Introduction
The current severe acute respiratory syndrome coronavirus-2 (SARS-CoV-2)/COVID-19 pandemic, caused by the novel coronavirus, became a tough challenge for health care systems worldwide. The virus is characterised by high infectiousness, with 8,532,150 confirmed cases (registered till 18 June 2020) [1]. Surprisingly, the most severe disease course, with mortality of 5.3% (454,014 lethal cases till 18 June 2020), almost exclusively concerns adults [1]. Recent studies demonstrated that children usually are asymptomatic or develop only mild symptoms. In consequence, the case fatality rate of SARS-CoV-2/COVID-19 in children below 10 is nearly 0% [2, 3].
A reason for the benign presentation of SARS-CoV-2/COVID-19 in children is puzzling. It was suggested that mild symptoms may be attributed to different mechanisms of the immune response or higher levels of neutralizing antibodies, due to broader exposures to respiratory infections in winter. Also, lower maturity and binding ability of angiotensin converting enzyme II (ACE2), or dipeptidyl peptidase IV (DPP IV), the cell receptors for coronavirus, have been considered [2, 4].
Obviously, the understanding of underlying mechanisms for such a difference may be crucial in taming the SARS-CoV-2/COVID-19 pandemic. In that context, we would like to bring to the attention of the scientific community involved in research on COVID-19 the exceptional feature of children’s exhaled breath condensate (EBC). This condensed vapour solution, containing a mixture of non-volatile compounds present in exhaled air, is currently considered as a promising non-invasive tool in respiratory research. In our previous studies devoted to asthma-related angiogenesis [5], we found that children’s EBC exerted a dose-dependent inhibitory or cytotoxic effect on endothelial cell cultures [6].
However, we could not find any data on a similar effect caused by EBC samples collected from adults.
Aim
We aimed to evaluate and compare the biological effect of EBC in adults and children in the aforementioned experimental in vitro model. Furthermore, in order to explain the background of the observed effect and to identify an agent responsible for these unexpected biological properties of children’s EBC, we attempted to analyse the composition of selected EBC samples.
Material and methods
The study involved EBC samples excessively collected for our previous projects [6–8]. The samples were aliquoted and stored at –70°C, to avoid repetitive freezing-thawing cycles. We have used already stored samples of 28 children and 31 adults. Children samples were collected from patients with moderate or mild stable asthma (n = 16) and from their age-matched healthy controls (n = 12). Similarly, adults’ EBC samples included these collected from patients with moderate stable asthma (n = 15) and those from age-matched healthy controls (n = 16). The clinical characteristics of patients were described elsewhere [6–8]. The study was planned and performed according to the Declaration of Helsinki and European Medicines Agency Guidelines for Good Clinical Practice. The concept of the study was reviewed and approved by the local Ethics Committee of the Medical University of Warsaw.
All EBC samples were tested using in vitro cell cultures, as described in detail previously [6]. Briefly, C-166 endothelial cells (purchased from the American Type Culture Collection, Manassas, VA) were seeded in 100 µl of culture medium onto 96-well culture plates, at a density of 104 cells/well, and remained for 4 h, to allow their adhesion to the plastic. Then, 20 µl of EBC (tested sample), distilled water (baseline control) or Triton X-100 (negative control) were added to the culture in triplicates and incubated for 24 h in standard conditions, at 37°C, in humidified air with 5% CO2. Four hours before the test end 20 µl of 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT, from SIGMA-Aldrich, St. Louis, MO) in PBS were added to the cultures. Afterwards, cells were lysed in dimethyl sulfoxide with HCl and their absorbance was measured at λ = 540 nm using microplate reader (BIO-RAD, Hercules, CA). MTT reduction by target cells in the presence of tested EBC samples was expressed as the percent of counts from baseline control (cells with water), which was considered as 100%. Counts from negative control (with Triton X-100) were considered as 0% [6].
In order to verify whether the observed effect of EBC from the children group was independent from presumable microbial contamination, we tested eight randomly selected EBC samples (5 samples from the healthy group and 3 samples from asthma subjects) for the presence of endotoxin. The assessment was performed using ToxinSensorTM Chromogenic Limulus Amebocyte Lysate (LAL) Endotoxin Assay Kit, according to the detailed manufacturer’s protocol (GenScript, Piscataway, NJ).
EBC samples collected from two healthy children, which displayed the strongest inhibitory effect on MTT reduction, were used for further experiments. Briefly, samples were fractionated by size exclusion chromatography (SEC) using 2695 Alliance system (Waters, Milford, MA) equipped with 2489 UV/VIS detector. Data acquisition and processing were conducted using Empower software. The tested samples (0.1 ml each run) were injected on the BioSep™ 5 µm SEC-s2000 (145 Å, 300 × 4.6 mm) liquid chromatography column (from Phenomenex Inc.), conditioned at 25°C. The separation was carried out using the isocratic elution mode. As the eluent 0.2 M di-sodium hydrogen phosphate, pH 6.8 (obtained with the addition of 85% orthophosphoric acid) was used. The flow of the mobile phase was set at 0.25 ml/min, run time of the single analysis was 60 min. Separated components of the EBC mixture were collected with Fraction Collector III (Waters) set to the TIME mode using collection time intervals to acquire desired portions. Based on data from preliminary experiments, five fractions, corresponding to absorbance peaks, observed between 16 and 28 min of sample flow (16.00’ to 18.50’, 18.52’ to 20.00’, 20.02’ to 22.00’, 22.02’ to 24.50’, 24.52’ to 28.00’), were collected. Immediately after collection each fraction was deep-frozen and kept at –80°C until further use.
Afterwards, the collected fractions were thawed, passed through 0.22 µm Millex-GV polyvinylidene difluoride (PVDF) microfilter (Millipore-Merck, Darmstadt, Germany) and subjected to MTT assay with C-166 cells, using the same experimental conditions, as aforementioned for whole EBC samples. Fraction, which revealed the strongest effect in MTT assay, was then used for analysis in mass spectrometry.
Mass spectra were acquired in a positive-ion reflector mode using a tandem matrix-assisted laser desorption/ionization with time-of-flight mass spectrometry (MALDI-TOF MS) device – 4800 Plus MALDI-TOF/TOF Analyzer (Applied Biosystems/Thermo Fisher Scientific, CA), equipped with a pulsed solid-state YAG laser. A matrix solution consisted of a saturated solution of α-cyano-4-hydroxy-cinnamic acid in 50 : 50 water/acetonitrile with 0.1% trifluoroacetic acid. External calibration was achieved with a 4700 proteomics analyzer calibration mixture provided by Applied Biosystems. Samples were spotted onto a 384 Opti-TOF MALDI plate and analysed. Data Explorer Software, Version 4.9 was applied to process acquired spectra.
Results
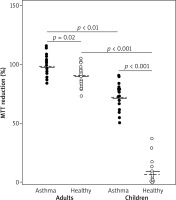
The influence of EBC on MTT reduction by endothelial cells was significantly different in children and adults. Weak or no effect of adults’ EBC was observed in both asthmatic and healthy individuals (mean percent of MTT reduction was 97.8 ±9.5 vs. 89.1 ±9.6, respectively). In contrast, notably decreased levels of MTT reduction, with mean percent values of 72.4 ±11.1 in asthma and 12.6 ±10.9 in healthy controls were demonstrated in cultures with children’s EBC. The values measured in cell cultures treated with EBC from both children groups were significantly lower as compared to values in respective adult groups (Figure 1).
Figure 1
EBC influence on in vitro MTT reduction in C-166 cell culture. Each dot represents percent value of MTT reduction ability, calculated from mean of three counts of individual EBC sample, compared to baseline (100%) and negative control (0%). Horizontal lines within each group represent mean (solid line) or median (dashed line) for the group. Horizontal whiskers with p-values (by Mann-Whitney U test) depict comparison between groups
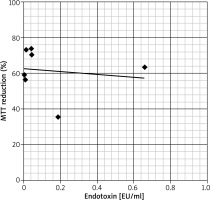
The assessment of endotoxin levels in randomly selected EBC samples from the children group has revealed that in seven of eight tested samples, the endotoxin concentration was below 0.5 U/ml, that is the upper limit allowed for water for injection, according to recommendations of the Food and Drug Administration [9]. One EBC sample from the asthma group revealed the endotoxin level slightly above the aforementioned limit (i.e. 0.65 U/ml), nevertheless, any significant correlation between endotoxin amounts and the cytotoxic effect of tested EBC samples was not observed (Figure 2).
Figure 2
Correlation between the concentration of endotoxin in Limulus Amebocyte Lysate assay (expressed in endotoxin units per millilitre – EU/ml) and biological activity in MTT reduction assay (expressed as % of MTT reduction ability in the control culture) of selected EBC samples. Each dot represents one EBC sample. The trend line on the graph shows that both variables seem to be independent
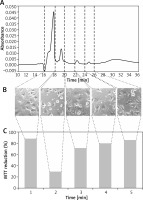
Five preselected fractions (0.5 ml each), collected from EBC of healthy children between 16 and 28 min of sample flow, were tested in MTT reduction assay. The strongest cytotoxic effect, expressed as a significant impairment of mitochondrial MTT-reducing activity (to the level of 29.4%, as compared to test control), was observed for fraction no. 2. The moderate inhibitory effect, with the mitochondrial activity decreased to 71.4% of control, was observed for fraction no. 3. The incubation of C-166 cells with the remaining three fractions revealed their weak influence on MTT reduction, with the levels of 80–88%, compared to test control (Figure 3).
Figure 3
The absorbance profile of the EBC sample in size exclusion chromatography (A). The collected fractions were marked with dashed vertical lines, indicating the time of collection (in minutes). The phase-contrast images of C-166 cell cultures incubated with respective EBC fractions just before addition of MTT to the culture (B). The result of MTT reduction assay (shown as percent of the test control) for respective EBC fraction (C). The fraction numbers were shown at the bottom
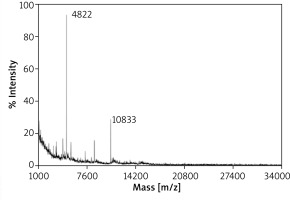
The mass spectrometry analysis of fraction no. 2 has revealed the presence of several signals of 4.3, 8.6 and 10.8 kDa and the prominent peak of molecular mass 4.8 kDa (Figure 4). Noteworthy, residual amounts of 4.8 kDa signal were detected also in mass spectra of fraction no. 3 (data not shown).
Discussion
Our study has shown that EBC influenced the ability of endothelial cells to reduce MTT in vitro. The intensity of the aforementioned effect varied between groups and among individuals within each of the tested groups, but EBC from healthy children exerted a significantly stronger inhibitory or even cytotoxic effect, compared to all other groups. Noticeably, recent experiments with EBC from healthy children have shown that this effect was similar also in other cell types, including fibroblasts and keratinocytes (data not shown).
The agent, or agents, responsible for this effect remain unknown. It is plausible that it could be the result of microbial contamination of tested EBC samples, e.g. by some endotoxins from microbiota of oral cavity. However, using ultrasensitive LAL assay we have found that only one of eight tested samples revealed the endotoxin concentration slightly exceeding the Food and Drug Administration (FDA) limit approved for parenteral drug products [9]. Moreover, the results of LAL assay did not show any correlation between endotoxin concentrations in tested EBC and their effect on MTT reduction in C-166 cultures. Thus, we have excluded the role of bacterial endotoxins in that phenomenon.
In order to determine the putative cytotoxic factor from breath condensate, we have used gel chromatography to fractionate selected EBC samples, followed by MTT test to find the fraction displaying the most cytotoxic activity. Then, the most effective fraction (no. 2) was used for subsequent analysis using MALDI-TOF MS, which has revealed the presence of 4.8 kDa compound. The latter was found in trace amounts also in fraction no. 3, displaying moderate cytotoxic activity. Thus, we hypothesize that aforementioned 4.8 kDa molecule may be responsible for the biological effect of EBC, observed in cell cultures. At present, the identification of this agent and the determination of its detailed characteristics are the subject of ongoing research.
The physiological role of the observed effect and the putative factor responsible for that remains elusive. One can hypothesize that it could be a component of body homeostasis, which may control cell function in the respiratory system. Apart from presumable anticancer defence, it could possibly be involved in antiviral resistance. Impaired mucus-mediated protection may result in increased susceptibility to viral infection. Since it may also expose epithelia to putative cytotoxic EBC component, the latter may eliminate infected host cells long before viral replication will accomplish. Although at the current stage it is a speculation only, our observation should be verified in the context of age-related epidemiology of SARS-CoV-2/COVID-19. In that aspect recent intriguing reports concerning co-incidence of COVID-19 and Kawasaki-like syndrome in young children may appear especially encouraging [10]. One can speculate that at least in some SARS-CoV2-infected children the extensive vascular damage could be mediated by the mechanism similar to that described in our report.
The main limitation of this study is a relatively small number of samples in each group. Therefore, our results should be considered as preliminary and will require verification in larger groups, preferably in a multicentre study. In further studies it will also be necessary to assess the mass spectra of fractionated EBC, collected from asthmatic children, and both adults groups. However, to enable further research on the putative factor, responsible for the observed cytotoxic effect of children’s EBC, especially on its postulated role in the clinical course of SARS-CoV-2/COVID-19 in children, a full identification of the aforementioned agent is necessary. On the “of supposing” hand, it is intriguing why the proposed mechanism of supposing it will be confirmed in subsequent research, seems to disappear or attenuate in the elderly? The further studies are in progress.