Introduction
Skin cancer is the most common malignancy, especially for elderly patients [1]. Several studies have estimated that non-melanoma skin cancer (NMSC), including basal cell carcinoma (BCC) and squamous cell carcinoma (SCC), affects more than 3 million Americans a year [2, 3]. For BCC, the main carcinogenic factor is ultraviolet light, which explains why most tumours are located on sun-exposed sites and the risk of developing BCC for white-skinned people is about 30% [4]. Surgical excision is the preferred therapy for non-melanoma skin cancer, with a reported < 5% local recurrence rate. But other loco-regional approaches such as radiotherapy (RT), cryotherapy, and photodynamic therapy are also available. Radiotherapy, both external beam and brachytherapy (BT), may be considered as the primary definitive treatment in patients who are unfit for surgery (locally advanced disease, comorbidities, or refused surgery) or when curative surgery is not possible due to a significant risk of poor aesthetic outcome [5]. When excision is incomplete and re-excision is not feasible, adjuvant RT is considered an option [6]. BT through the superficial deposition of dose within the tumour with a great saving of normal tissues has been reported to have an excellent cosmetic outcome. High-dose-rate (HDR) BT is a highly tailored treatment for lesions ≤ 5 mm deep; otherwise, interstitial BT is preferred. Patients treated with HDR-BT for non-melanoma skin malignancies show 85–100% of local control (LC) [7]. The new emerging electronic BT provides an alternative to radioactive isotope sources (usually iridium-192 [192Ir]) [8].
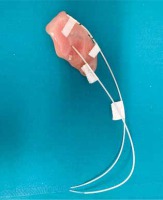
Mould BT allows the delivery of radiation therapy with moulds that are made to better fit the patient’s external surface. Moulds are mostly indicated for larger lesions or for those localized in areas such as the face or the scalp, where the skin thickness is not sufficient to implant catheters [9, 10]. Hence, several companies have developed applicators for treating superficial skin lesions with surface BT, but the applicators can also be created in the same Radiation Oncology Divisions, mostly with a mould or wax (Fig. 1).
Fig. 1
Tailored custom surface mould for treating a basal cell carcinoma of the ear with basal cell carcinoma brachytherapy
The aim of this study was to analyse retrospectively tumour control, toxicity, and aesthetic events for patients affected by NMSC treated with 192Ir-based HDR-BT using tailored custom moulds.
Material and methods
From January 2014 to December 2019, 37 patients affected by NMSC were treated with surface moulds with 192Ir-based HDR-BT at the Division of Radiotherapy, University of Pisa. We analysed 40 lesions with a depth ≤ 5 mm, measuring a median 2 cm (range, 0.3–6.0), located in 40.0% scalp (n = 16), 17.5% nose (n = 7), 25.0% face (n = 10), and 17.5% ear (n =7). A tailored custom surface mould was created for each patient, following the contour of the skin surface and size of the lesion, with 0.5–1-cm-thick bolus material with a median of 5 catheters (range, 1–9) spaced 5–10 mm apart and affixed externally. All patients underwent computerized tomography (CT) simulation (GE LightSpeed RT, GE Healthcare, MediPhysics Inc., Arlington Heights, IL, USA). CT images were acquired with 2.5 mm slice thickness in supine position, for a better delineation of the clinical target volume (CTV) [11], and dose to the normal tissue and structures, and afterwards it was transmitted to the planning system. The treatment program was to deliver ≥ 95% of the prescribed dose to the planning target volume (PTV) [11] up to a median total dose of 40 Gy (range, 25–50 Gy). Hence, the modal prescribed total dose was 40 Gy (48.5% of cases) in 8 fractions of 5 Gy, 2/3 fractions/week, with a minimum interval of 24 hours between fractions and with a biological effective dose (α/β = 10) of 60 Gy.
The equivalent dose in 2-Gy fractions (EQD2) was calculated by using the following formula:
EQD2 = D [( d+ α /β)/( 2 Gy+ α/β)],
where D is the total dose in Gy, d is the dose per fraction in Gy, and the α/β ratio is considered 10 Gy for the tumour [12].
The most used immobilization systems were tapes or thermoplastic masks. HDR-BT was delivered using a 192Ir source, HDR afterloader microSelectron Elekta. Each RT daily fraction must be administered in the presence of a radiotherapist highly experienced in BT, who takes care of the accuracy of the procedures and dose delivery by continuously monitoring the treatment via a video camera and audio connection with the treatment room.
Acute and late toxicities were evaluated according to the common terminology criteria for adverse events (CTCAE vs. 5.0) [13]. The cosmetic results were assessed at each follow-up visit according to the Radiation Therapy Oncology Group – European Organization for Research and Treatment of Cancer scale [14]. Follow-up visits were scheduled every 3 to 4 months for the first 2 years after BT completion, every 6 months for the next 3 years, and once a year after 5 years.
Results are presented as median or mean value for quantitative parameters. Frequencies and percentages were computed for qualitative parameters. Local control was evaluated with the Kaplan-Meier method.
All patients were periodically followed-up, until they died or up to December 2019.
Results
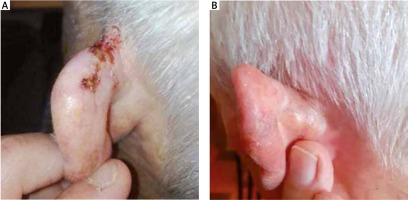
Non-melanoma skin cancer patients and lesions characteristics are shown in Table 1. Thirty-four lesions (85.0%) received a radical treatment and 6 lesions (15.0%) underwent adjuvant RT after surgery. Thirty-six lesions (90.0%) had a complete response (Fig. 2), and 4 (10.0%) had a partial response (PR) at clinical evaluation performed 3 months after treatment completion. Four (10.0%) PR were treated with HDR-BT after failed primary surgery and/or tailored chemotherapy.
Table 1
Patient and lesion characteristics
Fig. 2
A – An example of complete response of treatment of a basal cell carcinoma of the ear with high-dose-rate brachytherapy, using a tailored custom surface mould. B – After three months
Two (5.O%) PR lesions had local progression after a median time of 12 months (range, 6–23 months). Of these, 1 was affected by multifocal BCC of the face and 1 by multifocal BCC of the scalp, and both were treated with surgery and/or chemotherapy. The other 2 PR (5.0%) patients affected by multifocal BCC of the scalp had local and nodal progression and were treated with palliative RT and chemotherapy. No patients developed distant disease.
Four PR patients (10%) died of disease and 15 patients (40.5%) died of old age or comorbidities. The median follow-up of survivors was 25 months (range, 3–70 months). The 2-year LC rate was 90%. All 37 patients completed radiation treatment, and BT was well tolerated; no treatment was stopped for toxicity.
As shown in Table 2, the most common G1 toxicities were dermatitis (52%, n =21), pain (25%, n = 10), and ulceration (22%, n = 9). The only G2 acute toxicities were dermatitis and ulceration. The most common G1 late toxicities were fibrosis (17%, n =7), atrophy (15%, n = 6), and hypopigmentation (12%, n = 5). There was also no G3 or higher acute and late toxicity.
Table 2
Acute and late toxicity (CTCAE v. 5.0) in patients who underwent 192Ir–based HDR-BT, using customized surface moulds for NMSC
Excellent cosmetic results were observed in 65.0% of lesions (n = 26); only 1 treated lesion (2.5%) presented a poor cosmetic result (skin ulceration) (Table 3).
Discussion
The gold standard treatment for most primary NMSC cancer is surgical excision with histological control of excision margins, with recurrence rates for BCC from 2% to 8% at 5 years, as reviewed by Trakatelli et al. [6, 15]. Radiotherapy may be considered a primary definitive treatment in patients who are unfit for surgery (locally advanced disease, comorbidities, or refused surgery) or when curative surgery could lead to a poor aesthetics, but also RT could damage the surrounding normal tissues, resulting in toxicity or aesthetic changes, which are most often located within the radiation field [6]. Several RT techniques are used to treat skin cancer, such as superficial orthovoltage X-ray, electron beam, megavoltage photons, low-dose rate, or HDR-BT, and recently electronic BT [16]. In our study BT seems to be a highly effective and non-invasive therapeutic approach for NMSC, without high-grade toxicities, excellent cosmetic outcome, and good LC. We reported, as well as Jumeau et al., that no treatment was stopped for toxicity and there was no significant high-grade late skin toxicity [17]. The same results in terms of LC, late toxicity, and cosmetic effects, comparing BT with external beam radiation therapy (EBRT), were reported by Delishaj et al., with 62% excellent cosmetic results, 26% good results, and 5.5% poor results [18]. Guix et al. reported in 136 patients 10.3% G1 ulceration, 14% G2 erythema, and 92% good cosmetic results, 3 months after the completion of BT [9]. In a cohort of 200 patients of Gauden et al. G1 acute skin toxicity was detected in 168 treated lesions (71%), G2 in 81 (34%), and good or excellent cosmesis in 208 cases (88%). Late skin hypopigmentation was observed in 13 (5.5%) patients [19]. Unlike our cohort, a prospective study of Kalaghchi et al. reported, in 60 patients underwent radical or adjuvant BT, 6.7% G3 – 4 acute (3-month after BT) and no late toxicities (2 years after BT). The 2-year cosmetic results were good/excellent in 96.2% of patients [20].
The results of the studies on HDR-BT for NMSC showed LC rates from 83.3.% to 100%; the limit was due to few patients or different follow-up duration, cosmetic results, and toxicities reported. The different LC rates may be caused by tumour size (small vs. large), site (plain vs. curved surfaces), margin status, and histology (BCC vs. SCC) [18–22]. In our study, we showed no statistically significant difference in LC rates between 2 histological groups, perhaps due to the small number of patients (data not shown).
Gauden et al. [19] compared the LC of HDR-BT-treated patients (98%) with the LC of EBRT-treated patients and reported both LC rates from 87% to 100% with follow-up from 2 to 5 years. Drucker et al. [23] estimated the equivalent LC (95%) for Mohs surgery and EBRT. Our study reported a 2-year LC rate of 90%, according to other BT papers.
As found in the literature, we showed no statistically significant difference between definitive and adjuvant treatment groups (data not shown).
Several studies reported various dose prescriptions and target volume contouring: Jumeau et al., for example, used a PTV equal to CTV and prescribed 25 Gy in 5 fractions of 5 Gy for adjuvant treatments, 30 Gy in 6 fractions for exclusive treatments, and 8 Gy (one fraction) for palliative treatments [17]. Gauden et al. prescribed 36 Gy in 12 fractions and used PTV equal to gross tumour volume (GTV) plus 5–10 mm [19]. Casey et al. [7] used several doses, but 40 Gy in 10 daily fractions was the most commonly used dose fractionation (48.2%). Also, in our cohort we used different fractionation schedules, but the most common prescribed total dose was 40 Gy in 8 daily fractions.
In accordance with the literature, we considered a surface HDR-BT particularly tailored for elderly patients, who are often unfit for surgery or longer radiation treatments, or for performance status and/or concomitant comorbidities. A French study [17] on patients with a median age of 80 years, poor performance status, and scalp and face skin lesions, treated with customized applicators, reported a 2-year LC rate of 91%, with no high-grade skin toxicity and only G1 dermatitis [1, 24–26].
Conclusions
Our results suggest that surface mould HDR-BT is a safe and effective treatment modality for NMSC. BT is well tolerated with very poor high acute and late toxicities and good cosmesis. However, the limitation of our study as well as most literature studies is mainly due to the limited size and age of the population, because most late toxicity events can be noted only with a longer follow-up, while these frail patients did not continue follow-up or even died shortly after treatment was completed, limiting follow-up data.








