Introduction
Invasive lobular carcinoma (ILC) accounts for 10–15% of all breast cancers and is associated with higher stage at diagnosis. ILC tends to have more hormone receptor-positive and HER2 receptor-negative biological subtypes and is less responsive to chemotherapy [1].
Preoperative chemotherapy (PCT) is traditionally used in the treatment of patients with locally advanced breast cancer (LABC) in an attempt to downstage the disease and achieve breast-conserving surgery (BCS) [2]. Total pathologic complete response in breast and nodes (tpCR; ypT0/is ypN0) after PCT is associated with better outcome [3]. However, the pathologic complete response (pCR) rate according to histology is not usually considered in trials.
Material and methods
Patients and eligibility criteria
We conducted an exploratory analysis of tpCR rates in the subgroup of patients with ILC of the breast. Adult patients (> 18 years of age) with histologically confirmed locally advanced or inflammatory ILC of breast, from three phase II trials (AT, ATX, and TXH) [4–6], were eligible in this study. Patients were treated with one of the following regimens of PCT, as previously reported: TXH (docetaxel, capecitabine, and trastuzumab), ATX (doxorubicin, docetaxel, and capecitabine), or AT (doxorubicin and docetaxel). Adjuvant chemotherapy (CT), radiotherapy (RT), and hormone-therapy (HT) were allowed after surgery. Expression of markers and clinical phenotypes (CPh) were determined by immunohistochemistry before PCT [7]. Patients were required to give written, informed consent before inclusion in the trials. The study protocols were approved by the institutional review board at each study centre, and the study was conducted in accordance with the principles of the Declaration of Helsinki.
Study endpoints
The primary endpoint of the study was the tpCR rate, defined as no evidence of invasive cancer cells in breast tissue and lymph nodes removed during surgery (ypT0/is ypN0), after PCT in the subgroup of patients with ILC. Secondary endpoints were clinical response (CR) rate, BCS rate, pathologic tumour size, event-free survival (EFS), and overall survival (OS). EFS was defined as the time from the beginning of PCT until the date of the first occurrence of one of the following events: recurrence of ipsilateral breast tumour, recurrence of ipsilateral locoregional breast cancer, distant disease recurrence, or death from any cause. OS was defined as the time from the diagnosis of ILC of the breast until death from any cause.
Statistical analysis
Standard summary statistics for continuous variables were: median, range, standard deviation, and/or 95% confidence interval (95% CI). Standard summary statistics for discrete variables were: count and proportion. Response rates were summarised by proportions. Survival estimations (EFS and OS) were calculated with Kaplan-Meier methods. The Stata statistical program, version 14 (StataCorp. 2007. Stata Statistical Software: Release 14. College Station, TX: StataCorp LP) was used for all statistical analyses.
Results
Patient characteristics and pre- and post-surgical treatment
Of 185 patients with LABC treated with PCT in the three trials (AT, ATX, and TXH), 16 patients had ILC (8.65%): 10 patients in the AT trial (62.5%), five patients in the ATX trial (31.25%), and one patient in the TXH trial (6.25%). Patients with ILC were treated with a median of four cycles (range: 3–6) of PCT.
In this subgroup of patients the median age was 50 years (range: 38–66), 56.25% (n = 9) were premenopausal, median tumour size was 5 cm (range: 3–6), and 68.75% (n = 11) had clinical node involvement. Six patients (37.5%) had clinical stage II breast cancer and 10 (62.5%) had clinical stage III. Hormone receptor-positive disease was present in 93.75% (n = 15) of the patients, only one patient had an HER2-positive tumour (6.25%), and median Ki-67 was 25% (range: 5–70). CPh was Luminal A-like in 37.5% (n = 6), Luminal B-like in 50% (n = 8), HER2-positive in 6.25% (n = 1), and triple negative in 6.25% (n = 1) of tumours (Table 1).
Table 1
Baseline clinical and pathological characteristics of the study population (n = 16)
Ten patients (62.5%) were treated with adjuvant CT, four patients (25%) with CMF (cyclophosphamide, methotrexate, and fluorouracil), and six patients (37.5%) with anthracycline and taxane-based CT (AT and ATX), with a median of two cycles (range: 1–2). One patient (6.25%) with HER2-positive tumour was treated with adjuvant trastuzumab. Eleven patients (68.75%) were treated with adjuvant RT, with a median dose of 50 Gy (IC 95%: 41–60 Gy). Fifteen patients (93.75%) with hormone receptor-positive tumours were treated with HT, eight patients (50%) with tamoxifen, and seven patients (43.75%) with an aromatase inhibitor (letrozole or anastrozole).
Primary and secondary endpoints
Of the 16 evaluable patients with ILC, only one patient (6.25%) had a tpCR; this patient had a triple-negative tumour. Another patient achieved a pCR only in the breast (pCR in the breast of 12.5%; Table 2).
Table 2
Clinical and pathologic responses to preoperative chemotherapy (n = 16)
The overall CR rate was 68.75% (11/16), with 12.5% (2/16) of clinical complete response and 56.25% (9/16) of clinical partial response. One patient (6.25%) had a progression during PCT. The BCS rate was only 12.5% (2/16), and the median pathologic tumour size after PCT, in the 15 patients without a tpCR, was 2.4 cm (IC 95%: 0–3.3 cm).
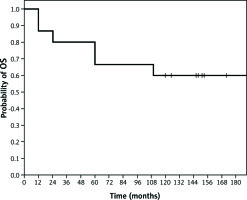
With a median follow-up of 146 months (95% CI: 113–179 months), median EFS was 120 months (95% CI: 68–139 months). The EFS probability at three years, five years, and 10 years was 60%, 53%, and 47%, respectively (Fig. 1). Median OS in this subgroup of patients was not reached; and three-year, five-year, and 10-year OS probability was 80%, 67%, and 60%, respectively (Fig. 2).
Fig. 1
Kaplan-Meier estimates of event-free survival (EFS). In the subgroup of patients with locally advanced invasive lobular breast carcinoma treated with preoperative chemotherapy (n = 16), with a median follow-up of 146 months (95% CI: 113–179 months), median EFS was 120 months (95% CI: 68–139 months), and five-year and 10-year EFS probability was 53% and 47%, respectively
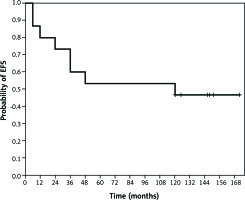
Fig. 2
Kaplan-Meier Estimates of overall survival (OS). In the subgroup of patients with locally advanced invasive lobular breast carcinoma treated with preoperative chemotherapy (n = 16), with a median follow-up of 146 months (95% CI: 113–179 months), median OS was not reached, and five-year and 10-year OS probability was 67% and 60%, respectively
In this population, 50% (8/16) of patients had an EFS event: six patients (37.5%) had visceral metastasis as first recurrence, one patient (6.25%) had only ipsilateral breast recurrence, and one patient (6.25%) had ipsilateral locoregional recurrence. Seven patients (43.75%) died due to tumour progression during the study.
Discussion
The goal of PCT in breast cancer is to downsize the tumour and nodal burden, allowing for options of less aggressive surgical management converting the need for mastectomy or axillary lymph node dissection to lumpectomy or sentinel lymph node biopsy. As the use of PCT increases, it is important to evaluate which patient subgroups will derive most benefit from PCT. One must weigh the benefits and risks of giving systemic CT. While the goal of less aggressive surgery is a great option, the risk of giving CT to a tumour type not likely to respond to CT risks the side effects of overtreatment.
In our study, the tpCR rate in patients with locally advanced ILC of breast treated with PCT was low (tpCR of 6.25%), and these patients had poor outcome, with 10-year EFS and OS probability of only 47% and 60%, respectively, despite the fact that they were treated with adjuvant CT (62.5%), RT (68.75%), and/or HT (93.75%). In addition, only 12.5% of patients underwent a BCS. Similar results in terms of BCS rate after PCT have been published in the literature [8].
Thus, PCT might not be a good treatment option in patients with locally advanced ILC of the breast because the vast majority of these patients have Luminal A and B tumours. In fact, in our study hormone receptor-positive disease was present in 93.75% of the patients, and 87.5% of patients had Luminal like tumours (37.5% Luminal A-like and 50% Luminal B-like). Moreover, the only patient with ILC who achieved a tpCR after PCT had a triple negative tumour. The results of our study are similar to previous studies, which have shown low tpCR rates in patients with ILC of the breast [9–12].
An important limitation of our study is the small number of patients with locally advanced ILC of the breast. Only 16 patients were included in this analysis, but the results of our study are similar to recently published articles, with low tpCR rates in patients with ILC of the breast after neoadjuvant chemotherapy [11, 12]. In the study conducted by Petruolo et al. [12] patients with ILC of the breast were unlikely to downstage in the breast or axilla after chemotherapy compared to patients with invasive ductal breast carcinoma. Patients with ILC initially ineligible for breast-conserving surgery (n = 56) were less likely to downstage than those with invasive ductal carcinoma (n = 183, 16% vs. 48%, p≤ 0.0001), with a similar trend in the axilla (p = 0.086). In another study, patients with ILC were also significantly less likely to have a tpCR after PCT than patients with invasive ductal breast carcinoma (11% vs. 25%, p = 0.01) [11].
Another issue to consider in our study is that the vast majority of eligible patients have high Ki-67 rates (median of 25%), This fact might be the consequence of the patients’ selection for neoadjuvant chemotherapy. However, despite the high Ki-67 rate there was a low tpCR rate after PCT in this subgroup of patients.








