Introduction
Inflammation is a biological reaction to disrupted tissue homeostasis, triggered by any interfering factor [1-4]. The World Health Organization (WHO) considers chronic inflammatory diseases as the greatest burden to human health, affecting any organ of the body and among the major causes of death worldwide [3, 5]. However, its role in the pathogenesis of the reproductive system disorders is still debated [3]. Given the alarming epidemiology of chronic inflammation and the availability of suggestive data, non-microbial inflammation deserves further investigations to clarify whether it plays a key role (at least in part) for obstetric and gynecological disease.
This work aims to review the concept of impaired inflammatory state of the endometrium (IISE) as a multifaceted approach to the problem of endometrial inflammation, in contrast to the entity termed chronic endometritis (CE), currently restricted to infectious etiology and managed with antibiotics. It gives insights into the endometrial inflammation as a fundamental process, related to infectious and non-infectious factors, and involved both in physiologic and pathogenic conditions in obstetrics and gynecology, such as infertility, endometrial polyps, miscarriage and abnormal placentation-related diseases.
Methods
A non-systematic review was done through a search on the following databases: MEDLINE, EMBASE, Global Health, The Cochrane Library (Cochrane Database of Systematic Reviews, Cochrane Central Register of Controlled Trials, Cochrane Methodology Register), Health Technology Assessment Database and Web of Science, research registers (such as www.cliniclatrials.gov); we used the medical subject heading (MeSH) term Inflammation (MeSH Unique ID: D007249) in combination with: Endometrium (MeSH Unique ID: D004717); Endometritis (MeSH Unique ID: D004716). We selected pertinent original and review articles, published in English or French until 31 December 2019, focusing about CE, endometriosis, endometrial polyps, infertility and placenta-related complications, endometrial inflammation assessment and pharmacological targeting.
General features of the inflammatory response
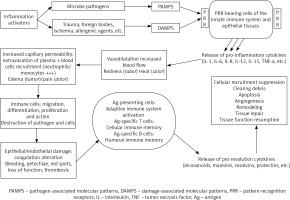
Microbial pathogens, but also mechanical trauma, extreme temperatures, ischemia, hypoxia, foreign bodies, pollutants, toxic, irritant, metabolic and allergenic agents are potent activators of the inflammatory response [1-11]. Pathogen-associated molecular patterns (PAMPS) and/or damage-associated molecular patterns (DAMPS) are derived either from pathogens or damaged cells and subsequently recognized by the pattern-recognition-receptors of the host’s cells [7-9, 12, 13]. The innate immune system, involving a variety of soluble molecules (cytokines) and immune cells, is first triggered, leading to a cascade of vascular and immune reactions. Subsequently, 4 to 7 days later, the adaptive immune system is eventually involved and consists of a T or B-cells-mediated specific response against the antigen [10, 14-17]. The return to homeostasis is orchestrated by active pro-resolution pathways, limiting damage to the cells by clearing leukocytes and tissue debris, thus allowing prompt restoration of tissue and vascular functions [18-22] (Fig. 1).
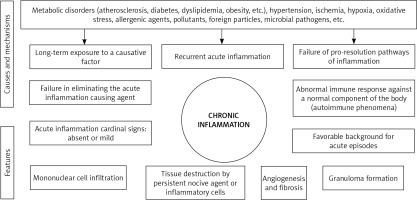
Fig. 1
Schematic illustration of acute inflammation orchestrated by the innate and adaptive immune system
Acute inflammation lasts less than 2 weeks and is characterized by 5 cardinal clinical signs: redness (rubor), heat (calor), edema (tumor), pain (dolor) and loss of function (functio laesa) [17]. Ideally, this results in a restitutio ad intergrum of the tissue. However, inflammation could evolve, after a subacute 2-to-6-week phase, to chronicity, displaying different symptoms, as the acute inflammation cardinal signs become mild or absent. These include constant fatigue, sleep disorders, depression, mood disorders, gastrointestinal complaints, weight gain, frequent infections, allergies and body pain [5, 23, 24]. Chronic inflammation is characterized by different cellular, vascular and histological features [5, 23, 24]. Furthermore, various causes and different mechanisms explain its pathogenesis [3, 5, 17, 20, 25-28] (Fig. 2). The challenging deeper understanding of the inflammatory response in its molecular, humoral, cellular and histopathological aspects should contribute to improved diagnostic and therapeutic strategies of inflammation-mediated pathologies.
The physiological inflammatory state of normal endometrium
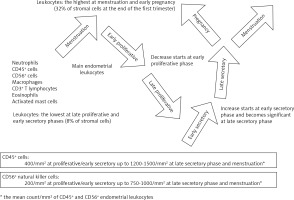
A vast amount of literature acknowledges inflammation as a key mechanism in physiological reproductive events, such as menstruation, ovulation, implantation and gestation [3, 17, 23, 29-35]. Endometrial physiology underlies a cyclical injury-healing process, displaying all the hallmarks of a self-limiting inflammation [17, 30]. Leukocytes are scattered in the stroma with aggregations around the glands [36] in a menstrual cycle-dependent fashion: their proportion among stromal cells ranges from 8% in proliferative endometrium to 32% in early pregnancy decidua [31]. Neutrophils are the prominent leukocytes during menstruation and start increasing after progesterone withdrawal in late secretory phase [37]. Macrophages, eosinophils and activated mast cells have a similar distribution, yet in smaller proportions [17, 30, 37]. The other main leukocytes of normal endometrium are CD56+ uterine natural killer (uNK) cells which account for 2% of stromal cells in proliferative endometrium, 17% during late secretory phase and more than 70% of endometrial leukocytes at the end of the first trimester of pregnancy where they play a role in trophoblast invasion and increased spiral artery blood flow [31, 33-36, 38, 39]. They are however characterized by gradual decrease of their cytotoxic activity until suppression in late secretory phase and early stage of pregnancy, thus allowing a favorable environment for implantation [33, 40].
Studies have revealed fluctuating time-dependent quantitative and qualitative changes of the recruited leukocytes (Fig. 3), the involved cytokines and the vascular aspects throughout the menstrual cycle and pregnancy [17, 23, 30-32, 34, 36-40]. The vascular changes mainly consist of vasoconstriction, vasodilatation, increased capillary permeability and angiogenesis. Interleukin (IL)-1, IL-6, IL-8 and peptide growth factors are demonstrated to have a key role in reproductive physiology and pathology, mainly via the prostaglandin biosynthetic pathway [17, 39, 41, 42]. Furthermore, studies have demonstrated the key role of active pro-resolution pathways aimed to control inflammation, thus allowing resumption of normal reproductive functions [17, 19-21]. Exacerbated activation of pro-inflammatory pathways and/or failure of pro-resolution pathways have already been described as a cause of reproductive disorders [17, 40].
Fig. 3
Time-dependent fluctuations of the main endometrial leukocyte populations throughout the menstrual cycle and early pregnancy
The impact of estradiol and progesterone on inflammation has also been demonstrated as their receptors are expressed in all the cells of the innate and the adaptive immune systems [5, 43-46]. They can suppress the production and secretion of several pro-inflammatory markers, such as IL-6 and IL-1β [45, 46]. Furthermore, progesterone decreases the cytotoxic activity of uNK-cells during secretory phase and pregnancy; and when decreased, it causes activation of some pro-inflammatory mediators and neutrophils recruitment in the endometrium [17, 33, 37, 47].
The concept of impaired inflammatory state of the endometrium
The delicate balance of pro-inflammatory and pro-resolution mechanisms in normal endometrium is well documented, thus explaining how over-activation of inflammation is as problematic as its suppression [17, 45]. An objective assessment of “normal endometrial inflammatory state” requires considering the time-changing quantitative and qualitative inflammatory patterns of the endometrium. However, to date this evaluation is totally operator-dependent with a consistent inter-observer variability. The diagnosis should rely on objective scores though, considering the stromal edema, the vascular patterns as well as the distribution and counts of the endometrial inflammatory markers for each reproductive phase. The observation of a “late-secretory-phase-pattern” during proliferative phase, for instance, may indicate an “impaired inflammatory state of the endometrium” (IISE).
IISE is a more appropriate terminology than “endometritis”, especially because the latter literally means “inflammation of the endometrium”, regardless of its causative factor. Yet, inflammation is already a consistent part of normal endometrium, the term “impaired” is thus more convenient for anomaly.
Depending on the nature of the causative factor and the attenuating circumstances, the IISE is transient, repeated or persistent. A transient IISE is an “out-of-phase” inflammatory disorder caused by any interfering factor. In fact, the monthly chance of pregnancy in the average young and healthy fertile human couple is around 30% [48]. A transient IISE partially explains this low percentage. If persistent or recurrent, an IISE can have considerable consequences on fertility. It is well established today that repeated acute inflammation is one of the forms of chronic inflammation [5].
However, to date, most acknowledged forms of impaired endometrial inflammation only consider germs or foreign bodies as causative factors. “Acute endometritis”, for instance, is defined as an acute response to pathogens. It is characterized by excessive exudation and leukocyte infiltration, predominantly granulocytes [23]. Specific endometritis is characterized by specific granulomatous lesions, such as the foreign body granuloma and the tuberculosis granuloma [5]. Immunological changes are also described in category E of abnormal uterine bleeding of the International Federation of Gynecology and Obstetrics (FIGO AUB-E), previously denominated dysfunctional uterine bleeding. Among these changes: impaired prostaglandin activity [41, 42, 46, 47], in addition to increased CD56+ cells during proliferative and early secretory phases (5% of stromal cells instead of 2%) [38].
“Persistent or recurrent IISE” is a more accurate terminology than CE. To date, CE is an anatomo-clinical entity displaying a number of paradoxical features. In fact, histological diagnosis of CE is mainly based on the presence of plasma cells, normally absent or very low in the endometrium [23, 49-54]. Therefore, this definition does not take into consideration the other impaired inflammatory parameters of the endometrium.
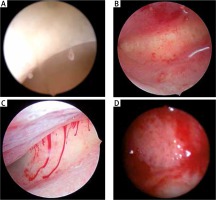
In addition, hysteroscopic diagnosis of CE is consensually based on the presence of strawberry pattern, focal hyperemia, hemorrhagic spots, micro-polyps and stromal edema [51, 53, 55, 56] (Fig. 4). However, it is well established in immunology that hyperemia and edema are typical cardinal signs for acute inflammation, not the chronic one which is rather silent [5, 24]. The presence of acute inflammation signs can be explained though by the fact that chronic inflammation makes perfect ground for repeated acute episodes. CE has already been histologically demonstrated in patients free of hysteroscopic signs, since negative predictive values range from 83 to 89%, i.e. in about 10 to 20% of patients with normal hysteroscopy, there was histological evidence of CE [55-57]. Limited observer agreement is a possible explanation. Nonetheless, recent literature concludes that absence of hysteroscopic features does not rule out CE [57]. Conversely, positive predictive values range from 42 to 94%, which is low in most studies [57]. This indicates either a transient IISE, or limited histological diagnostic tools due to the absence of a rigorous definition of endometrial inflammatory patterns.
Fig. 4
Hysteroscopic signs of what is termed chronic endometritis. A) micropolyps; B) hyperemia: red spots and petechiae; C) spontaneous bleeding and stromal edema; D) “chronic endometritis-like” redness and edema at day 14 of the menstrual cycle: progestin-only contraception
Furthermore, germs and/or altered microbiota are, to date, the unique cause admitted for chronic inflammation in human endometrium, and exclusively managed with antibiotics [35, 49, 53-56, 58-60]. Despite absence of evidence of pathogens in 25% of women diagnosed for CE [56], and despite hysteroscopic persistence of CE after antibiogram-based antibiotic therapy in 28% of patients [59], only germs are targeted, and never inflammation as an independent process, nor the other possible causes of impaired inflammation.
Migration of circulating B lymphocytes to the endometrial stromal compartment has been found to be induced by gram-negative bacteria [54]. However, the growing concept of microbiota has demonstrated a potential co-existence between microorganisms and healthy endometrium without causing disease [50, 60]. Underlying conditions predisposing to impaired endometrial inflammation are susceptible of altering the microenvironment, leading to impaired microbiota or pathogens, thus causing a vicious circle. For that matter, recently published results of a trans-sectional preliminary analysis of an ongoing case-control study [61], evaluated with DNA sequencing the endometrial and vaginal microbiota during the implantation window in patients suffering from repeated implantation failure (RIF) versus control group. Despite limited number of enrolled patients (28 in RIF and 18 in control group), one of the main findings was that endometrial microbiota, and not the vaginal one, showed a marked variance between the individuals. One confounding factor claimed to explain it was the different types of cycles (natural, hCG-triggered and hormone replacement cycles).
Regarding non-infectious inflammatory etiology, a higher prevalence of CE was correlated with endometriosis, a history of prolonged menstrual bleeding episodes, infertility, abortion and fallopian tube obstruction [52, 62]. Furthermore, there is evidence regarding the impact of non-infectious conditions on the endometrial fine-regulated inflammatory balance. In fact, multiple impaired inflammatory mechanisms of the endometrium have been demonstrated in heavy menstrual bleeding, abnormal implantation/placentation–related issues, such as infertility, recurrent miscarriage, congenital anomalies, stillbirth, preterm delivery, fetal growth restriction and pregnancy-induced hypertension. They consist of increased uNK-cells during proliferative and early secretory phases; higher percentage of uNK-cells expressing activation-antigens; elevated levels of T helper (Th) 1 cells contrasting with low Th2-cells and over-activation of the prostaglandin inflammatory pathway [33-35, 38-40, 45, 63-67]. There is clear evidence that controlled immune functions play a pivotal role in initiating a normal pregnancy, as they modulate the decidual response, cytokine balance, vascular adaptation, epithelial embryo attachment, trophoblast invasion, placental morphogenesis and also immune tolerance [45]. It is well documented that any disequilibrium in favor of suppressed or excessive inflammation in the endometrium significantly affects fertility or placental morphogenesis, subsequently leading to the above-cited gestational disorders [45, 67].
In addition, analysis of the acknowledged risk factors for preeclampsia by the U.S. Preventive Services Task Force guideline [68] shows that most of them are risk factors for chronic inflammation too: age of 35 years or older, body mass index greater than 30, chronic hypertension, type 1 or 2 diabetes, renal disease and autoimmune disease (such as systemic lupus erythematosus and antiphospholipid syndrome).
Metabolic disorders, such as diabetes, dyslipidemia, hypertension and obesity, have been ascertained to over activate the immune system, causing leukocyte activation, with greater numbers of leukocytes with pro-inflammatory phenotypes, thus explaining predisposition to chronic inflammatory diseases [25, 27]. They are well established risk factors for endometrial cancer too, which is already acknowledged as a chronic inflammation-mediated condition [17, 69, 70]. They are also significantly associated with increased risk of abnormal uterine bleeding, fertility issues as well as miscarriage and stillbirth, with remarkable improvement obtained with stricter metabolic control [71-73].
The oxidative stress, mediated by toxic habits (such as smoking), excessive pollution, allergies, sleep disorders and increasing age, is also demonstrated to predispose to chronic inflammatory state. Correlation with unexplained infertility, spontaneous abortion, recurrent pregnancy loss and preeclampsia has been documented [5, 74-77].
Additionally, the physiological effect of sex hormones on endometrial inflammation may explain both transient and persistent IISE in case of hormonal imbalances [17, 40-44, 78]. The borderline between “hormone-dependent” and “inflammation-mediated” disorder thus becomes blurred, as hormone imbalances induce impaired inflammation. Conversely, the latter alters hormone receptors’ expression both in epithelial and stromal cells, thus maintaining a vicious circle [54].
Stress was also demonstrated to induce localized inflammation in the uterus and cause an immune–endocrine disequilibrium involved in infertility, miscarriage, late pregnancy complications and impaired fetal development, mainly via elevated levels of cortisol as well as significant reduction in progesterone levels followed by impaired activity of uNK-cells [35, 78, 79].
In endometriosis, altered expression of estrogen and progesterone receptors has been associated with resistance to progesterone and over-expression of pro-inflammatory cytokines, such as IL-1, IL-6 and tumor necrosis factor α (TNF-α). Auto-antibodies against endometrial cells have also been described in endometriosis [32, 35, 63, 80-82]. Recent data found in patients with endometriosis a different behavior of some endometrial immune cells (macrophages, dendritic cells and T cells). The latter display more pro-inflammatory properties, compared to women without the disease [83, 84].
In adenomyotic nodules, high levels of inflammatory and neurogenic mediators have been reported [85]. The latter is demonstrated to be correlated with a higher incidence of implantation failure and abnormal placentation-related complications like miscarriage and pregnancy-induced hypertension [64, 86-88].
Auto-immune, allergenic and chronic inflammatory diseases directly interfere with the immune system and are correlated with decreased fertility [11, 25, 28, 35]. Even in the absence of a clinically overt autoimmune disease, anti-phospholipid, anti-thyroid, or antinuclear autoantibodies have been reported to be correlated with implantation failure and early pregnancy loss, especially when associated to vasculitis [65].
Semen-induced inflammation is another possible cause for IISE that has never been properly assessed in human reproductive medicine. In several animal species however, a transient post-mating uterine inflammation, triggered by semen components, is considered physiological [89-91]. It becomes pathological in case of inflammatory disequilibrium in favor of pro-inflammation, which is demonstrated to cause persistent endometritis, correlated with decreased fertility, requiring treatment like immuno-stimulants and corticosteroids [92]. In humans, the only acknowledged form of semen-induced inflammation is semen allergy, a rare and most likely underrated condition, poorly known by practitioners and thus, poorly investigated [93-96].
Furthermore, endometrial polyp (EP) is an anatomo-clinical entity that appears strongly related to IISE. It is a benign hyperplastic overgrowth of endometrial glands and stroma affecting 7.8% to 50% of women, with a prevalence of malignancy ranging from 1 to 3%, particularly in post-menopausal patients [97-103]. Spontaneous regression has been reported in 6.3 to 25% of cases [97, 104]. Although its etiopathogenesis is still unknown, it is usually presented as a hormone-related condition [103, 105, 106]. Yet, there is to date sufficient evidence indicating an inflammation-mediated pathogenesis, especially as hormonal imbalances are demonstrated to interfere with the inflammatory state of the endometrium [65, 99, 106]. Risk factors for EP are those for chronic inflammation: increasing age, menopause, obesity and hypertension [99, 102]. Higher prevalence of EP has been reported in women with diabetes mellitus [107-109], adenomyosis [110], endometriosis [111] and CE [112, 113]. In addition, studies have demonstrated increased expression of transforming growth factor β1 (TGF-β1) and vascular endothelial growth factor (VEGF) in EP [101, 113, 114]. Both are important pro-inflammatory mediators identified in many chronic inflammatory diseases [115-120]. The prevalence of EP in patients with IUD does not seem to have been assessed. Meanwhile, studies have reported cases for ParaGard copper and levonorgestrel IUD [121], despite therapeutic properties of this latter on endometrial hyperplasia [122]. The causal relationship between EP and infertility has been confirmed in only one randomized trial [102]. The acknowledged explanation is that EPs cause endometrial inflammation and sometimes constitute a mechanical barrier to sperm transportation or embryo implantation [102, 103, 105]. Current evidence rather indicates EP as an inflammation-mediated neo-process, subsequently maintaining the inflammatory state. This vicious circle partially explains why resection of EP is associated with increased rates of pregnancy in infertile women [100, 102].
Very importantly, gynecological malignancies, mainly cervical and endometrial ones, have also been demonstrated to be related to chronic inflammation, be it mediated by infectious or non-infectious conditions [17, 69, 70, 123]. This highlights the long-term importance of proper diagnosis and management of chronic inflammation.
IISE assessment and management: future perspectives
To date, there are no guidelines allowing proper assessment or management of endometrial inflammation. Elaborating clinical scores to assess the global inflammatory risk for patients appears pertinent. The patient’s clinical assessment should consider: her age, lifestyle, body mass index, blood pressure, atopy, stress factor, chronic inflammation signs, as well as a medical history of inflammation-mediated diseases and abnormal placentation-related disorders. Hysteroscopy should be scheduled during proliferative phase, as inflammation-hallmarks are the poorest at this stage.
Acute inflammation signs suggest either a transient or a persistent IISE. Absence of signs should not exclude persistent IISE as it is normally macroscopically silent. If semen-allergy is suspected, introducing the concept of “post coital hysteroscopy” appears relevant and remains to define in order to assess post-coital inflammation both macroscopically (redness, edema) and microscopically (assessment of mast-cells and eosinophils’ distribution). More attention has to be given to micro-hysteroscopy as it already defines vascular patterns for the endometrium [124]. Further research is highly needed, also for histopathology that should be revised and based on normal and impaired inflammatory patterns, and performed by trained pathologists. Despite its cost, the superiority of immunohistochemistry is indisputable as it allows a better identification of the positively stained cells for all antigens [52].
The optimal inflammatory balance allowing a proper implantation and progression of a healthy pregnancy seems quite delicate. In fact, spontaneous resolution of reproductive issues may occur without any medication. This highlights the importance of a healthy lifestyle, such as weight loss, controlling metabolic diseases, exercise and/or reduced stress.
Etiologic context must be targeted whenever possible. Anti-inflammatory therapy has to be considered as well. There is increasing evidence about its effectiveness in animal models for instance [19, 92]. In humans, several authors stress the necessity of introducing effective therapies capable of modulating the immune endometrial reproductive functions [30, 45, 80-82, 85]. In order to restore the normal endometrial inflammatory balance, minimal effective doses of pro-resolving anti-inflammatory drugs appear more relevant.
Although anti-inflammatory therapy has poorly been considered in the management of impaired endometrial inflammation, there is suggestive data highlighting the benefits of some molecules, such as low-dose acetylsalicylic acid (ASA). Its mechanism of action in the prevention of preeclampsia has never been elucidated [125] although there is evidence demonstrating its pro-resolving anti-inflammatory effect [22, 126]. Depending on the modalities of prescription, encouraging outcomes were reported regarding perinatal death, preeclampsia, fetal growth, preterm birth and fecundability [125, 127-129]. Furthermore, it is very often well-tolerated by women, fetuses and neonates [130].
Corticosteroids’ pro-resolving anti-inflammatory properties are widely demonstrated as well [67, 131-133]. Positive live birth outcomes in unexplained infertility and idiopathic recurrent miscarriage were obtained with low (or medium) dose of dexamethasone, prednisolone or prednisone, yet with low evidence level [67, 132, 134, 135]. Nevertheless, many adverse maternal and fetal effects were reported in the studies where corticosteroids were administered at high doses [45, 136]. Moreover, other molecules need to be assessed such as non-steroid-anti-inflammatory drugs (NSAIDs), whose prolonged prescription could oppose to follicle rupture though [137]. Prostaglandin E2 inhibitors have already been suggested to decrease the inflammatory environment caused by endometriosis and thus enhance endometrial receptivity for improved implantation [138]; metformin and dehydroepiandrosterone (DHEA), have been found to have anti-inflammatory properties [135, 139, 140]. Finally low-dose heparin, used in antiphospholipid autoimmune syndrome, has shown safe and effective anti-inflammatory properties in addition to its anticoagulant effect [141, 142].
Conclusions
Endometrial inflammation is a complex key-part of the endometrial physiology, regulated by a fine-regulated balance between pro-inflammatory and pro-resolving mechanisms, and involved in all reproductive events.
Infectious agents should no longer be considered as the only disturbing agent causing IISE. Any condition interfering with the immune system could result in a transient, repeated or persistent IISE. The latter could display very poor clinical and paraclinical signs. Optimal diagnostic and therapeutic tools remain to determine, yet, minimal effective doses of inflammation-pro-resolving therapies appear promising. Further research is needed for a better assessment of the inflammatory pathways involved in the reproductive tract, as well as the hysteroscopic and pathological patterns of normal and impaired endometrium, in order to improve prevention, diagnosis and treatment of obstetric and gynecological disorders.