Summary
Long-term data on Lotus® (Boston Scientific, USA) transcatheter aortic valve prostheses are lacking. Our study suggests that higher cardiovascular mortality rates during mid-term follow-up were associated with Lotus compared with Evolut valves. Higher gradients on the Lotus valves at the end of follow-up suggest the possibility of accelerated prosthesis degeneration.
Introduction
The Lotus® valve (Boston Scientific, USA), designed for transcatheter aortic valve implantation (TAVI), was promising because of the repositionable and fully retrievable prosthesis with excellent procedural [1, 2] and 1-year follow-up results [2, 3]. The Lotus valve also included an adaptive seal to effectively minimize paravalvular leak (PVL) [4] and thus enabled TAVI also in complex anatomies such as stenosis of bicuspid aortic valve [5–7]. However, long-term data on Lotus are lacking compared with results for other currently used TAVI valves [8–10]. With the extension of TAVI indications to younger, lower-risk patients [11], data on long-term transcatheter valve durability are essential [12]. Although the Lotus valve was recalled in 2020 [13, 14] and is not available for clinical use, thousands of valves were implanted over the last decade.
Aim
We evaluated the mid-term outcomes of patients with Lotus and compared them with those of patients with Evolut R® valves (Medtronic, USA).
Material and methods
Design
We conducted a single-centre, retrospective, observational study comparing outcomes of patients with severe aortic stenosis (AS), who underwent TAVI with Lotus or Evolut R valves. The study was approved by a multicentre Ethics Committee and was conducted in accordance with the principles of the Declaration of Helsinki.
Patients and procedures
A total of 197 consecutive patients with severe AS, who underwent TAVI between August 2015 and January 2020, were enrolled in this study. Patients were either admitted haemodynamically stable for planned diagnostic evaluation or admitted acutely with decompensated heart failure. Diagnosis of severe AS was made according to echocardiographic criteria. The indication for TAVI was established as a consensus of members of the heart team. All TAVI procedures were performed by highly experienced interventional cardiologists in a single tertiary centre. Clinical, demographic, and echocardiographic data were recorded at baseline and during follow-up. Patients had a clinical and echocardiographic examination before discharge from the hospital after the index procedure, 30 days after TAVI, and 1 year after TAVI. Information on specific events was obtained from follow-up visits and from the National Institutes of Health Information and Statistics. Because of COVID, it was difficult to distinguish infection from other causes of death. Therefore, we ended the clinical follow-up in March 2020 to prevent mortality bias resulting from COVID infection, but in 2020–2021 we collected all available echocardiography studies from living patients with the Lotus valve and compared them with patients with the Evolut valve and similar length of follow-up.
The Lotus® (Boston Scientific, USA) valve is a bioprosthetic aortic valve comprising a braided nitinol wire frame with 3 bovine pericardial leaflets and a polymer membrane surrounding the lower half of the valve to reduce PVL. The valve is pre-mounted on a delivery catheter and deployed via controlled mechanical expansion enabling repositioning or retrieval of the valve at any point before release [3]. The Medtronic CoreValve Evolut R® System (Medtronic, U.S.A.) comprises the Evolut R valve and the EnVeo R Delivery Catheter System (DCS) with the InLine sheath. The trileaflet valve and sealing skirt are made from porcine pericardial tissue, sutured in a supra-annular position on a compressible and self-expandable nitinol frame. The Enveo R DCS enables the valve to be fully repositionable and recapturable before full release by turning the delivery handle [15]. All TAVI procedures were performed with a Lotus® (Boston Scientific) Introducer set.
Outcomes
Primary outcomes were mid-term (I) all-cause mortality and (II) cardiovascular mortality. Secondary outcomes included clinical outcomes as a periprocedural myocardial infarction, an overt central nervous system injury (CNS) at 30 days, bleeding complications type 2, 3, or 4 at 30 days, major vascular complications at 30 days, acute kidney failure, stage 2 or 3, and valve malposition. Outcomes were defined according to standardized endpoint definitions for transcatheter aortic valve implantation clinical trials in a consensus report from the Valve Academic Research Consortium (VARC3) [16].
Statistical analysis
Data are presented as means ± standard deviations (SD) for continuous variables and median ± interquartile range for categorical variables. Continuous variables were compared by Welch two-sample t-tests for the normally distributed variables and by two-sample Wilcoxon tests for the variables with some outliers. Categorical variables were compared using the Pearson χ2 test of independence or by Fisher exact tests (in the case of expected low counts). P-value < 0.05 was considered statistically significant.
The log rank test was used to compare survival times in 2 groups. Kaplan-Meier estimates of the survival curves are presented. The Cox proportional hazards model was used to identify predictors of all-cause mortality and cardiovascular mortality and to adjust for potential baseline differences between the Lotus and Evolut groups. All analyses were performed using statistical program R and GrafPad Prism Version 6.05.
Results
Baseline characteristics
A total of 197 consecutive single-centre patients with symptomatic aortic stenosis received either Lotus or Evolut valves. Valve selection was at the discretion of clinicians. Seven (3.6%) patients died after the TAVI procedure during initial hospitalization. Therefore, our sample under investigation comprised 190 patients (71 Lotus and 119 Evolut valves). No significant differences existed in baseline clinical and echocardiographic characteristics in the Lotus and Evolut groups, respectively (Table I).
Table I
Clinical and echocardiography characteristics*
| Parameter | Evolut (N = 119) | Lotus (N = 71) | P-value |
|---|---|---|---|
| Clinical characteristics: | |||
| Age [years] | 79.1 ±6.2 | 78.9 ±5.8 | 0.8 |
| Males, n (%) | 63 (53) | 33 (47) | 0.4 |
| Body mass index [kg/m2] | 28.6 ±5.2 | 28.7 ±4.7 | 1.0 |
| Follow-up [years] | 1.8 ±0.7 | 2.4 ±0.9 | < 0.0001 |
| NYHA class† | 3.0 ±1 | 3.0 ±1 | 0.2 |
| NYHA class ≥ III, n (%) | 60 (52.2) | 47 (66.2) | 0.06 |
| Diabetes mellitus, n (%) | 41 (34.5) | 22 (31.0) | 0.6 |
| Arterial hypertension, n (%) | 98 (82.4) | 51 (71.8) | 0.08 |
| Atrial fibrillation, n (%) | 44 (37.0) | 29 (40.9) | 0.6 |
| Coronary artery disease, n (%) | 51 (42.9) | 31 (43.7) | 0.9 |
| Creatinine [μmol/l]† | 87.0 ±34.5 | 84.0 ±39.5 | 0.5 |
| Haemoglobin [g/l] | 127.6 ±15.8 | 125.2 ±16.6 | 0.3 |
| Platelets [× 109/l] | 203.2 ±70 | 192.6 ±65.6 | 0.3 |
| Pacemaker before TAVI, n (%) | 12 (10.1) | 7 (9.9) | 1.0 |
| STS score† | 2.4 ±1.5 | 2.3 ±2.3 | 0.7 |
| Acute procedure, n (%) | 14 (11.8) | 6 (8.5) | 0.5 |
| Agatston score† | 2738 ±2440 | 2398 ±1581 | 0.4 |
| Echocardiography characteristics: | |||
| Mean gradient [mm Hg] | 43.2 ±14.9 | 42.4 ±13.1 | 0.7 |
| Aortic valve area [cm2] | 0.7 ±0.2 | 0.7 ±0.2 | 0.7 |
| Aortic regurgitation grade ≥ 3, n (%) | 6 (5.1) | 5 (7.0) | 0.8 |
| LVEF, %† | 55 ±15 | 60 ±20 | 0.2 |
| Mitral regurgitation grade ≥ 3, n (%) | 13 (10.5) | 6 (8.2) | 0.8 |
Procedural and postprocedural characteristics (Table II)
We used balloon valvuloplasty more often in the Evolut group. Open surgical access was used more often in the Lotus group. We observed a slightly but significantly higher drop in haemoglobin and platelets in the Lotus group. The rate of new pacemaker implantation was the same in both groups. Also, consumption of contrast dye during the procedure and the level of troponin and creatinine after the procedure were without significant differences between the study groups.
Table II
Procedural and postprocedural characteristics*
| Parameter | Evolut (N = 119) | Lotus (N = 71) | P-value |
|---|---|---|---|
| Balloon valvuloplasty, n (%) | 34 (28.8) | 4 (5.8) | < 0.001 |
| Open surgical access, n (%) | 87 (73.1) | 71 (100.0) | < 0.001 |
| Contrast dye [ml]† | 100.0 ±34.4 | 100.0 ±45.0 | 0.1 |
| Troponin in 24 h [ng/l]† | 574.0 ±923.6 | 607.4 ±653.5 | 0.6 |
| Creatinine [μmol/l]† | 75.0 ±21.6 | 80.0 ±40.3 | 0.4 |
| Haemoglobin [g/l] | 114.4 ±15.8 | 107.6 ±19.5 | 0.014 |
| Platelets [× 109/l] | 146.3 ±54.2 | 108.8 ±50.0 | < 0.001 |
| AVB requiring PM implantation, n (%) | 34 (28.6) | 20 (28.2) | 1.000 |
| Length of hospital stay after TAVI [days] | 6 ±2 | 7 ±3 | < 0.002 |
Secondary outcomes
The rate of procedural complications was low and without significant differences between groups (2 (3%) patients in the Lotus group and 5 (4%) in the Evolut group died within 30 days of TAVI; no myocardial infarctions or acute kidney injury occurred during TAVI; rates of overt periprocedural CNS injury were 2.8% vs. 2.5%; bleeding complications type 2, 3, or 4 at 30 days 2.8% vs. 1.7%; major vascular complications 4.1% vs. 1.6 %; valve malposition 0 vs. 1.7% in the Lotus and Evolut groups, respectively). All-cause mortality the first year was not significantly different (12.7% vs. 11.8% in the Lotus and Evolut groups, respectively) (Table III).
Table III
Primary and secondary outcomes
Primary outcomes
A total of 52 all-cause deaths occurred during 388 patient-years (mean follow-up: 1.9 ±0.9 years), which translates into 16.4 and 11.0 deaths per 100 patient-years in the Lotus and Evolut groups, respectively. During initial hospitalization 3 patients died because of periprocedural aortic dissection, 3 patients because of heart failure/cardiogenic shock that developed before TAVI, and 1 patient died because of periprocedural cardiac tamponade. Our survival analysis was based on deaths occurring after initial hospitalization for TAVI.
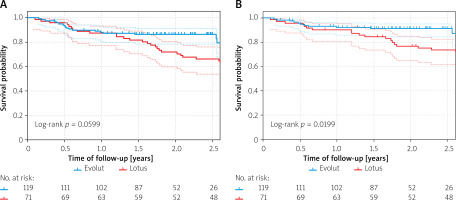
A total of 45 all-cause deaths occurred after initial hospitalization during 388 patient-years (mean follow-up: 2.0 ±0.9 years), which translates into 15.3 and 8.7 deaths per 100 patient-years in the Lotus and Evolut groups, respectively. The logrank test showed higher mortality in the Lotus group for all-cause deaths, but this difference did not reach statistical significance (p = 0.06; RR = 1.8, 95% CI: 0.97–3.3; Figure 1 A).
A total of 30 cardiovascular deaths occurred after initial hospitalization during follow-up, which translates into 11.2 and 5.1 deaths per 100 patient-years in the Lotus and Evolut groups, respectively. The logrank test showed significantly higher cardiovascular mortality in the Lotus group (p = 0.02; RR = 2.4, 95% CI: 1.123–5.075; Figure 1 B).
Subanalysis
In a univariate analysis, we did not find any significant association between all-cause mortality or cardiovascular mortality and the studied categorical variables (Table IV). We also studied the association between all-cause and cardiovascular mortality and continuous variables. We observed a significant association between all-cause mortality and higher creatinine levels after TAVI, and lower haemoglobin level before and after TAVI (Table IV). We revealed an association between cardiovascular mortality and lower haemoglobin and higher creatinine levels before TAVI (Table IV).
Table IV
Univariate analysis of all-cause and cardiovascular mortality and its association with categorical and continuous variables*
| Parameter | All-cause mortality | Cardiovascular mortality | ||
|---|---|---|---|---|
| RR (95% CI) | P-value | RR (95% CI) | P-value | |
| Categorical variables: | ||||
| Male gender | 1.147 (0.629–2.060) | 0.6 | 1.253 (0.611–2.569) | 0.5 |
| NYHA ≥ III before TAVI | 1.179 (0.645–2.155) | 0.6 | 1.667 (0.763–3.641) | 0.2 |
| Diabetes mellitus | 0.784 (0.412–1.495) | 0.5 | 0.970 (0.454–2.072) | 0.9 |
| Arterial hypertension | 0.748 (0.386–1.449) | 0.4 | 0.628 (0.287–1.371) | 0.2 |
| Atrial fibrillation | 1.765 (0.984–3.168) | 0.05 | 1.280 (0.622–2.637) | 0.5 |
| Coronary artery disease | 1.019 (0.566–1.836) | 1.0 | 0.862 (0.415–1.790) | 0.7 |
| Pacemaker before TAVI | 1.135 (0.448–2.877) | 0.8 | 1.007 (0.305–3.320) | 1.0 |
| Balloon valvuloplasty during TAVI | 0.718 (0.303–1.703) | 0.5 | 0.511 (0.154–1.690) | 0.3 |
| Open surgical access | 0.933 (0.358–2.431) | 0.9 | 0.796 (0.269–2.356) | 0.7 |
| Creatinine worsening after the procedure | 1.031 (0.507–2.097) | 0.9 | 0.730 (0.278–1.917) | 0.5 |
| Continuous variables: | ||||
| Age | 1.009 (0.958–1.062) | 0.7 | 0.993 (0.933–1.056) | 0.8 |
| Body mass index | 0.956 (0.898–1.017) | 0.2 | 0.966 (0.896–1.041) | 0.4 |
| Creatinine before TAVI† | 1.063 (0.996–1.138) | 0.07 | 1.079 (1.004–1.161) | 0.04 |
| Creatinine after TAVI† | 1.076 (1.020–1.138) | 0.009 | 1.066 (0.993–1.138) | 0.08 |
| Haemoglobin before TAVI† | 0.757 (0.629–0.911) | 0.003 | 0.728 (0.580–0.914) | 0.006 |
| Haemoglobin after TAVI† | 0.821 (0.680–0.992) | 0.04 | 0.827 (0.656–1.041) | 0.1 |
| Platelets before TAVI† | 0.972 (0.925–1.020) | 0.3 | 0.971 (0.914–1.030) | 0.3 |
| Platelets after TAVI† | 0.999 (0.938–1.051) | 0.8 | 0.976 (0.908–1.051) | 0.5 |
| Troponin after TAVI‡ | 0.994 (0.980–1.008) | 0.5 | 0.997 (0.980–1.017) | 0.7 |
| Agatston score‡ | 0.992 (0.961–1.023) | 0.6 | 1.000 (0.970–1.030) | 1.0 |
| STS score | 1.027 (0.938–1.124) | 0.6 | 0.998 (0.879–1.134) | 1.0 |
| Mean aortic valve gradient before TAVI | 1.001 (0.981–1.022) | 0.9 | 1.003 (0.978–1.028) | 0.8 |
| Mean aortic valve gradient after TAVI | 1.020 (0.971–1.072) | 0.4 | 1.029 (0.999–1.060) | 0.09 |
| Aortic valve area before TAVI | 3.611 (0.767–17.00) | 0.1 | 4.676 (0.689–31.75) | 0.1 |
| Left ventricle ejection fraction | 1.015 (0.982–1.037) | 0.2 | 1.014 (0.987–1.041) | 0.1 |
| Septal wall thickness | 1.039 (0.912–1.182) | 0.6 | 1.072 (0.924–1.244) | 0.4 |
| Inferior wall thickness | 0.998 (0.948–1.367) | 0.2 | 1.232 (0.988–1.535) | 0.06 |
Multivariate analysis using variables identified as significantly associated with mortality in univariate analysis revealed that only the Lotus valve (p = 0.03) and lower haemoglobin level before TAVI were independently associated with cardiovascular mortality. Patients with the same haemoglobin level before TAVI had 2.3-times higher risk of cardiovascular death with the Lotus valve than patients with the Evolut valve (95% CI: 1.1–4.9).
Echocardiography findings (Table V)
The mean aortic valve gradient (AVGm) at 30 days was significantly higher in the Lotus group; this difference remained significant at year 1. No significant aortic regurgitation occurred during the first year in either group. In 2020–2021, we collected 30 echocardiography studies from 40 (75%) living patients with a Lotus valve and compared them with 34 patients with Evolut R, with a mean follow-up of 4.1 ±0.9 years; we found a significantly higher AVGm in the Lotus than in the Evolut group (17.9 ±9.5 vs. 10.2 ±3.5 mm Hg; p = 0.0006). In the Lotus group, 9 (29%) had AVGm ≥ 20 mm Hg, and 3 (10%) suffered from symptomatic re-stenosis needing reintervention. Conversely, in the Evolut group, only 1 patient with moderate aortic regurgitation and AVGm ≥ 20 mm Hg was observed.
Table V
Echocardiographic characteristics after TAVI*
Discussion
To the best of our knowledge, this is the first study evaluating the mid-term outcome of patients with the Lotus valve. Here, we report 2 essential findings: (I) use of the Lotus TAVI valve was independently associated with cardiovascular mortality compared with the Evolut R valve; (II) the gradient with the Lotus valve was significantly higher at 30 days and 1 year after the procedure, and this difference increased during mid-term follow-up.
The Lotus valve was the only fully retrievable prosthesis, with excellent results with regards to minimal paravalvular leak [3, 4, 10]. The disadvantage of this valve is a high risk (about 35%) of significant atrioventricular block during the procedure, requiring permanent pacemaker implantation (PPI) [1, 2] and issues associated with the product delivery system, which was the reason for the valve recall [13, 14]. Although the valve is no longer commercially available, thousands of prostheses were implanted either in controlled trials [3, 10] or in current clinical practice worldwide. Therefore, we are convinced that long-term outcome evaluation of this valve is of the utmost importance, but so far only one study has published 2-year results [10], one has published 5-year results of 11 patients from the initial REPRISE I study [8], and one recent research correspondence of Dumpies et al. [17] published 3- and 5-year all-cause mortality data without further details of the studied patients.
We observed favourable 1-year all-cause mortality (12.7%) in agreement with published studies (11.7% in the RESPOND study [3] and 11.9% in the REPRISE III study [2]). Our results suggest that during the second year after the TAVI procedure survival of patients with the Lotus valve significantly worsens (Figure 1). This is in apparent contrast with the 2-year results of the REPRISE III study [10] in which no difference in all-cause mortality was observed (21.3% vs. 22.5% in the Lotus and CoreValve groups, respectively). The 2-year mortality of patients with CoreValve/Evolut valves in REPRISE III seems to be unexpectedly high, because in the study by Reardon et al. [18] in patients with similar risk profiles (STS 6.9 vs. 7%) 2-year mortality was 15%, which is in agreement with our observation (13.4%).
Despite the nonrandomized character of our study, both groups were well balanced. TAVI procedures (Table III) were accompanied by the low rate of complications without significant differences between groups. The lower number of balloon valvuloplasties in the Lotus group was enabled by valve radial force and its ability to fully retrieve and thus facilitate precise valve deployment. A higher percentage of open surgical access comes from the preferred strategy in this period of our TAVI program, and it is most probably the cause of lower haemoglobin levels after the procedure and longer hospital stay of the patients with the Lotus valve (Table II). Nevertheless, the Lotus valve was independently associated with cardiovascular mortality in multivariate analysis. It should be emphasized that we did not reveal any causal relationship between death and implanted valve.
CoreValve demonstrated lower gradients and larger aortic valve area than Lotus at discharge, but these haemodynamic differences were not associated with any clinical differences at 1 year of follow-up [19]. We also revealed a significantly higher valve gradient in the Lotus group compared with the Evolut group (Table V), and this difference even increased by the end of 4 years of follow-up.
The higher postprocedural gradient in the Lotus valve is most probably based on the intra-annular position of the prosthesis compared with the supra-annular placement of the Evolut valve. The higher position of the TAVI valve in relation to the annulus is associated with lower turbulence intensity. The development of turbulence after TAVI implantation may have effects on the long-term performance and durability of the valves [20]. Nevertheless, in a study of a surgical bioprostheses [21], the superior haemodynamics of the supra-annular position of the valve conferred no apparent clinical benefits.
Five patients with the Lotus valve (7%) suffered from a transitional increase in the gradient of the TAVI prosthesis, which disappeared after anticoagulation therapy and therefore was most probably caused by prosthetic thrombosis. Clinical transcatheter heart valve thrombosis is probably more common than previously considered [22]. It might contribute to valve degeneration [23]. The data about the incidence of clinical valve thrombosis with the Lotus valve are limited [24], but it seems to be higher than that with the self-expanding TAVI prosthesis [22]. Dumpies et al. [17] revealed clinical valve thrombosis in 6.1% and endocarditis in 5.2% of patients with the Lotus valve.
This study has several limitations. First, the retrospective, observational, single-centre design has its own inherent limitations that should be considered before generalization of the results. Second, the high number of PPI in both groups is not consistent with current practice, especially in the Evolut group, whereas in the Lotus group a 28% risk of PPI is appropriate. Although PPI after TAVI is associated with increased long-term mortality [25, 26], identical pacemaker prevalence in the studied groups excluded onset of conduction disturbances associated with TAVI as a cause of increased mortality in the Lotus group. With our increasing experience and the development of implantation technique, the rate of PPI decreased in the last 3 years to a rate similar to contemporary reports (PPI rate 16.8%) [27]. Third, our small sample under investigation and limited number of echocardiography studies in the end of follow-up does not allow definite evaluation of a worse outcome cause in Lotus patients.
Conclusions
Despite several inherent limitations associated with the nonrandomized design of this study, our results suggest that higher rates for cardiovascular mortality during mid-term follow-up were associated with the Lotus compared with the Evolut valve. Occurrence of higher AVGm in Lotus valves found during follow-up suggests the possibility of accelerated prosthesis degeneration as a potential cause of worse outcome.