The fetus is dependent on the maternal supply of calcium, and alteration in maternal calcium homeostasis can affect calcium homeostasis in fetus and neonate [1, 2].
A 16-days old female presented with recurrent, brief (10–15 seconds), multifocal seizures beginning at day 14 of life, not associated with fever, poor feeding or lethargy. She was born at full term following an uncomplicated pregnancy with a birth weight of 3300 g. The baby had a smooth perinatal transition with APGAR score of 8/10, discharged on day 2 of life and was mixed fed with breast and diluted cow’s milk. Baby was receiving 400 IU of vitamin D3 from birth.
Metabolic investigations for seizure revealed normal blood glucose (82 mg/dl), low serum total calcium level 5.8 mg/dl (8.5–10.5) with ionized calcium 0.44 mmol/l (1.05–1.37), serum phosphate (P) 9.1 mg/dl (2.7–4.5), and Alkaline phosphatase (ALP) was 299 U/L (42–128). Her PTH and 25-hydroxy vitamin D [25(OH)D] levels were relatively low, 5.45 pg/ml (10–65) and 18.56 ng/ml (20–50 ng/ml) respectively. The baby was managed with intravenous calcium initially followed by oral activated vitamin D (Rocaltrol; 0.25 µg OD), oral calcium (calcium carbonate; 150 mg/kg/day), and oral 25OH vitamin D (cholecalciferol; 2000 IU OD). Phenobarbitone initiated at outside hospital was discontinued at discharge. The vitamin D and calcium were discontinued after two months, and subsequent calcium of baby was normal (Ca – 9.1 mg/dl, P – 4.2 mg/dl).
Mother, 23 years old, belonged to lower middle class and had 1–2 hour of sunlight exposure during household work. She was clinically asymptomatic. Her evaluation revealed a serum total Ca level of 12.1 mg/dl with serum phosphate level of 2.7 mg/dl, and serum ALP level was 359 U/l. Urinary calcium 21.4 mg/dl and phosphate 41.9 mg/dl and urinary calcium creatinine ratio (0.4) were elevated; however, no evidence of nephrocalcinosis on sonography. The PTH level was high (299 pg/ml; 330 pg/ml) on two separate occasions, while 25(OH)D level was 19.6 ng/ml. Ultrasound neck was suggestive of parathyroid adenoma (right inferior parathyroid 1.3 cm × 2.8 cm), and the Sestamibi scan confirmed parathyroid adenoma (Fig. 1). Mother has undergone surgery for the same and presently doing well. Baby is asymptomatic after one year of follow up with no further episodes of any hypocalcemia.
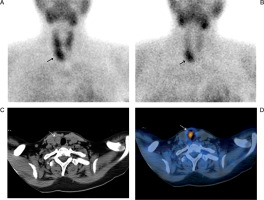
Figure 1
Early 10 min Tc99m MIBI (A) and delayed 1 hour Tc99m MIBI images (B) show an area of increased tracer activity below the lower pole of right lobe of thyroid gland (solid arrows) with wash out of tracer activity from the thyroid gland in the delayed 1 hour images. Transaxial CT (C) and fused SPECT/CT images (D) localizes the increased tracer activity to a soft tissue lesion below the right lobe of thyroid gland (dashed arrows) consistent with parathyroid adenoma
Increased serum calcium levels in mother due to unrecognized hyperparathyroidism results in fetal hypercalcemia, which in turn leads to suppression of parathyroid gland activity in the fetus, and thereby, resulting in impaired parathyroid responsiveness to hypocalcemia after birth. This results in hypocalcemia in the newborn, which may be severe and prolonged [3, 4]. Thus, this should be kept in mind while evaluating an apparently healthy new born with late onset hypocalcemic seizures. Maternal primary hyperparathyroidism is difficult to diagnose early in pregnancy as the symptoms are usually subtle and are easily confused with physiological changes of pregnancy. Routine serum calcium and phosphorous screening in pregnancy and maternal investigations for neonatal hypocalcemia may unmask maternal diagnosis and save mother and baby from complications [4–7].
to the neonatal illnesses in a preterm baby, which may lead to suppression of the HPT axis. Considering the importance of early therapy for congenital hypothyroidism, it is essential that all preterms are retested at term or prior to discharge, irrespective of the initial thyroid screening results.
The Indian Society of Paediatric and Adolescent Endocrinology (ISPAE) have given guidelines for management of congenital hypothyroidism [22, 23]. However, we used the guidelines recommended by the ESPE [19] because these babies were managed prior to the publishing of the ISPAE guidelines. We attempted to reanalyse our data using the ISPAE guidelines: 7 babies were screen negative, as per the ISPAE guidelines, and screen positive as per the ESPE guidelines. Of these 7 babies, 3 were found to have congenital hypothyroidism at term. None of the babies with congenital hypothyroidism identified as preterm would have been missed as per either guidelines. Thus, the new ISPAE guidelines may be more cost effective in developing countries like ours.
Our study is not without limitations. Aetiological profiling was possible in 8 out of 15 cases due to logistic issues. However, the remaining 7 babies are being followed up in the thyroid clinic. We are planning a trial off therapy at 3 years of age and aetiological assessment in these infants at 3 years of age. Also, the small sample size in our study is a significant limitation.
To summarise, we have described the experience of our unit in screening and evaluating preterm babies for congenital hypothyroidism. We observed a high prevalence (1 in 77). A need for repeat venous testing, irrespective of initial screening, and significant permanent congenital hypothyroidism (50%) were observed in our series.








