Minimally invasive robotic mitral valve repair (rMVR) using the da Vinci system is an alternative to other minimally invasive mitral valve (MV) surgical techniques with important advantages such as small skin incisions, reduced risk of wound infection and arrhythmia, less blood loss and shorter hospitalization [1, 2]. The implementation of new robotic technologies, intensified training of surgeons and a greater demand from patients with MV disease may influence the wider use of this technique in clinical practice.
The first procedures of rMVR using the da Vinci system were performed in two Polish patients in autumn 2018 due to symptomatic severe mitral regurgitation (MR) caused by posterior mitral leaflet prolapse (NYHA II/III).
According to a paper published in 2019, rMVR procedures were guided with the transoesophageal echocardiographic support with a 3D option and their good final result was confirmed [3].
Currently, we would like to present the results of transthoracic echocardiography (TTE) performed in 2 patients 30 months after rMVR. Preoperatively, the first patient (No. 1: 46-year-old man) had a deep P2 prolapse with a significant cleft of the scallop causing a large MR jet (Figure 1 A). During the rMVR the P2 cleft was closed, two neochords were placed in the papillary muscles and a Simulus 31 flexible partial band was implanted. In the second patient (No. 2: 56-year-old man) with at least three broken native chords to the P2 scallop and a strongly eccentric jet of MR (Figure 1 B) a triangular resection of the prolapsing segment was performed (Figure 1 C) and a Simulus 35 partial band was also implanted.
Figure 1
A – Severe mitral regurgitation jet in patient No. 1 (arrow); color Doppler. B – Eccentric jet of severe mitral regurgitation in patient No. 2 (arrow); color Doppler. C – Intraoperative view of the mitral valve during robotic surgery in patient No. 2. D – Echo of an artificial chord implanted in the lateral papillary muscle in patient No. 1 (arrow); the absence of regurgitation in color Doppler assessment (small picture). E – Trivial residual mitral regurgitation after robotic surgery in patient No. 2 (arrow)
AML – anterior mitral leaflet, LA – left atrium, LV – left ventricle, PML – posterior mitral leaflet.
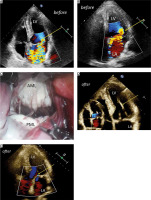
Both patients were followed up carefully (6, 12, 24 and 30 months). Thirty months after rMVR, the first patient had no MR jet (Figure 1 D) and no significant transmitral gradient, and the second patient had only trivial MR (Figure 1 F) and the mean transmitral gradient 3 mm Hg.
In addition to the significant reduction of MR degree, decreased left-sided heart dimensions, tricuspid regurgitation peak gradient and right ventricular systolic pressure were also observed. Important cardiac parameters measured immediately before and 30 months after robotic mitral valve repair with the da Vinci system are included in Table I.
Table I
Echocardiographic parameters before and 30 months after robotic mitral valve repair (rMVR) using the da Vinci system
[i] a-p – antero-posterior, E/A – ratio of maximal velocities of early and atrial wave in mitral inflow, LA – left atrium, LAV – left atrial volume, LAX – parasternal long axis view, LVEDD – left ventricular end diastolic diameter, LVEF – left ventricular ejection fraction, MV – mitral valve, RVSP – right ventricular systolic pressure, TAPSE – tricuspid annulus peak systolic excursion, TRPG – tricuspid regurgitation peak gradient, 4C – apical four-chamber view.
The clinical condition and exercise tolerance of both operated patients were good throughout the entire follow-up. Heart murmurs and arrhythmias were not found.
Although robotic surgery is now widely used in urology or general surgery, it does not appear to be sufficiently used in mitral valve surgery. This minimally invasive surgical procedure has many advantages and benefits for patients, but we should be aware of its high costs and long learning curve [2].
In Chitwood’s report on 279 patients after rMVR using the da Vinci system in East Carolina (the mean follow-up of 815 ±459 days) no/trivial MR in 68.8% of patients, mild in 23.6%, moderate in 5.4% and severe in 2.2% were found [4]. Similarly, the European report published by Navarra et al. on 134 patients after rMVR showed freedom from recurrence of MR greater than mild of 92.5% and 80.7% at 12 and 36 months follow-up, respectively [5]. Chinese authors published their own results of TTE follow-up (range: 14.3–59.4 months, median: 36) in 81 patients who underwent rMVR and only 4.9% of them had mild MR, and 95.1% had no or trace MR [6].
The results of longer follow-up after rMVR were presented in 2021 by Barack et al. They summarized 133 patients after this procedure. In 5-year follow-up the incidence of moderate MR was 18 ±6%, severe MR was 4 ± 3%, and mitral valve replacement 9 ±5% and 5-year survival was 96 ±3%. In the opinion of these authors, the number of 16 operated cases per year was not sufficient to improve the pump time or length of stay over time [7].
Compared to other techniques, the results of rMVR can be impressive. However, it is worth emphasizing that a team approach and excellent communication in the operating room are crucial. To become proficient in robotic surgery, properly selected patients and a significant number of procedures per year are required [2].
Performing the rMVR procedure leads to a significant reduction in MR grade. This method seems to be the least invasive treatment of an MV degenerative disease, which results in good repair durability, especially when used in experienced centers. Good results of rMVR are possible even with the first procedures, despite the long learning curve.











