Summary
Kidney transplant recipients (KTR) represent a high-risk population for cardiovascular disease and often have to undergo coronary intervention (CI), which can be associated with the risk of acute kidney injury (AKI). This retrospective single-center study presents the incidence and outcome of AKI after CI, analyzing 135 CIs in KTR between 2005 and 2015 at University Hospital Frankfurt. The incidence of AKI was 23%; 1-year-survival was significantly higher in the non-AKI group (94% vs. 81%, p = 0.02). AKI was associated with periprocedural bleeding events, whereas well-known risk factors such as contrast agent or diabetes were of minor impact in our study population.
Introduction
Kidney transplant recipients (KTR) undergoing invasive coronary angiography and percutaneous coronary intervention (CI) represent a high-risk cohort in different ways:
Both incidence and severity of coronary artery disease are more pronounced in patients suffering from chronic kidney disease (CKD) [1–3]. KTR often experienced long-term renal replacement therapy (RRT) while awaiting kidney transplantation with a known adverse impact on atherosclerotic burden and a high coincidence of cardiovascular risk factors such as diabetes or hypertension [1, 4, 5]. Even after successful transplantation, cardiovascular disease is highly prevalent in this population and the most frequent cause of death in KTR [3–6].
When performing CI in KTR, the interventional cardiologist is often faced with a more severe grade of coronary artery disease (CAD) with calcified lesions bearing a higher risk of prolonged CI procedures and procedural complications, necessitating larger volumes of nephrotoxic contrast dye than standard procedures.
The accurate choice of arterial access is an utmost safety issue for every patient undergoing coronary angiography and CI [7–10]. In recent years, the transradial access has gained a IA recommendation in the guidelines due to reduced bleeding rates translating into improved survival [11]. However, end-stage CKD and KTR are still often deemed unsuitable for transradial access as the risk of an irreversible injury of the radial artery as the potential donor vessel for dialysis shunt in the future is judged as too high [11, 12].
The transplanted kidney often displays reduced clearing function and higher vulnerability to contrast dye due to various reasons: sympathetic denervation during explantation impairs the kidney vessels’ autoregulation, calcineurin inhibitors are known to have adverse effects on donor kidneys [13–15], and the donor organ might have been affected by pre- and peri-transplant organ handling as well as subclinical presence of chronic renal impairment before transplantation [16].
Aim
The aim of the present study is to describe the incidence and risk factors of AKI, periprocedural bleeding and their prognostic impact on 1-year mortality in KTR undergoing CI in a retrospective, single-center analysis. The primary endpoint was acute kidney injury defined by the AKIN classification (as described in the methods section). Secondary endpoints were periprocedural bleeding events as defined by the BARC classification and 1-year survival.
Material and methods
Patients’ data collection and follow-up
For this single-center retrospective analysis, all patients with a history of kidney transplantation who underwent coronary angiography with or without PCI were selected according to the ICD-10 code for kidney transplantation and OPS codes for invasive coronary diagnostics and PCI at Goethe University Hospital Frankfurt, Germany between 2005 and 2015. For every patient, demographic, clinical, laboratory and procedural data were collected from the electronic hospital information system (Orbis, Agfa). Glomerular filtration rate was estimated according the MDRD formula with the creatinine value at baseline. Follow-up data were collected from the electronic patient files, affiliated outpatient clinics and requests from the patients’ general practitioners. Patients were followed up from the day of index procedure until 12 months after or all-cause death.
This study complies with the local standards of the institutional ethics committee (Approval file No. 312/16) and the Helsinki Declaration from 1975.
Classification of kidney injury
Serum creatinine value and glomerular filtration rate estimated by the 4-variable MDRD formula (eGFR) were used as baseline kidney function. Acute kidney injury was defined according to the AKIN classification (Acute Kidney Injury Network) [17]. AKIN 1 is defined as an increase of serum creatinine of > 0.3 mg/dl or a 1.5 to 2-fold increase from baseline. AKIN 2 is defined as an increase of 2 to 3-fold and AKIN 3 as an increase of more than 3-fold or creatinine > 4 mg/dl with an acute increase of > 0.5 mg/dl.
Classification of periprocedural bleeding events
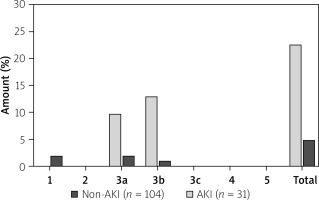
Complications due to bleeding were defined by the BARC criteria (Bleeding Academic Research Consortium) [18]. Type 0 means no signs of bleeding. Type 1 bleeding does not require any treatment. Overt signs of hemorrhage without the need for action are defined as type 2. Type 3 bleeding is divided as follows: type 3a – overt bleeding with hemoglobin drop 3.5 g/dl + transfusion; type 3b hemoglobin drop > 5 g/dl/cardiac tamponade/need for surgical intervention/IV vasoactive agents; type 3c intracranial bleeding. Any coronary artery bypass grafting (CABG)-related bleeding is classified as type 4, and fatal bleeding is classified as type 5.
Statistical analysis
Statistical analyses were performed using Prism 7 (GraphPad Software Inc.; San Diego, USA) and SPSS 23.0 (IBM). Continuous variables are shown as median and interquartile range (IQR), while categorical variables are reported as frequencies and percentages. Differences between patient cohorts were determined using Fisher’s exact test for categorical variables; for quantitative variables the Mann-Whitney-U test was used. The Kaplan-Meier estimator and log-rank test were used for survival analysis. Predictors of survival and endpoints were determined with Cox regression analysis. Binary univariate and multivariate logistic regression analyses were performed to study the determinants of independent predictors of AKI. Odds ratio (OR) and corresponding 95% confidence intervals were calculated simultaneously. A two-sided p-value of < 0.05 was considered significant.
Results
Patients
As depicted in Table I, patients were predominantly male (79% male gender), with a median age of 65 years, a high burden of preexisting chronic transplant kidney disease (median serum creatinine 1.8 mg/dl) and a high prevalence of diabetes mellitus (36%) and coronary multivessel disease (72%). Left ventricular ejection fraction – LVEF) was preserved in the majority of patients.
Table I
Baseline characteristics, indications and complications after coronary angiography of AKI vs. non-AKI group
During the study period, 135 invasive coronary procedures were performed in our institution (see Table I for details). Of those, 53 (39%) procedures were diagnostic. In 82 (61%) procedures, PCI of one or more coronary arteries was performed. The indication for the invasive approach was predominantly elective (69%) while 31% were urgent indications in the setting of acute coronary syndromes. Transfemoral access was performed in 93% of the procedures, while 7% of the procedures were performed via transradial access.
Acute kidney injury
AKI occurred after 31 of the 135 procedures, representing an incidence of 23%. These two groups are named “AKI” and “non-AKI” below. AKIN 1 was observed in 77% of the AKI patients, AKIN 3 with the need for transient RRT in 23% of the AKI group (Table II). No patient developed AKIN 3 with permanent RRT during the study period. There was no case of AKIN 2. No significant differences in the incidence of AKI were found after diagnostic angiography (25%) or PCI (22%; p = 0.83). Furthermore, the incidence of AKI after transfemoral access (24%) and the incidence of AKI after transradial access (11%) did not show a significant difference (p = 0.68). Patients from the AKI group were significantly older (Table I) and displayed a higher proportion of acute coronary syndromes as the indication for invasive approach (p = NS, Table I). Further, AKI patients suffered more often from diabetes mellitus and coronary multivessel disease, without reaching statistical significance for these parameters. The total amount of contrast dye was not significantly higher in the AKI group (155 ml (IQR: 70–225) vs. 127 ml (IQR: 71–200), p = 0.43).
Table II
Incidence and extent of acute kidney injury, need for acute hemodialysis and graft loss
| Parameter | All (n = 135) | AKI (n = 31) |
|---|---|---|
| AKIN 1–3, n (%): | 31 (23) | 31 (100) |
| AKIN 1 | 24 (18) | 24 (77) |
| AKIN 2 | 0 (0) | 0 (0) |
| AKIN 3 | 7 (5) | 7 (23) |
| Need for acute hemodialysis | 7 (5) | 7 (23) |
| Graft loss | 0 (0) | 0 (0) |
There was no difference in percentages of male patients in the AKI and non-AKI group (71% vs. 81%, p = 0.32). Patients of the AKI group tended to have higher creatinine levels at baseline (2.0 mg/dl vs. 1.8 mg/dl, p = 0.12) and significantly lower eGFR (32 ml/min vs. 36 ml/min; p ≤ 0.05). Most of the patients suffered from chronic glomerulonephritis as chronic kidney disease (62% in the AKI group and 44% in the non-AKI group; p = 0.11). There were no significant differences in the immunosuppression scheme between the groups.
In multivariate analysis, periprocedural bleeding persisted as a single independent predictor of AKI with an OR of 6.43 (95% CI: 1.78–23.20, p = 0.01; Table III).
Table III
Predictors and potential risk factors for development of AKI – univariate and multivariate analysis for endpoint AKI
Periprocedural bleeding complications
Bleeding events were registered throughout hospitalization; bleeding events after hospital discharge have not been recorded. There were 10 patients with access site bleeding, 2 patients with pericardial tamponade. Periprocedural bleeding as defined by the BARC criteria occurred in 23% of the AKI group and only 5% of the non-AKI group (p < 0.01). Bleedings were mainly categorized as type 3a or 3b (see Classification of periprocedural bleeding events and Figure 1). Access-site bleeding occurred only in patients with femoral access. An extended univariate analysis was performed for the secondary endpoint without detecting predictors of periprocedural bleeding. Surgical treatment after access site bleeding was necessary for 2 patients of the AKI group and 1 patient of the non-AKI group.
Mortality and follow-up
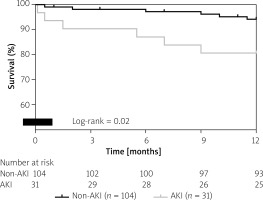
In-hospital mortality was 3% in the AKI group; no patient of the non-AKI group died during hospitalization (p = 0.2). One-year survival was 81% in the AKI group and 94% in the non-AKI group (p = 0.02; Figure 2).
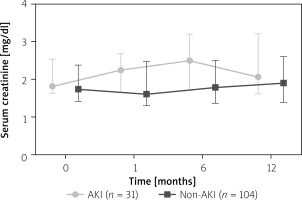
Kidney function was analyzed for 1, 6 and 12 months after CI. Kidney function was stable during follow-up. Figure 3 shows serum creatinine values and IQR 30 days, 6 and 12 months after CI. There was no significant difference in serum creatinine after 12 months (p = 0.28).
In multivariate analysis, only the occurrence of periprocedural bleeding complications persisted as a risk factor of acute kidney injury in these patients (Table III). The amount of contrast dye used during the CI was not an independent predictor of acute kidney injury in this study.
Discussion
The accurate choice of coronary revascularization technique in KTR remains a complex task [19–22]. In patients undergoing invasive coronary angiography and PCI, acute kidney injury following coronary angiography and PCI is a topic of high interest. This study of kidney transplant recipients receiving CI analyzed the incidence and outcome of acute kidney injury and interaction with periprocedural bleeding.
In our study, the incidence of AKI after CI was 23.0%; no patient needed permanent RRT. Our results are comparable to several studies regarding the incidence of AKI in KTR: Lees et al. found an incidence of 18.9% [23], Neves et al. reported an incidence of AKI of 17.5% in 7800 patients after coronary angiography [24]. Fähling et al. found an incidence of 30% for contrast-induced nephropathy [25]. Victor et al. indicated the amount of contrast dye as an important risk factor for AKI and included it in their risk scoring model together with GFR, diabetes, anemia, hypotension and peripheral artery disease [26].
In order to avoid AKI in patients with renal disease undergoing CI, advanced PCI technologies to reduce contrast dye are evaluated such as ultra-low contrast angiography and zero-contrast PCI [27]. Ali et al. performed CI without contrast in 31 patients with advanced CKD using IVUS, without changes in creatinine levels or eGFR in the follow-up period [28]. The study of Sacha et al. showed that zero-contrast PCI was a safe and effective method in 20 patients with severe CKD undergoing 29 procedures and also reduced the estimated risk for AKI from 26% to a prevalence of 10% without specific complications [29].
However, Caspi et al. found similar rates of AKI in patients with STEMI with and without contrast exposure [30], indicating the presence of several factors for development of AKI in cardiovascular patients: age over 70 years, GFR, diabetes mellitus and heart failure [30].
In our study, univariate analysis indicated that the amount of contrast dye was not a significant risk factor for development of AKI; both the AKI and non-AKI group received substantial amounts of dye during CI. However, we found periprocedural bleeding complications to be associated significantly with acute kidney injury. Our findings correspond to those of Neves et al., who found major bleeding as a risk factor for AKI with higher in-hospital mortality and lower 1-year survival [24]. The vast majority of PCIs in our study were performed using a transfemoral access. Several studies have described the association of arterial access and AKI in terms of less AKI in patients with transradial access (TRA). TRA is associated with significantly reduced rates of major bleeding [31]. Whether the pathophysiological mechanism for increased AKI rates in patients with major bleeding is caused by hemodynamic changes in terms of hypovolemic shock under bleeding or a loss of hemoglobin remains unclear [32]. In the study of Pancholy et al., TRA was associated with a 43% relative reduction of post-PCI AKI compared to transfemoral access [33].
Pathogenesis of AKI in patients suffering from bleeding after CI may involve several pathways: first, acute hypovolemic shock due to bleeding will induce shock-induced nephropathy. Second, the need for transfusion after bleeding also has a negative impact on kidney function. The paper by Ohno et al. describes an increased risk of AKI also in patients with a drop in hemoglobin levels without hemodynamic change [34]. Third, the possibility of catheter-induced microembolization into the transplant donor vessel during aortal catheter passage has to be taken into account in patients with transfemoral access [35].
This study bears several important limitations: first, the lack of information about intraprocedural hemodynamics in patients with and without AKI. Moreover, serum creatinine could not be analyzed for all patients during the follow-up; only one patient underwent CI in cardiogenic shock. Further, this study is limited by its design as a retrospective observational study. Hence, we could not describe independent risk factors of periprocedural bleeding due to the non-randomized study fashion with predominant transfemoral arterial access and a small absolute number of bleeding events. Therefore, we only can speculate that improved access safety in terms of more frequent use of transradial access in a comparable KTR population will be associated with fewer bleeding events, less AKI and improved survival.
Conclusions
Cardiovascular disease is the most common cause of morbidity and mortality in patients with impaired kidney function. This study analyzed the incidence, risk factors and outcome of acute kidney injury and bleeding complications in kidney transplant recipients receiving coronary interventions in diagnostic or therapeutic intention. Kidney transplant recipients of our cohort with acute kidney injury after coronary intervention showed lower 1-year-survival. Graft function was stable 12 months after the procedure. In this subset of patients at high risk for AKI receiving substantial amounts of contrast dye for CI, periprocedural bleeding could be identified as an important independent risk factor for AKI. Optimized vascular access and the avoidance of periprocedural bleeding complications seem to be essential in this high-risk population.