Summary
Carotid stenosis management guideline recommendation classes are organized according to lesion severity expressed as “percent stenosis”. The “percent stenosis”, along with the presence/absence of neurologic symptoms, serves as the principle parameter in guideline-based decisions on revascularization. Computed tomography angiography (CTA) is a widely used, guideline-approved imaging modality with area stenosis (AS)-derived calculations commonly applied to determine “percent stenosis”. In a study of 300 consecutive patients referred to a major tertiary vascular centre for potential carotid revascularization, we found that replacing the reference standard intra-arterial quantitative angiography diameter stenosis measurement with CTA-AS “percent stenosis” calculation reclassified 76% of “< 50%” cQA-DS measurements to the “50–69%” group, and 58% of “50–69%” measurements to the “≥ 70%” group (symptomatic lesion thresholds). For asymptomatic lesion thresholds, 78% of “< 60%” reference measurements were reclassified to the “60–79%” class, and 42% of “60–79%” crossed to “≥ 80%” class. This magnitude of reclassification error may lead to many unnecessary carotid procedures and patient exposure to potential complications if (and where) CTA-AS is used to determine “percent stenosis”. The CTA-AS reclassification error of carotid stenosis severity is applicable in particular to carotid endarterectomy (when CEA is based on CTA-AS) but not to carotid artery stenting or stent-assisted trans-carotid revascularization that includes verification of stenosis severity with intra-arterial angiography. Until stenosis severity measurement methods applicable to CTA (MRI) become rectified by future guidelines, the AS-based calculations of “percent stenosis” should not be used for decision-making.
Introduction
Despite growing evidence of the role played by plaque morphology in modulating the risk of carotid stenosis-related stroke [1, 2], according to international guidelines [3–6] the degree of carotid lumen reduction (along with the presence/absence of neurologic symptoms) remains the principle parameter in decision-making on revascularization (Table I). Reduction of diameter stenosis (DS) on intra-arterial (catheter) quantitative angiography (cQA) has been the reference standard in reporting carotid artery stenosis severity as “% stenosis” [7, 8]. The cQA-DS measurement has been applied in pivotal revascularization trials as a sole [7, 8] or prevailing [9, 10] technique. Duplex ultrasound (DUS) flow velocities were then formally validated against cQA-DS as a method to estimate stenosis severity [11]. Although DUS has limited precision in “% stenosis” determination [12, 13], it has the fundamental advantage of being easily accessible, non-invasive, and it does not require contrast. Some clinical trials accepted DUS, even using any local-lab criteria of stenosis severity, as the basis for patient enrolment [14, 15]. Other study protocols employed detailed algorithms to verify DUS estimate of stenosis severity with cQA-DS as a prevailing technique [9, 10]. Growing access to non-invasive imaging modalities widely considered less observer-dependent than DUS, such as computed tomography angiography (CTA) or magnetic resonance angiography (MRA), has led to their incorporation in the guidelines [3–6] as a basis for decisions on carotid revascularization (Table II). Since a 3-dimensional imaging technique is typically believed to “naturally” offer more precision than a 2D modality, the “% stenosis” thresholds previously defined with cQA-DS were considered applicable to CTA/MRA [3–6] (Table I, II). With cross-sectional planar visualization of the lumen of stenosed artery (Figure 1), CTA and MRA quantification of carotid stenosis severity is typically based on reduction of the lumen area (area stenosis – AS) [16–18]. Only some [19] radiology reports of carotid stenosis severity by CTA (or MRA) provide also the measurement of DS reduction [19], with the latter calculation closer, at least in theory, to the standard of reference NASCET measurement [20, 21]. The non-linear relationship between AS-based and DS-based “percentage” stenosis measurements (governed by the fundamental formula “area = π • (diameter/2)2”, indicates an “automatic” overestimation of “% stenosis” severity with the AS-based calculation (Figure 2). However, only some carotid stenoses are circular, and several lesion characteristics other than non-circularity (such as calcifications and ulcerations) may affect the relationship between CTA-AS-based “% stenosis” and the cQA-DS standard of reference [19, 21]. Because the guideline recommendation classes are organized according to “% stenosis” categories (Table I), a clinically relevant error might occur when the CTA-AS-based measurements are used for decision-making. However the magnitude of CTA-AS error in stenosis severity classification, and its impact on “gaining” an inappropriate indication to revascularization or lesion (patient) “migration” to another guideline indication class, remains undetermined.
Table I
Carotid “percentage stenosis” thresholds in decision-making on carotid revascularization in primary (asymptomatic patients) and secondary (symptomatic patients) prevention of carotid-related stroke
| Group | ESC/ESVS 2017 | ESO 2021 | SVS 2022 | ESVS 2023 |
|---|---|---|---|---|
| Symptomatic patients | 50–69% CEA (IIa) CAS (IIb) | 50–69% CEA (IIa) CAS (IIb)* | 50–99% CEA “rather than” CAS (Ib) | 50–69% CEA (IIa) CAS (IIb) |
| Symptomatic patients | 70–99% CEA (I) CAS (IIa/b) | 70–99% CEA (I) CAS (IIb)* | (See above) | 70–99% CEA (I) CAS (IIa/b) |
| Asymptomatic patients | 60–99% CEA (IIa) CAS (IIb) | 60–99% CEA (I) | 70–99% CEA/CAS (IIc) | 60–99% CEA (IIa) CAS (IIb) |
The recommendation class for each stenosis category in symptomatic and asymptomatic patients is provided in parentheses that follow the type of revascularization (CEA or CAS). For symptomatic carotid lesions, the FDA-CMS threshold is “≥ 70%“; in addition, CAS can be performed in symptomatic carotid artery stenoses between 50% and 70% in patients at high risk for CEA [22]. The FDA-CMS revascularization threshold in asymptomatic carotid stenosis is 80% [22]. For details see Refs. [3–6, 22].
Table II
Guideline position on the role of CTA (MRA) in evaluation of extracranial carotid stenosis severity as a basis for decision-making on revascularization
| Guideline | Role of CTA (MRA) imaging | Comments |
|---|---|---|
| ESVS 2023 [6] | CTA (MRA) is an alternative to DUS |
|
| SVS 2022 [5] | DUS or CTA (MRA) (can be used to determine lesion severity) | |
| ESC/ESVS 2017 [3] | DUS = first-line imaging modality (first step for screening and diagnosis) CTA (MRA) recommended for evaluating carotid stenosis severity |
|
Figure 1
Principles of diameter stenosis-based and area stenosis-based quantification of carotid lumen reduction expressed as “percent stenosis” using computed tomography angiography (CTA). A 56-year-old neurologically asymptomatic office worker, whose brother-in-law experienced a disabling stroke in relation to atherosclerotic carotid stenosis, presented in a vascular surgery outpatient clinic with DUS examination indicating a border-line left internal carotid artery (LICA) lesion (PSV/EDV 2.1/0.6 m/s consistent with “50–69%” stenosis [11]). CTA request was made to increase the precision of stenosis severity determination and aid further decision-making [22–24]. By CTA, the LICA lumen was not fully circular in the reference plane (A), but there was a rather concentric lumen reduction in the minimal lumen area plane (B). Diameter stenosis (DS)-based calculations, depending on the use of minimal vs. average diameter in the reference plane, show stenosis of “46%” (when the minimal reference diameter is used) or “51%” (with average reference diameter use). Thus, according to diameter stenosis-based calculations, the threshold of “≥ 60% stenosis” was not crossed to consider revascularization of this neurologically asymptomatic lesion [3, 6]. In contrast, with the area reduction-based calculation (see the bottom measurement in panel B) the “percent stenosis” is “71%”, placing this patient in the stenosis severity cohort where revascularization may be performed to reduce stroke risk according to both European and North American guidelines [3, 6]. Surgical revascularization (CEA) could thus be performed, particularly in the case of a radiology report limited to the area-based calculation of stenosis severity, which is not infrequent [19]. It is common that in the case of both AS- and DS-derived calculations provided in the radiology report [19], the “more severe” measurement could be taken as the basis for the clinical decision. Note that carotid artery stenting or trans-carotid stent-assisted revascularization, with their inherent “final” confirmation of lesion severity by intra-arterial angiography (standard of reference), would probably have resulted in deferring any interventional management because in this study patient catheter quantitative angiography (cQA, NASCET method) revealed LICA stenosis severity of 47%. Medical therapy was maximized to reduce stroke risk [3, 25, 26], and the patient is monitored yearly in a certified DUS lab for potential stenosis progression [3, 4, 6] along with imaging and clinical observation for de novo presence of other increased stroke risk characteristics [3, 4, 6, 25, 26]. A documented increase in stenosis severity would trigger the NeuroVascular Team to re-consider recommended management, which would involve the patient’s opinion (based on complete information on treatment options [26]) in the decision-making process
PSV – peak systolic velocity, EDV – end diastolic velocity, CTA – computed tomography angiography, CEA – carotid endarterectomy, cQA – catheter (intra-arterial) quantitative angiography, NASCET – North American Symptomatic Carotid Endarterectomy Trial.
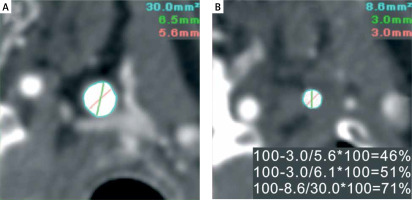
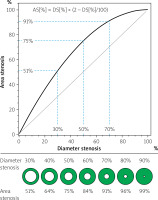
Figure 2
Relationship between DS-based and AS-based calculation of “percentage stenosis” in circular lesions. The graph shows a curvilinear relationship between area stenosis (AS) and diameter stenosis (DS) in concentric lesions described by the equation AS[%] = DS[%] × (2 – DS[%]/100) arising from the fundamental mathematical formula “area = π • ( d/2)2”. The panel below the graph shows “% stenosis” with the diameter-based calculation (top) and area-based calculation (bottom) for typical guideline-indicated thresholds for decision-making, including decisions on revascularization to reduce the risk (Table I). With these 2 methods of calculating stenosis severity in clinical use [19] the numeric “outcome” of stenosis severity determination is fundamentally dependent on the calculation method. Note that despite yielding elementarily different numeric “outcomes” of “% stenosis”, the 2 methods are perfectly correlated (r = 1, p = 0.000). Any comparison of these 2 methods limited to assessment of “correlation” as a basis for replacing one technique with the other would be fundamentally flawed because other statistical approaches (such as Bland-Altman analysis, kappa statistics, frequency distribution analysis, etc.) would be required. Presentation concept based on Alexandrov et al. [27] and Ota et al. [28]; modified
Aim
In a series of consecutive patients referred for potential carotid revascularization to a tertiary vascular centre, we evaluated the impact of replacing the pivotal trials standard measurement of carotid stenosis severity (cQA diameter stenosis) with stenosis severity determination using CTA area-based calculations on misclassification of carotid lesion severity as suggested by the mathematical relationship between “% area stenosis” and “% diameter stenosis” (Figure 2).
Material and methods
This was a prospective study in consecutive patients referred for potential carotid revascularization to a tertiary vascular centre performing high-volume carotid revascularization by means of CEA, CAS, and TCAR in patients with indications established by the multispecialty NeuroVascular Team and a final decision on the treatment modality incorporating the patient’s informed opinion [29–38]. Referral stenosis severity had to be at least “50%” according to the modality and measurement method used by the referring centre. The referral centre imaging could involve any of the guideline-accepted non-invasive techniques (Table I). Chronic kidney disease with glomerular filtration rate < 30 ml/min was an exclusion criterion. In every study patient, CTA and catheter angiography were performed within 1 month of each other. In patients with bilateral carotid stenosis, the symptomatic or more severe lesion was studied.
Computed tomography angiography
Image acquisition of the supra-aortic vessels was performed on a 64-multi-detector-row CT system (Somatom 64, Siemens, Erlangen, Germany) using a routine imaging protocol. A slice thickness of 1.5 mm (1 mm collimation, feed 5 mm/s) and a reconstruction interval of 1.0 mm were used. Bolus tracking technique was used to ensure optimal intravascular contrast media density. The contrast agent (Ultravist 300 mg/ml, Schering AG, Berlin, Germany) volume for CT angiography was 80 ml with a saline chaser bolus of 30 ml using a flow rate of 3 ml/s via a 1.3 mm (18G) cannula through the antecubital vein. CTA analysis was performed by agreement of 2 senior radiologists with, respectively, > 20 years and > 15 years of experience in reporting carotid CTA. The %AS was computed as 100-MLA/RA*100%, where MLA is the minimal lumen area and RA is the reference area (example in Figure 1). In each case, 3 measurements were performed, and the average %AS was taken for further analysis.
Intra-arterial angiography
Selective digital angiography of the index carotid artery was performed with an Axiom Artis Zee angiograph (Siemens) in multiple (4-7, median 4) angulated projections to identify the narrowest lumen diameter while minimizing foreshortening and avoiding overlap of side branches. The view where the stenosis was tightest was used for quantitative measurements (Quantcor QA v5.0, Siemens). Stenosis severity measurements were performed by agreement of 2 angiographic core lab analysts and were verified by an angiographic corelab supervisor. The %DS was computed as [(RD-DS)/RD]*100% according to NASCET (North American Symptomatic Carotid Endarterectomy Trial) method [7], where DS is diameter stenosis and RD is reference diameter. In the projection showing the narrowest lumen diameter 3 measurements were performed and the average %DS was taken for further analysis.
The CTA and cQA analyses were blinded against each other. The study protocol was approved by the local Ethics Committee, and all patients provided written informed consent.
Treatment decisions were based on cQA-DS and guideline criteria other than stenosis severity.
Statistical analysis and classification error demonstration
Categorical variables were presented as numbers and percentages. Continuous variables were expressed as medians [Q1–Q3]. The Mann-Whitney U test was used to assess differences between the groups. Correct stenosis severity classification by CTA-AS measurement was defined as CTA-AS measurement placing the patient (lesion) in the same guideline-defined category (i.e. < 50%, 50–69%, and ≥ 70% “percentage stenosis severity” for symptomatic patient management thresholds and < 60%, 60–79%, and ≥ 80% “percentage stenosis severity” for asymptomatic patient management thresholds) as the cQA-DS (reference) measurement [3–6]. Stenosis severity misclassification by CTA-AS measurement was defined as CTA-AS calculation placing the lesion (patient) in another guideline category. The magnitude of misclassification error was expressed, for each specific lesion severity category according to ESC/ESVS [3], ESO [4], SVS [5], ESVS [6], or CMS-FDA [22], as the proportion of patients (lesions) migrating to another category of the same guideline when stenosis severity was calculated as CTA-AS %reduction rather than the reference cQA-DS %reduction. Probability values < 0.05 were considered statistically significant.
Results
Baseline clinical data of the 300 study patients are provided in Table III.
Table III
Baseline characteristics of the study group
| Parameter | Total study group n = 300 | Symptomatic* patients n = 108 (36.0%) | Asymptomatic patients n = 192 (64.0%) | P-value# |
|---|---|---|---|---|
| Age [years] | 66 [60.0–72.0] | 67.0 [59.0–71.5] | 66.0 [61.0–72.0] | 0.365 |
| Female gender | 108 (36.0) | 63 (58.3) | 130 (67.7) | 0.121 |
| Arterial hypertension | 266 (88.7) | 93 (86.1) | 174 (90.6) | 0.20 |
| Diabetes | 96 (32.0) | 28 (25.9) | 68 (35.4) | 0.064 |
| Insulin | 31 (10.3) | 9 (8.3) | 22 (11.5) | 0.277 |
| h/o myocardial infarction | 76 (25.3) | 32 (29.6) | 44 (22.9) | 0.152 |
| Smoking (current or past) | 160 (53.3) | 59 (54.6) | 101 (52.6) | 0.923 |
| CAD | 201 (67.0) | 73 (67.6) | 128 (66.7) | 0.87 |
| PAD | 45 (15.0) | 18 (16.7) | 26 (13.5) | 0.17 |
| BMI | 27.7 [25.7–30.1] | 27.8 [25.9–30.2] | 27.7 [25.5–30.1] | 0.95 |
| Creatinine [μmol/l] | 85 [74–101] | 83 [74–103] | 85 [74–100] | 0.66 |
| eGFR < 60 [ml/min] | 65 (22.2) | 22 (20.4) | 43 (22.4) | 0.47 |
Based on the (reference) cQA %DS measurement, with regard to lesion severity decision-making categories in symptomatic patients, there were 14.7% lesions in the “< 50% stenosis” category, 72.0% lesions in the “50–69% stenosis” category, and 13.3% in the “≥ 70% stenosis” category (Figure 3 left bar). As per decision-making thresholds in asymptomatic lesions, based on the (reference) cQA %DS measurement, there were 54.0% lesions in the “< 60% stenosis” category, 42.3% lesions in the “60–79% stenosis” category, and 3.7% in the “≥ 80% stenosis” category (ESC/ESVS/ESCO and CMS-FDA thresholds, Figure 4 A, left bar). With regard to the SVS threshold in asymptomatic lesions, 86.7% of lesions fell into the “< 70% stenosis” category and 13.3% into the “≥ 70% stenosis” category (Figure 4 B, left bar) according to the (reference) cQA %DS measurement.
Figure 3
Effect of replacing – in symptomatic lesions – cQA diameter stenosis-based measurement of carotid “% stenosis” (reference standard) with CTA-derived area stenosis calculation on lesion (patient) migration to another guideline category. Stenosis severity-based grounds for decision-making in symptomatic lesions (patients) are presented in a bar graph format. The bar on the left shows proportions of lesions (patients) in respective guideline categories according to lesion severity (“% stenosis”) by the reference standard, cQA. The bar on the right illustrates the same lesions (patients) stratified according to stenosis severity by CTA-AS calculation. Note that change from cQA-DS to CTA-AS-based calculation results in a major “migration” of lesions below the revascularization threshold (green) to revascularization consideration (yellow). Similarly, there is a major “migration” from the “50–69%” lesion severity category (yellow box on the left) to a “higher” class of indications to revascularization (“≥ 70% stenosis, red on the right). Proportions of “migrating” lesions (patients) are provided below each of the arrows. See text for details. ESC – European Society of Cardiology, ESVS – European Society of Vascular Surgery, ESO – European Stroke Organisation, SVS – Society for Vascular Surgery, CMS – Centres for Medicare & Medicaid Services, FDA – Food and Drug Administration, cQA – catheter (intra-arterial) quantitative angiography, CTA – computed tomography angiography, DS – diameter stenosis, AS – area stenosis. *SVS Guidelines pool “50–69% stenosis” and “≥ 70% stenosis” in one decision category
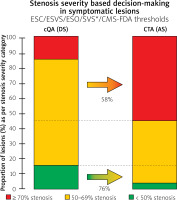
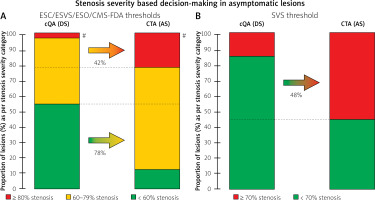
Figure 4
Effect of replacing – in asymptomatic lesions – cQA diameter stenosis-based measurement of carotid “% stenosis” (reference standard) with CTA-derived area stenosis calculation on lesion (patient) migration to another guideline category. Stenosis severity-based grounds for decision-making in a symptomatic lesions (patients) are presented in a bar graph format. In A (ESC/ESVS/ESO/CMS-FDA thresholds) and B (SVS threshold) the bar on the left shows proportions of (lesions) patients in respective guideline categories according to lesion severity (“% stenosis”) by the reference standard, cQA. The right bar in A and B illustrates the same lesions (patients) stratified according to stenosis severity by CTA-AS calculation. Note that change from cQA-DS to CTA-AS-derived calculation results in a major “migration” of lesions below the revascularization threshold (green) to revascularization consideration (yellow in the right bar in A) or revascularization indication (red in the right bar in B). As per CMS-FDA decision-making threshold in asymptomatic lesions (A), there is also a large magnitude of “migration” of “60–79%” lesions (i.e. below the threshold for intervention) to erroneously “achieving” stenosis severity above the revascularization threshold. As a result, the “60–79%” group is increased 1.6-fold and the “≥ 80%” group is increased 5.8-fold. For the SVS guideline threshold (B) nearly half of the (lesions) patients with no indications to intervention (green) “migrate” to the revascularization group (red); thus, the group of patients considered for intervention is increased 4.15-fold. See text for details. ESC – European Society of Cardiology, ESVS – European Society of Vascular Surgery, ESO – European Stroke Organisation, SVS – Society for Vascular Surgery, CMS – Centres for Medicare & Medicaid Services, FDA – Food and Drug Administration, cQA – catheter (intra-arterial) quantitative angiography, CTA – computed tomography angiography, DS – diameter stenosis, AS – area stenosis
Reclassification with CTA-AS-based “% stenosis” measurement
For symptomatic lesion categories, CTA-AS-based calculation of “% stenosis” reclassified 76% of “< 50% cQA-DS stenoses” to the “50–69% stenosis” category (resulting in an erroneous “de novo” indication to revascularization) whereas 58% of “50–69% cQA-DS stenosis” measurements migrated to the “≥ 70% stenosis” category (Figure 3, right bar).
For asymptomatic lesion categories, 78% of cQA-DS measurements showing “< 60% stenosis” (i.e. below the threshold for revascularization) became CTA-AS reclassified to the “60–79% stenosis” recommendation group, consistent with migration from the lack of an indication to revascularization (Table I) to revascularization “considered” or “indicated” according to ESC/ESVS/ESO thresholds. Regarding the CMS-FDA revascularization threshold of “≥ 80% stenosis” in asymptomatic patients, 42% of “60–79%” cQA-DS-based measurements (revascularization not indicated) crossed to the “≥ 80%” class (“revascularization may be performed”; Figure 4 A). For the SVS threshold of “70% stenosis”, 48% of lesions (patients) migrated from the “no revascularization” to the “revascularization” class (Figure 4 B).
In aggregate, using CTA-AS instead of cQA-DS enlarged the “60–79%” and “≥ 80%” lesion severity classes by 1.6- and 5.8-fold respectively, whereas the “≥ 70%” class increased 4.15-fold. No underestimation of stenosis severity occurred with CTA-AS measurement against the cQA-DS reference standard.
Discussion
Carotid stenosis management guideline recommendation classes are organized, in both neurologically symptomatic and asymptomatic patients, according to lesion severity expressed as “% stenosis” (Table I) [3–6].
The principal finding from our present analysis is that the change from cQA-DS (reference standard) to stenosis severity calculation derived from AS on CTA results in a “migration” of a major proportion of lesions (patients) from below the revascularization threshold to the guideline lesion severity category indicating consideration of revascularization, in both the symptomatic (Figure 3) and asymptomatic (Figure 4) cohorts (an erroneous “gain” of revascularization indication). In addition, use of the CTA-AS-based calculation causes a large “migration” of “50–69%” and “60–79%” lesions to a “higher” class of indications to revascularization. Such an erroneous “de novo” (i.e. resulting purely from the method of calculation) “indication” to revascularization (or consideration of revascularization) is particularly concerning because the patients, with lack of guideline-based criterion for surgery, become “operable” if the method to measure the stenosis severity is changed from cQA (standard reference) to CTA-AS-based calculation. This indicates that a significant number of unnecessary carotid procedures may be performed where/when these are based on CTA-AS-derived “lesion severity” in the absence of verification with intra-arterial angiography.
Recent large-scale analysis from the US Vascular Quality Initiative (VQI) indicates that the preoperative imaging modality used before CEA varies widely, with 57% of patients receiving only a single preoperative imaging study (DUS – 46%, CTA – 7.5%, MRA – 2.0%, catheter angiography – 1.3%) [39]. Thus, today less than half of patients seem to receive > 1 preoperative imaging study [39]. In clinical practice DUS is considered the fundamental, first-line imaging method to detect carotid stenosis [3]. Nevertheless, noninvasive vascular laboratories show large variation in the diagnostic criteria used to classify the severity of carotid artery stenosis using DUS [23, 24]. Despite improvements in ultrasound scanners and standardization of scanning protocols, considerable variability remains in reporting the severity of internal carotid artery stenosis [40]. Differences in diagnostic criteria to interpret DUS result in significant variation in classification of carotid artery stenosis, probably leading to differences in the number of revascularizations [23] if DUS alone is used as a single or protocol-mandated [14, 15] imaging modality. Although there is a definite correlation between the velocities obtained by DUS and percent stenosis on intra-arterial arteriography, this relationship is variable [12]. DUS velocity measurement may be affected by a number of variables other than the lesion characteristics, such as heart rate/cardiac output, blood pressure, or contralateral occlusion [12]. It is thus unlikely that any further refinements in the diagnostic velocity thresholds could lead to improved DUS accuracy compared to arteriography [40]. Overall, there is a growing consensus that DUS should not be used as a single modality in carotid stenosis decision-making. On the other hand, it is important to realize that DUS when used as a “second” imaging modality may fail to correct any potential error of CTA (or MRA) [41]. The notion that “No technique on its own is accurate enough to replace catheter angiography. Two non-invasive techniques in combination, and adding a third if the first 2 disagree, appears more accurate, but may still result in diagnostic errors” [41] continues to be valid.
VQI data suggest that in nearly 50% of patients today CTA is the sole imaging modality before CEA [39]. According to current SVS [5] and ESVS [6] guidelines, DUS and CTA (MRA) are considered “equivalent” or “alternative” (Table II). However, it must be noted that the role of CTA in decision-making in everyday clinical practice is particularly important because CTA is widely believed to be more “objective” and “observer-independent” than DUS. Several CTA limitations (e.g. the “blooming” artifact of calcifications) are well-known [42], but there seems to be a widespread absence of understanding that the “numeric” outcome of CTA evaluation of carotid stenosis severity, reported as “% stenosis”, may be largely dependent on the calculation method used (Figure 2).
Although the theoretical basis for fundamental differences between DS- vs. AS-based calculations of carotid stenosis severity, arising from the “area = π • (diameter/2)2” formula, were noted in early studies by Alexandrov et al. [27] three decades ago and were further highlighted by Ota et al. [28], the mathematics-based relationship (Figure 2) has made it neither to the guidelines [3–6] nor to common clinical practice [19]. Furthermore, radiology reports rarely aid in resolving the decision-making problem [19]. A recent survey indicates that the majority of radiology reports are limited to providing either DS- or AS-based calculation, with only a minority (≈ 10%) including both “% diameter” and “% area” reduction [19]. The latter, paradoxically, may be further confusing to (at least some) clinicians, who may tend to choose the “more severe” of the 2 estimates of carotid stenosis as grounds for their decision (“worse result application scenario”) [43]. There is no “mathematical” (Figure 2) doubt that area stenosis-based reports can lead to unnecessary carotid revascularization procedures, particularly with CEA as the revascularization modality not involving the cQA-DS verification of stenosis severity [44]. Our present work demonstrates, for the first time, the size of the potential clinical error as a result of CTA-AS determination of carotid “% stenosis”. Using the guideline thresholds of carotid stenosis severity applicable to the clinical decisions, we estimate the proportion of asymptomatic and symptomatic lesions (patients) wrongly classified to another cohort of the guideline recommendation as a result of using the area stenosis measurement as a replacement for the standard of reference (Figures 3, 4). By evaluating the size of misclassification error (expressed as proportion of lesions “migrating” to another class of indications), we estimate the magnitude of the grounds for erroneous revascularization with CTA-AS measurement of carotid “% stenosis”.
It is surprising that, with its potential significant impact on erroneous decision-making (Figure 2), the issue of “diameter” versus “area” stenosis severity calculation has not received the attention it would deserve from the standpoint of its mathematical basis. While initial clinical studies used cQA-determined DS as the basis to test hypotheses linking stenosis severity to the stroke risk (and the impact of revascularization on reducing the stroke risk) [7], a rapid development of non-invasive techniques (along with a small but non-negligible risk of catheter angiography when used as the first-line modality to determine stenosis severity) has led to a nearly automatic incorporation of non-invasive techniques into every-day clinical decision-making and the study inclusion criteria.
For several reasons, the relationship between DS-derived and AS-derived “% stenosis” in the clinical setting may be more complex than that arising from the mathematical formula “area = π • (d/2)2”. First, only a proportion of lesions are circular [19]. Secondly, lesion ulceration(s) and calcification(s) affect the differences between CTA and cQA visualization as well as the CTA-AS vs. cQA-DS measurement of stenosis severity [19]. In calcified plaques, conventional CTA determination of stenosis severity, particularly at higher stenosis grades, may be hampered by blooming artifacts (present in up to 90% of calcified arteries [42]), resulting in an overestimation of the actual stenosis grade [42]. In addition dense calcium deposits may cause streak and beam hardening artifacts [42], impeding the accurate diagnosis of stenosis severity. Recent work by Horev et al. [21], who compared NASCET measurements of carotid stenosis severity using cQA and CTA, revealed that out of 90 patients with significant stenosis on CTA (thus being candidates for CEA), only 70 (78%) were found to have a significant stenosis on cQA by the NASCET method. Thus, the CTA overestimation error of “% diameter stenosis” wrongly classified 22% of lesions (patients) to the revascularization cohort. These authors concluded that despite ongoing radiological progress, the specificity of CTA in accurately assessing carotid stenosis remains relatively low, particularly in patients with both moderate stenosis and heavily calcified plaques [21]; consequently, patients could be referred for unnecessary CEA surgery and may become exposed to associated potential complications [21]. Although Horev et al. [21] did not move on to evaluate, as a “next” step, the error arising from the use of area reduction calculations rather than (or in addition to) the NASCET-type measurements [19, 21] – their critically important finding should not be ignored by advocates of using CTA-DS as the basis for clinical decisions as per the guideline thresholds (Table I).
With the stenosis “percent severity” as the key guideline-indicated parameter in decision-making on revascularization in carotid-related stroke prevention (Table I) and the CTA position as a guideline-approved imaging modality (Table II), it may be considered surprising that the CTA evaluation of carotid stenosis seems to have never undergone a formal, rigorous validation. Table IV lists key studies that evaluated CTA (with a focus on CTA-AS calculation use) as a method to determine carotid stenosis severity. A PubMed search (and references cross-search) identified 8 studies, with the average number of evaluated vessels being ≈ 180 [17, 18, 20, 41, 44–47]. Most of the studies performed so far, including the largest one (575 arteries) [17], used correlation as the fundamental tool to compare the different imaging methods (Table IV). Only 2 studies included the reference standard cQA-DS (NASCET) measurements [20, 41]. Reported correlation coefficients between CTA-AS and CTA-DS (“NASCET-type” measurement) varied from “weak (r = 0.32) and statistically not significant” [46] to “strong (r = 0.938) and statistically significant” [17], with the latter finding interpreted as “no significant difference” between the area-derived and diameter-derived measurements [17]. One study [47] concluded that “CTA-AS correctly classifies vessels needing surgical intervention” on the basis of comparing CTA-AS measurements with DUS evaluation that is known to have a poor discriminatory value [12, 23, 24, 40, 48, 49]. Only 2 studies [20, 44] (Table IV) applied κ statistics, which is an appropriate tool for comparing diagnostic methods.
Table IV
Key studies evaluating CTA-based determination of carotid stenosis severity, with a focus on the “% area stenosis” measurement
| Author Year | No of vessels | Main findings | Comments |
|---|---|---|---|
| Patel SG 2002 [41] | 34 |
| |
| Cinat M 2003 [45] | 216 |
| |
| Zhang Z 2005 [20] | 72 |
|
|
| van Prehn J 2008 [46] | 37 |
| |
| Carnicelli AP 2013 [17] | 575 |
|
|
| Müller M 2015 [18] | 85 |
| |
| Samarzija K 2018 [47] | 113 |
| |
| Arous EJ 2021 [44] | 320 | and |
It needs to be noted that a “correlation” of any 2 methods, even if strong (as per the correlation coefficient value approaching 1) and “highly” statistically significant, is not sufficient as a basis to replace one imaging technique with another. In an illustrative example, a series of measurements “1.0, 2.0, 3.0, 4.0, 5.0, 6.0, etc.” performed according to “Method A” show a perfect (r = 1, p = 0.000) correlation with a series of “Method B” measurements (“1.8, 2.8, 3.8, 4.8, 5.8, 6.8, etc.”) and show a similarly perfect correlation with another series of measurements using “Method C” (“1.3, 2.6, 3.9, 5.2, 6.5, 7.8, etc). However, the “Method A” measurements (of, say, “lesion severity”) show values greater by 13.3–80.0% than measurements in series A. The “Method C” measurements are all systematically overestimated by 30% in relation to “Method A” measurements. It is obvious that while “Method B” cannot simply replace “Method A”, what “Method C” requires is 30% correction (re-setting the threshold) to achieve values identical with those obtained with “Method A”. Comparisons involving established techniques to compare diagnostic methods other than an isolated analysis of “correlation”, such as Bland-Altman [50], frequency distribution (histograms, with evaluation of a potential shift) [44, 51], receiver operating characteristics analysis (for cut-offs of interest) [49, 52, 53], and κ statistics [20, 44], are needed before stating that one diagnostic method can be replaced with another.
With the stenosis severity as a key diagnostic and revascularization parameter, evolution of inclusion criteria in clinical trials of stroke prevention suggests that at least some investigators have realized the importance of rigorous assessment of stenosis severity. Approaches have varied, from freedom to use any local DUS criteria to enrol on the one end [14, 15] to detailed algorithms incorporating the recommendation by Patel et al. [41] (Table II) of cQA-DS as the “final” resolving technique in case of discrepancies in non-invasive assessment [9, 10]. CREST-1 involved the use of cQA-DS as the basis of patient enrolment or cQA-DS as the verification method in the case of lack of agreement between DUS and CTA/MRI (with a priori greater “% severity” thresholds for CTA/MRI than those with DUS) [9]. In ACT-1, angiogram demonstrating ≥ 80% DS was accepted as an inclusion criterion [10] or, if DUS was compromised and the degree of stenosis could not be established, cQA-DS was recommended as the prevailing technique [10]. Another standardization approach, applied in more recent FDA-IDE studies, incorporates screening committees that verify the protocol-required stenosis severity using at least 2 techniques prior to patient entry into the study [54]. Such an approach minimizes potential erroneous entry into the trial of patients with overrated “% stenosis” lesion severity that as we presently show (Figures 3, 4) might arise, at least in some cases, from adoption of CTA-AS-based measurements.
A large degree of reclassification error with CTA-AS calculations, as identified in this study (Figures 3, 4), is broadly consistent with the recent observation that calculations based on CTA-AS led to a 1.9-fold increase in the number of stenoses characterized as “> 70%” and a 3.8-fold increase in the number of stenoses characterized as “> 80%”, in comparison to NASCET DS calculations [44]. This prompted the conclusion that “AS calculations can lead to unnecessary carotid revascularization procedures” [44] that is broadly consistent with our findings (Figures 3, 4), particularly as ≈ 30–50% of carotid CTA reports include the AS-based measurements [19] while ≈ 20–30% are limited to the calculation of area reduction [19]. The clinical problem of CTA-AS-based unnecessary carotid revascularization procedures is largely limited to conventional surgical revascularization (CEA) because percutaneous (CAS) or transcarotid stent-assisted (TCAR, TCR) revascularization involves a conventional angiographic (re-)evaluation of stenosis severity.
Limitations
Our evaluation is based on corelab-type assessments; thus, there may be some differences with real-life reports [19] that, for instance, do not always seek agreement between observers. Furthermore, the exact proportions of “reclassified” patients (lesions), as a result of using CTA-AS calculation rather than cQA-DS, may vary somewhat with differences in the referral cohorts such as proportions of, e.g. eccentric vs. concentric lesions, calcific vs. non-calcified lesions or “borderline” lesions. Thus, the exact proportion of patients (lesions) affected by the carotid stenosis severity overestimation with use of the AS-based measurement may, to some extent, vary with different referral population characteristics. It will also vary with the proportion of radiology reports including – or limited to – the AS-based measurements [19], and the decisions of clinicians to use “one” or “the other” measurement in case both “% diameter stenosis” and “% area stenosis” are provided (or the decision to proceed to a further verification of stenosis severity using intra-arterial angiography or intravascular ultrasound [55]). However, the fundamental rule of mathematics that the area equals π • (d/2)2 holds as the basis for AS-determined vs. DS-determined carotid stenosis severity, with the AS overestimation remarkably relevant clinically for lesions at (and above) the revascularization threshold (Figures 3, 4). We have deliberately related CT-AS measurements to cQA-DS by conventional angiography (rather than CTA-DS) because cQA-DS (rather than CTA-DS) is the standard of reference measurement [7–10]. Thus, our analysis did not include the “intermediate” step of comparing CT-AS measurements with CT-DS measurements, which was the focus of recent analysis from the University of Massachusetts in a cohort similar to ours (n = 320) [44]. Rather, we compared CT-AS directly with cQA-DS, which is the clinically-relevant comparison [7, 8, 11], particularly because CTA-DS measurements incorporate the CTA “general” limitations in relation to cQA [21, 55]. Although semi-quantitative scoring systems exist to classify the degree of calcifications [35], blooming continues to pose a significant challenge in CTA evaluations of carotid severity [21, 42, 56]. Indeed, whether CTA-based calculation of “% diameter stenosis” (rather than the “% area stenosis” as evaluated in the present study) could routinely replace the pivotal trials standard of cQA-DS NASCET measurements remains uncertain [18, 41, 45–47], and this subject is beyond the scope of the present study. The relationship between cQA-DS and CTA-DS measurements has recently been evaluated in a cohort similar to ours (n = 320) [44] (Table IV). Concordance between CTA-AS and CTA-DS was poor both for ≥ 70% stenosis (κ = 0.32) and ≥ 80% stenosis (κ = 0.25) [44]. The CTA-AS calculation of “≥ 70% stenosis” was confirmed by CTA-DS measurement in 51.4% lesions whereas the CTA-AS reading of “≥ 80% stenosis” was confirmed as “≥ 80% stenosis” by CTA-DA in only 26% [44]. Unfortunately, these investigators did not present their findings in a way clearly indicating the scope of patient (lesion) “migration” to another guideline indication class (i.e. the clinical practice-relevant impact of the magnitude of CTA-AS “% stenosis” severity overestimation error, cf., Figures 3, 4). Thus our results relating CTA-AS to cQA-DS measurement are largely consistent with those relating CTA-AS to CTA-DS [18, 47] while the CTA-DS measurement incorporates the CTA limitations in relation to cQA [21, 42, 56] and lacks any specific validation in clinical trials. MRA, not assessed in the present study of AS-based measurements, is not typically used in our institution as a diagnostic modality, consistent with its overall limited use in the determination of carotid stenosis diagnosis and severity in clinical practice [39]. However, the mathematical principle determining the relationship between AS and DS (Figure 2) applies, in principle, also to MRA evaluation of carotid “% stenosis”.
In summary, the present work evaluated the impact of using the CTA-AS calculation method of carotid stenosis severity in consecutive patients referred for potential carotid revascularization on gaining an erroneous indication to revascularization or change in the revascularization indication level. Stenosis severity categories were according to guideline-recommended thresholds for specific decision-making, including an indication to revascularization (or recommended consideration of revascularization) (Table I). We found that the mathematical basis for CTA-AS overestimation of stenosis severity against the standard of reference (Figure 2) has a clinically relevant impact that we demonstrate, for the first time, as proportions of lesions (patients) “migrating” to another guideline category of indications if (when) the CTA-AS-based calculation is used instead of the cQA-DS standard measurement (reclassification error, Figures 3, 4). With the significant proportion of CTA-AS calculations reported in clinical practice as the measurement of carotid “% stenosis” [19], the clinical impact of AS-based overestimation of carotid stenosis severity (Figures 1–4) is likely to be major. This is particularly relevant to surgery (CEA) because both CAS and TCAR/TCT involve routine angiographic verification of stenosis severity (enabling us to avoid unnecessary procedures) while CEA is typically performed on the basis of CTA that may [3, 39] (or may not [5, 6, 39]) be interpreted in combination with DUS assessment, the discriminatory role of which is limited [12, 40, 41].
Further work from our group will involve systematic validation of DUS, CTA, and cQA measurements of carotid stenosis severity against the vascular imaging gold standard – intravascular ultrasound. In this context, it needs to be noted that the lumen reduction represents “only” the luminal effect of the atherosclerotic disease process occurring in the arterial wall. Evidence is accumulating that plaque morphology, even with optimized medical therapy employed [57], may play a clinically relevant role that is, in its relation to stroke risk, at least as important as the stenosis luminal severity [2, 58]. Carotid-related stroke risk stratification models are needed that incorporate the novel imaging and clinical features together with the conventional parameter of stenosis severity [3–6, 26].
Conclusions
Replacing the pivotal carotid trials reference standard cQA-DS measurement with CTA-AS-based calculations to determine carotid “% stenosis” results in a major reclassification of stenosis severity as per the guideline decision-making thresholds. In consequence, many unnecessary carotid procedures may be performed (where/when) based on CTA-AS-derived “lesion severity” in the absence of verification by catheter angiography. Future guidelines should address stenosis severity measurement methods with the different imaging modalities approved by the guidelines; the CTA-AS “% stenosis” thresholds may require adjustment.







