedural stroke were at 1.74× the risk of death compared with those without stroke (adjusted hazard ratio = 1.74; 95% CI: 1.21–2.50; p < 0.003) while patients with MI were at 3.61× increased risk of death compared with those without MI (adjusted hazard ratio =3.61; 95% CI: 2.28–5.73; p < 0.0001) [12]. Stroke, however, has a higher negative impact on HR-QoL than MI or heart failure [1, 12–15].
There is increasing understanding that quality of life (rather than major adverse clinical endpoints in isolation) should be receiving more attention in contemporary evaluation of the overall clinical evidence on CEA and CAS, and in determining the relative place of each in everyday clinical practice. Unfortunately, data on health-related quality of life (HR-QoL) after CEA and CAS are limited. In particular, no comparison of HR-QoL evolution in symptomatic CS patients treated with CEA vs CAS has been performed so far.
Aim
We have evaluated the effect of CS revascularization by CEA vs. CAS on HR-QoL in stroke survivors by performing a cross-sectional analysis controlled for major (other than stroke) HR-QoL determinants, and controlled for the subject’s ability to adequately express their views by assessing subjects with their ischaemic cerebral infarct location in the non-dominant hemisphere.
Material and methods
We performed a cross-sectional analysis based on 856 carotid revascularization procedures performed over 3 years in the Department of Vascular Surgery and Endovascular Interventions at John Paul II Hospital in Krakow. Symptomatic carotid stenosis (n = 417) accounted for 48.7% of the total carotid revascularization procedures. The decision on which particular method of carotid revascularization (CEA vs. CAS) to apply in which patient was, in general, guideline-based [4], but the patient and physician preference was also considered. Eversion CEA was conducted under general anaesthesia with brain protection using shunt devices. CAS was performed using conventional carotid stents and endovascular cerebral protection devices according to the “tailored CAS” algorithm [16] under local anaesthesia in the access site (typically groin). CEA and CAS were performed by experienced vascular surgeons and interventionalists who had previously performed a minimum of 100 procedures. During the study period there were 437 CEAs and 419 CAS procedures.
Health-related quality of life (HR-QoL) was assessed, as part of the Quality of Life in Carotid Revascularization Project carried out in our institution, using standardized questionnaires. The questionnaires were administered in written fashion. Medical Outcomes Study Short Form 36 (SF-36) [17] was used to determine the patients’ perception of their health status. SF-36 is a 36-item, commonly used health survey that assesses eight dimensions of health status, including physical health (4 domains) and mental health (4 domains), with a weighted “value” of complaints (and their severity) in each domain (physical functioning, role-physical, bodily pain, and general health in the physical health section; vitality, social functioning, role-emotional and well-being in the mental health section) [17]. SF-36 has been validated to evaluate HR-QoL in stroke patients [1, 15] as well as in patients with CS subjected to revascularization [13, 14, 18]. Scores for the SF-36 are in the range 0–100; a difference of 5–10 points is considered a clinically important change for an individual, whereas smaller differences may be important for group comparisons [13, 17]. Moreover, the SF-36 provides a summary scale (“total”) for overall physical and mental health, for which individual differences of 2.5–5 points are considered statistically significant [13, 17]. In the present investigation, SF-36 outcomes were presented in a fashion where higher scores correspond to fewer complaints (and/or their smaller severity), indicating better health status and higher quality of life [13, 17, 18].
The patients were encouraged to fill in a SF-36 questionnaire (Polish language version [19, 20]) within 7 days before carotid revascularization, and during the three scheduled post-revascularization visits in the vascular outpatient clinic; 14 days after, 6 months after, 12 months after CEA or CAS. Neurologic examination was performed at baseline, prior to discharge and at 12 months. The return rate of completed SF-36 forms was 94.1% at baseline, 85.8% at 14 days, 81.5% at 6 months, and 77.9% at 12 months.
To evaluate the effect of CS revascularization by CEA vs. CAS on HR-QoL in stroke survivors, we controlled for not only the size of neurologic deficit at baseline but also for major (other than stroke) HR-QoL determinants [19–24]. Because periprocedural stroke, similar to that within the follow-up period, has a negative impact on HR-QoL [13], subjects with any stroke during follow-up (or with any other major adverse clinical event) were not included in an analysis aimed to evaluate the “clean” effect of either of the CS revascularization methods on the quality of life. To control for the subject’s ability to adequately express their views, the present analysis was limited to patients with cerebral infarct location in the non-dominant hemisphere (for study group homogeneity, only those with left-dominant hemisphere and right-hemispheric stroke were considered).
The nearest neighbour search algorithm [25] was applied to identify similar patient pairs (CEA-CAS), matched for National Institutes of Health Stroke Scale (NIH-SS) score as well as presence of major known determinants of HR-QoL other than stroke including age and sex, educational and marital status, hypertension, diabetes, and the presence of heart failure [19–24].
The study was approved by the local Ethics Committee, and all subjects provided informed written consent.
Statistical analysis
The results were presented separately for the CAS and CEA cohort. Differences between qualitative variables were calculated using the χ2 test. For eight SF-36 domains the total score for all measurement points (baseline, after 2 weeks, 6 months and 12 months) was presented as the mean with 95% CI for the mean. In each study cohort, differences between the time points were checked with one-way repeated measures ANOVA, whereas differences between the CAS and CEA cohort were checked by the t-paired test for all measurement points separately. For all analyses, p-values of < 0.05 for two-sided tests were considered statistically significant. All calculations were carried out using IBM SPSS Statistics 24.
Results
The nearest neighbour search identified, amongst 667 carotid revascularization subjects who returned completed SF-36 questionnaires by 12 months after the procedure, 42 (CEA-CAS) pairs of left hemispheric-dominant survivors of right hemispheric stroke who – besides the degree of neurologic impairment as per the NIH-SS – could be matched for age, sex, marital and educational status, hypertension, heart failure and diabetes, and who (1) underwent an uneventful carotid revascularization procedure, (2) had no major adverse clinical events throughout 12 months, and (3) completed the SF-36 questionnaire (i) within 7 days before, (ii) 14 days after, (iii) 6 months after, and (iv) 12 months after carotid revascularization.
Clinical characteristics of the CEA and CAS cohort are provided in Table I.
Table I
Characteristics of the CEA cohort and CAS cohort
| Parameter | CEA (n = 42) | CAS (n = 42) | P-value |
|---|---|---|---|
| Sex: | – | ||
| Men | 60% (25) | 60% (25) | |
| Women | 40% (17) | 40% (17) | |
| Age: | |||
| Men | 70.4 ±5.1 | 71.3 ±4.1 | 0.375 |
| Women | 73.3 ±4.3 | 74.3 ±4.7 | 0.312 |
| Marital status: | 0.660 | ||
| Married or with partner | 52% (22) | 57% (24) | |
| Alone | 48% (20) | 43% (18) | |
| Education: | 0.660 | ||
| Obtained degree or professional qualification | 55% (23) | 60% (25) | |
| Unqualified | 45% (19) | 40% (17) | |
| Hypertension | 93% (39) | 98% (41) | 0.308 |
| Diabetes mellitus | 38% (16) | 43% (18) | 0.660 |
| Coronary artery disease | 57% (24) | 62% (26) | 0.660 |
| Heart failure | 19% (8) | 31% (13) | 0.208 |
| Peripheral vascular disease | 52% (22) | 57% (24) | 0.660 |
| Left dominant hemisphere | 100% (42) | 100% (42) | – |
| Right hemispheric stroke ≤ 6 months and RICA stenosis ≥ 60% | 100% (42) | 100% (42) | – |
| h/o left hemispheric stroke | 0% (0) | 0% (0) | – |
| Occlusion of LICA* | 2.3% (1) | 29% (12) | < 0.001 |
| CT evidence of right hemispheric infarct | 100% (42) | 100% (42) | – |
| CT evidence of left hemispheric infarct | 0% (0) | 0% (0) | – |
| NIH-SS | 5.4 ±2.8 | 5.9 ±3.1 | 0.440 |
The HR-QoL results are given for CEA vs. CAS respectively. At baseline, HR-QoL perception was low and was similar in both groups (30.8 ±4.6% vs. 29.1 ±3.9%, CEA-CAS difference of 1.7 ±0.9%; p = 0.68). Hospitalization length was significantly higher in the CEA group (p < 0.001). Evolution of the HR-QoL total score is shown in Figure 1.
Figure 1
Evolution of total HR-QoL score in survivors of CS-related stroke, subjected to revascularization with CEA or CAS. Higher scores indicate better quality of life. Plotted values at each time point represent mean values for the eight SF-36 domains in each of the two study cohorts and associated 95% confidence interval for the mean
CEA – carotid endarterectomy, CAS – carotid artery stenting, SF-36 – Medical Outcomes Study Short Form 36, HR-QoL – health-related quality of life.
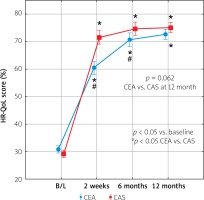
The CS revascularization was associated with a major HR-QoL improvement that was significantly greater in CAS than in CEA at 2 weeks after the procedure (60.4 ±9.2% vs. 71.5 ±6.2%, CEA-CAS absolute difference of –11.1 ±1.7% p < 0.001). At 6 months there was a further increase in the HR-QoL score that in each cohort was statistically significant both against the baseline value and against that at 2 weeks. However, at 6 months the CEA-CAS difference became significantly narrower (70.7 ±9.7% vs. 74.6 ±5.9%, absolute difference of –4.1 ±1.8%; p = 0.026) than 2 weeks after revascularization. At 12 months there was no statistically significant HR-QoL difference between the study cohorts (72.6 ±6.7% vs. 75.1 ±5.1%, absolute difference of –2.5 ±1.3%; p = 0.062), consistent with a similar effect of either CS revascularization method on the patient perception of their quality of life (Figure 1).
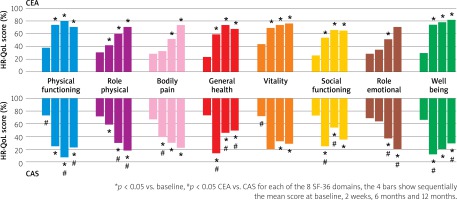
Analysis of the HR-QoL evolution in the individual SF-36 domains (Figure 2) showed that the early CEA-CAS difference was driven in the physical health domain by a less bodily pain and better physical functioning and role-physical and, in the mental health domain, by higher general well-being scores in CAS (p < 0.05 for all). Of note, there was no difference between CEA and CAS in the role-emotional and vitality domain at 2 weeks. Patients after CEA reported more frequently difficulties with eating/swallowing, neck pain, and headaches but less with walking and hemiparetic leg pain. These complaints were associated with the presence of a post-operative neck wound, and sometimes with the formation of a subcutaneous haematoma or ecchymosis. The areas of hypoaesthesia were mostly limited to the closest surgical scar area due to damage of the peripheral sensory skin nerve branches. In this case, the complainants were mainly men because of the limited superficial sensation in the skin area during shaving, consistent with the likelihood of cranial nerve palsy with CEA [11]. In the physical functioning domain, patients in the CAS cohort complained mainly of difficulties with walking because of the punctured groin pain. The site of the complaint was consistent with the site of the puncture (left or right common femoral artery), with a not infrequent haematoma (arterial access site closure devices were barely used in the study cohort, and manual compression followed by a pressure dressing was routine).
Figure 2
Evolution of HR-QoL scores in each of the eight SF-36 domains in survivors of CS-related stroke, subjected to revascularization with CEA or CAS. Higher scores indicate better quality of life. Bars represent mean values for each SF-36 domain in the two study cohorts at each time point
CEA – carotid endarterectomy, CAS – carotid artery stenting, SF-36 – Medical Outcomes Study Short Form 36, HR-QoL – health-related quality of life.
Discussion
Principal findings from this first study to evaluate HR-QoL evolution in stroke survivors undergoing carotid revascularization by CEA or CAS are the following:
Prior to revascularization, HR-QoL scores are very low in stroke survivors.
Carotid revascularization – both by CEA and CAS – has a major positive impact on patient-reported HR-QoL.
In stroke survivors subjected to CEA there is no initial post-procedural drop in HR-QoL that was previously reported to occur in mostly asymptomatic cohorts subjected to CEA.
Confounder-controlled analysis demonstrated that the early post-revascularization improvement is greater with CAS, due to less bodily pain and better physical functioning and role-physical in the physical health domain, and by better role-emotional and higher general well-being scores in the mental health domain.
At 6 months the remaining CEA-CAS difference in HR-QoL was small – to become not statistically significant at 12 months.
The debate on whether and which CS revascularization method should be applied to which patient populations has been focused mainly on (sub-)analyses of adverse clinical event components [4, 11]. In contrast, little is known about the quality of life in relation to the two carotid revascularization methods, particularly in subjects with symptomatic CS. In a recent meta-analysis on quality of life and functional status after carotid revascularization [26] the symptomatic status, surprisingly, was not taken into separate consideration. Similarly, in the CREST study, which included ≈ 50% symptomatic CS patients, the role of the symptomatic status on the quality of life appears undetermined [12, 13]. In CREST the overall CAS cohort had better outcomes for multiple components of the SF-36, with large differences for role-physical, pain, and the physical component (all p < 0.01) at both 2 weeks and 1 month [13]. On the disease-specific scales, CAS patients reported less difficulty with driving, eating/swallowing, neck pain and headaches but more difficulty with walking and leg pain (all p < 0.05) [13]. However, at 1 year there were no differences in any HR-QoL measure between CAS and CEA [13]. This line of findings is, in general, confirmed in our study focused specifically on stroke survivors with CS.
In stroke survivors with different stroke pathomechanisms, quality-adjusted survival is very low over 5 years after stroke [1]. Stroke often causes impairment of physical, cognitive, emotional, behavioural, and social functions, negatively affecting the quality of life [14]. Patients with symptomatic CS have been reported to exhibit significantly poorer HR-QoL than the general population [14, 27]. Our study cohorts exhibited, at baseline, extremely low HR-QoL – lower than reported in other studies of symptomatic patients and lower than might be considered to be expected (Figure 1). However, other studies have included, in the symptomatic cohorts, patients with transient ischaemic attacks the impact of which on HR-QoL is significantly smaller than that of stroke [1, 28]. Also, the stroke victims in the present study had a substantial neurological deficit (Table I). This low baseline HR-QoL level (Figure 1) may explain why, in contrast to the general expectation of an initial decline in HR-QoL after CEA [26], the CEA cohort in our study showed an increase HR-QoL after the procedure (Figure 1). The post-procedural increase in HR-QoL, however, was significantly smaller in the CEA cohort than in CAS patients (Figure 1), likely including the initial drop reported for mixed (asymptomatic and symptomatic) CEA groups [26].
Presentation with stroke has been suggested, by some, to predict poor outcomes after CEA [27], a finding not confirmed by data from the present study in relation to HR-QoL, with average scores, at 12 months, at the level of 70–75% (Figure 1). The magnitude of the improvement seen in the present study is consistent with the mean increase in patients’ quality of life evaluated previously in the Polish population using a 10-point Likert scale (increase from 3.9 to 6.3 points after CEA, p < 0.001) [29]. Indeed, our data are also consistent with the prior finding by the Oxford group that patients with symptomatic carotid artery disease undergoing an uncomplicated CEA perceive improved quality of life and overall health [7].
In the present investigation, by performing a matched-pair analysis with the maximally close-to-each-other CEA-CAS patient pairs identified using a nearest neighbour search algorithm [25], we controlled not only for the severity of neurologic impairment after stroke but also for major HR-QoL determinants (Table I). The guidelines and vascular surgery clinical practice favour CAS in higher surgical risk patients, such as those with heart failure or contralateral internal carotid artery (ICA) occlusion [4]. Accordingly, the CAS cohort in this study included numerically more patients with heart failure (31% vs. 19%, p = 0.208, Table I) and more subjects with contralateral ICA occlusion (2.4% vs. 28.6%, p < 0.001, Table I) despite our maximized optimal pair (CEA-CAS) match. Nevertheless, CAS patients reported a better overall quality of life at 2 weeks and 6 months after the procedure than those managed with CEA (Figure 1).
Strengths
One fundamental strength of this work is our unique investigational approach minimizing, in the evaluation of the relative value of CEA and CAS for the quality of life after carotid revascularization in stroke victims, the role of HR-QoL determinants other than the ischaemic brain infarct (Table I). Another is that by limiting the study subjects to those with non-dominant hemisphere stroke (Table I), we minimized the effect of stroke on the subject’s ability to adequately express the detailed HR-QoL characteristics required by the SF-36 questionnaire. Finally, we used the full SF-36 rather than its short version (SF-10) or a (limited) 10-point Likert scale (1 – poor to 10 – very good; evaluation with scores from 1 to 4 points classified as “poor”, from 5 to 7 – “good” and from 8 to 10 – “very good”) [29].
Limitations
The present study has several limitations that need to be acknowledged. First, our study was cross-sectional rather than randomized and (despite the very large baseline sample) the volume of matched pairs may be considered moderate. Secondly, we controlled for several major (Table I) but not all HR-QoL determinants other than ischaemic stroke. It is thus possible that conditions such as arthritis [23, 30] or lung disease [23, 31] could bias our findings of the effect of CEA vs. CAS on patient-reported HR-QoL (Figures 1, 2). Similarly, we were unable to take into consideration the effect of exercise [32, 33] or lifestyle [24], or that of social support [34] or living in a rural versus urban area [35]. Thirdly, it would be of interest to learn whether the post-revascularization improvement in HR-QoL (Figure 1) is affected by the degree of objective improvement as evidenced by the NIH-SS or modified Rankin score. Nevertheless, the size of the two study cohorts was not sufficient to test the potential relationship between the post-revascularization evolution of disability [36] and HR-QoL evolution. Moreover, neuromarker release [37] or cranial nerve injury that may occur with CEA but not with CAS (and may affect the quality of life [11]) was not routinely assessed. Finally, the present findings, in relation to HR-QoL after symptomatic CS revascularization using CAS, are applicable only to conventional (i.e., first-generation) carotid stents. The advent and increasing penetration of novel carotid stent technologies, including dual-layer stents that allow reduction of peri-procedural complications to ≈ 1% [36, 38, 39] and minimize the incremental risk related to the symptomatic status and other risk factors [40], may further impact the CEA vs. CAS relative effect on HR-QoL.
Conclusions
In stroke survivors matched for stroke severity and for major HR-QoL determinants beyond stroke, carotid revascularization has a major positive impact on patient-reported HR-QoL. The early post-revascularization improvement is greater with CAS, due to less bodily pain and better physical functioning and role-physical as well as better role-emotional and higher general well-being scores. At 6 months the remaining CEA-CAS difference is small – to become not statistically significant at 12 months. As surgeons are increasingly performing endovascular procedures, these findings may affect – on top of major adverse clinical events associated with CEA and CAS – the patient and physician preferences, though they require confirmation in larger cohorts and multi-centre settings.
In essence, our results show that in survivors of CS-related stroke carotid revascularization is associated with a significant improvement in patient-perceived HR-QoL, supporting its use in stroke survivors who were not lucky enough to benefit from carotid revascularization to prevent the devastating event.







