Introduction
Chronic viral hepatitis C, despite effective treatment, remains one of the most common causes of chronic liver diseases. It is estimated that 50 million people worldwide are infected with hepatitis C (HCV), with 1 million new cases emerging each year [1]. The HCV RNA prevalence rate in Slovakia is approx. 0.2%. However, its elimination is limited by several barriers [2, 3]. Transmission occurs via the parenteral route, with intravenous drug use (IDU) currently being the most common mode of transmission. The highest-risk populations include people who inject drugs (PWID), people in prisons and other closed settings, men who have sex with men, sex workers, and people living with human immunodeficiency virus (HIV) [4]. Incarceration is an independent and significant risk factor. It is estimated that at any given time, approx. 10.2 million people are imprisoned worldwide, with one in five prisoners incarcerated for drug-related crime [5]. The prevalence of HCV among prisoners is significantly higher compared to the general population. The estimated prevalence of anti-HCV among general prisoners is 26%, while among prisoners with a history of IDU, it reaches up to 64%. Globally, it is estimated that 2.2 million prisoners are anti-HCV positive [6]. The main cause of the high prevalence of HCV in prison settings is the criminalization of drug use in many countries. Additionally, harm reduction programs are often not available to prisoners, and risky behaviors such as drug use, non-sterile tattooing, and unprotected sexual intercourse frequently continue within the prison environment, contributing to the further spread of the infection [4]. These data suggest that treating chronic hepatitis C in the prison population is important for two reasons: preventing the spread of infection within the prison environment and reducing the risk of transmission by untreated individuals returning to the community after release. Prisons can be considered a controlled setting suitable for micro-elimination efforts. The World Health Organization has set key goals for the elimination of chronic hepatitis C, including reducing new cases to 350,000 per year, lowering new infections among PWID to 2 per 100 individuals, decreasing deaths from untreated HCV to 140,000 annually, diagnosing 90% of infected individuals, and ensuring that 80% receive treatment. One of the ways to achieve these targets is by eliminating hepatitis C in prison settings [7]. According to the latest EASL (European Association for the Study of the Liver) recommendations, all treatment-naïve and treatment-experienced patients with recently acquired or chronic HCV infection should be treated without delay, including individuals at high risk of transmitting HCV [4].
As of February 1, 2025, there were 7,078 incarcerated individuals in the Slovak Republic (SR) (6,566 men and 512 women), representing 130 prisoners per 100,000 inhabitants [8]. According to available data from single-center studies, the prevalence of anti-HCV antibodies in Slovak inmates ranges from 13.8% to 38.6% [9–11]. A screening program for chronic hepatitis C in Slovakia is currently being prepared. It is estimated that approximately 11% of prisoners with HCV receive treatment during incarceration [12].
Currently, two pangenotypic, panfibrotic treatment regimens for HCV are available in SR: sofosbuvir/velpatasvir and glecaprevir/pibrentasvir. In the past, genotype-specific regimens were also used, such as sofosbuvir/ledipasvir, elbasvir/grazoprevir, and ombitasvir/paritaprevir/ritonavir + dasabuvir. Strict treatment indication criteria requiring 12 months of drug use abstinence were in effect in SR, making it problematic to treat PWID, including inmates. This was particularly challenging for inmates with short sentences. A person diagnosed with HCV and with a history of IDU had to undergo toxicological urine tests every three months and a psychiatric evaluation confirming abstinence. It is estimated that before 2023, approximately 300 inmates with confirmed chronic hepatitis C but untreated were released into the community annually because they did not meet the treatment indication criteria at the time [12]. In June 2023 these indication criteria were first removed for sofosbuvir/velpatasvir and since March 2024 for both available treatment regimens. Now, every patient with HCV, including those with a history of IDU, can receive treatment without the need to prove abstinence. Moreover, patients with outstanding health insurance debt were unable to obtain treatment for selected chronic diseases, including chronic hepatitis C. On January 1, 2023, an amendment to the healthcare law (Amendment to Act No. 580/2024 of the Collection of Acts on Health Insurance) was adopted, which removed this barrier as well. Currently, even patients with healthcare insurance debt have approved treatment for chronic hepatitis C [13].
The main goal of our study was to assess the real-world experience of micro-elimination strategies for chronic hepatitis C in Slovak prisons with a focus on how the removal of treatment barriers affected patient treatment rates and access to care.
Material and methods
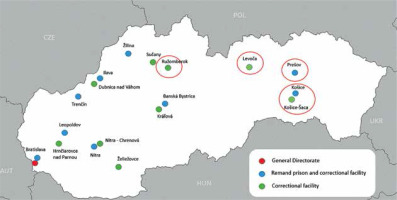
We retrospectively analyzed a cohort of 205 patients older than 18 years with confirmed chronic hepatitis C who were serving a sentence in five prisons in Eastern Slovakia. Four of these prisons fall directly under the jurisdiction of the Center for the Treatment of Chronic Viral Hepatitis at the Department of Infectology and Travel Medicine, Faculty of Medicine, P.J. Šafárik University, and L. Pasteur University Hospital. These include: a remand prison and correctional facility in Košice and Prešov, and correctional facilities in Košice-Šaca and Levoča. Additionally, our center is involved in the treatment of HCV in the correctional facility in Ružomberok. There are in total 1,659 prisoners in these five facilities [8] (Fig. 1). Screening for HCV in the mentioned correctional facilities is based on identifying prisoners with risk factors in their medical history, such as history of IDU, unsterile tattooing, risky sexual behaviors, etc. These prisoners are screened for anti-HCV antibodies, and subsequently HCV RNA testing is performed. After the diagnosis of chronic hepatitis C is confirmed, patients are referred to our center, and after treatment approval by the health insurance company, therapy is initiated. Patients are subsequently monitored by a physician from our center at monthly intervals during therapy.
Treatment with direct-acting antivirals was indicated and initiated in accordance with the currently valid indication criteria and treatment guidelines. Each patient signed a form titled Instructions and Written Consent of the Patient according to § 6 law No. 576/2004 of the Law Collection. As part of the demographic data collection, we recorded the patient’s age and sex. From the anamnesis, we evaluated the history of IDU. We also assessed selected virological (HCV RNA, HCV genotype) and histological (degree of fibrosis) parameters before treatment. The Cobas 5800 System (Roche Diagnostics, Basel, Switzerland) was used for quantitative detection of HCV RNA, and HCV genotyping was performed using a reverse hybridization-based test for differentiating HCV genotypes by the Versant HCV Genotype 2.0 Assay Line Probe Assay (Siemens, Berlin, Germany) at the Department of Microbiology Faculty of Medicine, P.J. Šafárik University, and L. Pasteur University Hospital, which is an accredited laboratory. The degree of fibrosis was assessed using the transient elastography method with the FibroScan Touch 502 device (Echosens, Paris, France) during the first visit in our center. Virological response was evaluated at the following time points: rapid virological response (RVR) – after the first 4 weeks of treatment; end-of-treatment response (ETR; defined as undetectable HCV RNA at the end of therapy) and sustained virological response (SVR; defined as undetectable HCV RNA 12 weeks after treatment completion). During treatment, patients were monitored on a monthly basis at our center. At the same time, prison healthcare staff supervised the regular intake of medications, which were dispensed to inmates on a weekly basis. We also analyzed the time from first HCV diagnosis to treatment initiation. The results are presented as mean values, medians, or percentages. For statistical analysis, a t-test and Mann-Whitney U test were used, with a p-value < 0.05 considered statistically significant. The study has been approved by Ethical Committee of L. Pasteur University Hospital, approval No. 2025/EK/02026. The committee waived the need for the specific patients’ informed consent due to the retrospective nature of the data collection. The study was performed in compliance with the Declaration of Helsinki.
Results
Between 2018 and 2024, a total of 205 inmates were referred for treatment evaluation to our center. The cohort consisted of 193 (94.1%) men and 12 (5.9%) women with a mean age of 32.3 ±7.9 years. A history of intravenous drug use was confirmed by 93.7% (n = 192) of the patients. One patient was co-infected with HBV and one with HIV (Table 1).
Table 1
Characteristics of study cohort
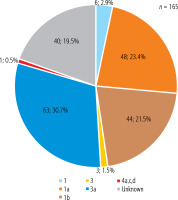
The HCV genotype was available in 165 patients. The most common genotype was 3a (63; 30.7%), followed by 1a (48; 23.4%) and 1b (44; 21.5%). Additionally, genotype 1 was identified in 6 patients (2.9%), and genotype 3 in 3 patients (1.5%). One patient was confirmed to have been infected with genotype 4a,c,d. The genotype was not determined in 40 patients (19.5%) (Fig. 2).
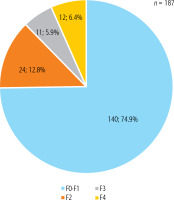
Liver fibrosis stage was assessed in 187 patients using the transient elastography method. In the remaining 18 patients, transient elastography could not be performed due to objective reasons; therefore, the Fib-4 score was used to estimate liver damage. Among the 187 evaluated patients, most (140; 74.9%) had liver fibrosis stage F0-F1, 24 patients (12.8%) had fibrosis stage F2, and 11 patients (5.9%) had fibrosis stage F3. Fibrosis stage F4 (liver cirrhosis) was confirmed in 12 patients (6.4%) (Fig. 3). In those with liver cirrhosis, the Child-Pugh score was assessed, and all patients had a Child-Pugh classification of A. No patient was diagnosed with decompensated liver cirrhosis or hepatocellular carcinoma. Among the 18 patients assessed using the Fib-4 score, none were diagnosed with advanced liver fibrosis. The Fib-4 score ranged from 0.21 to 0.94.
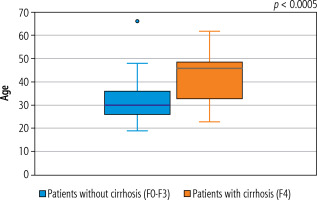
The relationship between patients’ age and the stage of liver fibrosis was analyzed by dividing the cohort into distinct fibrosis groups to assess statistical differences. Firstly, we divided patients into two groups: those without cirrhosis of liver (F0-F3, n = 175) and those with cirrhosis of liver (F4, n = 12). The average age of the no-cirrhosis group was lower (31.6 years) than the cirrhosis group (42.6 years). A boxplot visualization illustrates this trend, showing higher median age and a broader age distribution in the cirrhosis group (Fig. 4). These findings confirmed that cirrhosis of liver (F4) was significantly associated with older age in our cohort, p < 0.0005. The comparison of patient age between the mild fibrosis group (F0-F2, n = 164) and the advanced fibrosis group (F3-F4, n = 23) revealed no statistically significant difference.
Treatment with a direct-acting antiviral was started in 153 patients (74.6%). Of the 49 untreated patients (23.9%), 27 did not meet the treatment indication criteria at the time of evaluation and were subsequently lost to follow-up. Twenty-two patients met the indication criteria, but were not treated due to loss to follow-up, either because they were released from prison or transferred to another facility. Three patients refused treatment (1.5%). We recorded four cases of reinfection, and these patients were recommended for retreatment using a different regimen. Among all treated patients, 140 patients were treatment naïve and 13 patients underwent retreatment after previous unsuccessful therapy with pegylated interferon (Table 1).
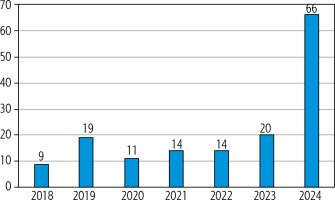
After the complete removal of treatment indication criteria in March 2024, we observed a significant increase in the number of treated patients per year, as shown in Figure 5. The number of treated patients in 2024 was more than three times higher compared to 2023 (66 patients vs. 20 patients), and in comparison with the previous average of approximately 15 treated patients per year, this increase is up to fourfold. By comparing patients indicated for treatment before and after the removal of indication criteria, we also observed a significant reduction in the time from the first positive test to the start of treatment, decreasing from 325 days to 91 days. This difference was statistically significant (p < 0.0005).
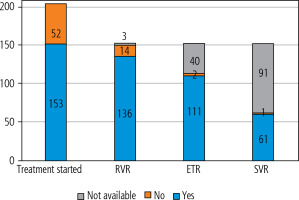
Among 153 treated patients, 15 patients (9.8%) received genotype-specific regimens (sofosbuvir/ledipasvir, elbasvir/grazoprevir), while 138 (90.2%) were treated with pangenotypic/panfibrotic treatment regimens (sofosbuvir/velpatasvir and glecaprevir/pibrentasvir). During treatment, the viral load was monitored. Among all treated patients, after the first 4 weeks of therapy, HCV RNA was undetectable in 136 patients (88.9%), while 14 patients (9.1%) still had detectable HCV RNA levels (ranging from 15 to 217 IU/ml). One patient had a higher viremia level of 18,500 IU/ml after the first month of therapy. In three patients, the result of HCV RNA after the first month of therapy was not available. Among the 153 treated patients, we were able to evaluate end-of-treatment response (ETR) in 113 patients. The remaining 40 patients were released from prison during treatment but were provided with the full course of therapy. Among these 113 patients, 111 (98.2%) achieved ETR. In the remaining two patients, HCV RNA was positive at the end of therapy, as well as after the first month of therapy. SVR was evaluated only in 63 patients, with 62 (98.4%) achieving SVR. The cascade of care is shown in Figure 6. For patients in whom ETR or SVR could not be assessed, the reason was loss to follow-up due to release from prison or transfer to another facility. No patient experienced treatment-related complications or severe drug interactions.
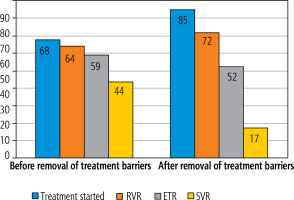
We compared the cascade of care before and after the removal of treatment barriers in March 2024. Before March 2024, 68 patients initiated treatment, 64 achieved RVR, 59 reached ETR and 44 achieved SVR. After the removal of treatment restrictions, the number of treated patients increased. The numbers of patients achieving RVR was 72 and ETR was 52. However, the number of patients evaluated for SVR decreased to 17, primarily due to loss to follow-up. Treatment failure was observed only in one case (Fig. 7).
Discussion
According to multiple studies, HCV is currently a disease predominantly affecting young men aged 25-44 years with a history of IDU [14]. These findings were confirmed in our cohort as well. The male-to-female ratio in the prison population in Slovakia is 13 : 1 [8]. In our study, 193 inmates with HCV (94.1%) were men, with an average age of 32.3 ±8.0 years. Similarly, in Hungarian prisons, the average age of incarcerated persons treated for HCV was 34.3 years, with a predominance of men (220; 84.3%), comparable to our findings. Hungarian authors reported that 99% of patients had a history of IDU, while in our cohort 192 inmates (93.7%) reported a history of IDU [15]. In contrast, Spanish authors, in their real-life experience study, confirmed a significantly higher average age of inmates with HCV (50.1 years), with half of the patients being between 51 and 60 years old, compared to the younger age profile observed in our cohort. Additionally, 70% of the inmates had a history of injection drug use [16].
The most common genotype in our cohort was genotype 3a (63; 30.7%), followed by 1a (48; 23.4%) and 1b (44; 21.5%). These three genotypes are also the most prevalent in our geographic region [17]. Similarly, Hungarian authors confirmed a high prevalence of these three genotypes, reporting 49.0% genotype 1a, 35.6% 1b, and 10.7% genotype 3 [15].
Given that the cohort consists predominantly of young patients, we can assume a shorter duration of infection, which aligns with the fact that 74.9% of patients had fibrosis stage F0-F1. Liver cirrhosis was confirmed in 12 patients, all of whom were in the compensated Child-Pugh A stage, with no cases of hepatocellular carcinoma. In our analysis of age and stage of liver injury stage, we found that a higher stage of liver fibrosis was significantly associated with older age. Patients with liver fibrosis stage F4 were significantly older than those with fibrosis stage F0-F3 (p < 0.0005).
Among other comorbidities, we investigated co-infections with HBV and HIV. HIV co-infection was detected in only one patient (0.4%), who has been receiving treatment for HIV infection for 13 years. Such a low prevalence of HIV co-infection is not surprising, as the Slovak Republic is among the EU and EEA countries with the lowest prevalence of HIV infection [18]. Similarly, in our cohort, HBV co-infection was detected in only one patient (0.4%). This patient has HBeAg-negative chronic HBV infection with low HBV DNA levels (< 2,000 IU/ml). After completing HCV treatment, we continued monitoring HBV DNA levels, which remained low, and hepatic enzyme activity normalized. The prevalence of HBV in Slovakia is low, estimated at 0.25%. Additionally, both hepatitis B and HIV are not widely prevalent among people who use drugs in the Slovak Republic [19].
Slovakia was among the countries with the strictest restriction for reimbursement of interferon-free direct-acting drugs for HCV infection. For individuals with a history of IDU, health insurance approval for treatment required 12 months of documented drug abstinence with toxicological urine tests every 3 months and a psychiatric evaluation confirming abstinence. After the evaluation, the therapy was fully covered by the health insurance company. Slovakia was the last country in the EU to completely abolish this barrier in March 2024. However, multiple studies have confirmed that SVR rates in people with a history of IDU are comparable to those in the general population without IDU history. These strict indication criteria made treatment in the population with a history of IDU quite problematic [20–22].
After the removal of treatment restrictions in Slovakia, we observed a significant increase in the number of treated patients. From 2018 to 2023, an average of 15 patients per year were treated. However, after the removal of treatment criteria (June 2023 for sofosbuvir/velpatasvir and March 2024 for glecaprevir/pibrentasvir), 66 patients were treated in 2024. Additionally, the time from initial diagnosis of anti-HCV positivity to treatment initiation was significantly reduced, from 325 days to 91 days. This difference was statistically significant (p < 0.0005).
Adherence to treatment during incarceration appears to be optimal, but after release from prison, many patients are lost to follow-up. A total of 49 untreated patients were lost to follow-up as a result of short prison sentences. Most of these untreated patients, 41 of 49 (83.7%), were lost to follow-up before 2023, when the requirement for 12 months of abstinence made treatment almost impossible for those with short sentences. Treatment was indicated and initiated in 74.6% of patients. The treatment regimen was selected based on: HCV genotype, potential drug interactions, and degree of liver damage. For patients with liver cirrhosis, sofosbuvir-based regimens were primarily chosen. If medical considerations did not dictate treatment selection, the duration of therapy was also considered in relation to the length of the prison sentence. For patients with short prison sentences, a shorter treatment regimen was preferred.
When evaluating ETR and SVR, patient loss to follow-up due to release or transfer to another facility was observed. ETR could be assessed only in 113 patients, and SVR only in 63 patients, yet high treatment efficacy was confirmed, with ETR of 98.2% and SVR of 98.4%. No treatment complications or severe drug interactions were recorded in any patient. Similar results were reported by authors from Slovakia in a real-life study on HCV elimination in one prison in western Slovakia. Out of 23 patients with confirmed chronic hepatitis C, treatment was initiated in 19 patients, achieving a 100% ETR rate. The remaining 4 patients were not treated due to short prison sentences and loss to follow-up [9].
The comparison of the cascade of care before and after the complete removal of treatment barriers in March 2024 shows a significant increase in treatment uptake and early virological response rates, although challenges in long-term follow-up after prison release remain. We suggest that after the removal of treatment barriers, even patients with very short sentences were referred to our center for treatment, which resulted in difficulties with follow-up after the end of therapy.
The treatment of HCV with direct-acting antivirals (DAAs) is particularly well suited for specific populations, as it is well tolerated and achieves high success rates. The prison environment appears to be an ideal setting for treating chronic hepatitis C. Inmates generally have better access to primary care, a structured routine, and fewer stressors compared to when they are released back into the community [23]. Harm-reduction programs in Slovak prisons are limited. Ongoing intravenous drug use is prohibited, and incarcerated individuals are provided with protective psychiatric services and addiction rehabilitation programs. Opioid substitution treatment and needle and syringe programs are not officially available in Slovak prisons [24]. Reinfection during incarceration is rare, and usually occurs outside of prison. In our cohort, we documented four re-infected patients.
The main goal of our research is to provide the first detailed analysis of the treatment of chronic hepatitis C in incarcerated individuals in Slovakia. Our study has several limitations. The research had a retrospective design. We included a relatively small number of patients from several prisons, mainly in Eastern Slovakia; however, our goal was to present real-life experiences. We were unable to detect SVR in all treated patients due to their release from prison.
Conclusions
The removal of treatment restrictions for chronic hepatitis C in Slovakia has led to a significant increase in the number of treated patients and a substantial reduction in the time from diagnosis to treatment initiation. The results demonstrate that micro-elimination efforts in prison settings are feasible and effective. These improvements highlight the critical role of removing unnecessary treatment barriers in enhancing access to care for high-risk populations. Our findings support the importance of HCV treatment programs in prison settings, both for reducing infection transmission within correctional facilities and for preventing untreated individuals from spreading the virus upon release. Given the high adherence rates and successful treatment outcomes, prison-based HCV treatment should be considered a key strategy in national elimination efforts. Continued efforts to enhance screening, facilitate access to treatment, and ensure post-release follow-up will be essential in further reducing the burden of HCV among incarcerated populations.