Introduction
Although often referred to as the ‘silent killer’ due to its asymptomatic early presentation, pancreatic cancer is associated with pain in approximately 75% of the patients at the time of diagnosis, and as much as 90% at the advanced stage [1]. Such pain is probably mediated through the celiac plexus [2] composed of 2 groups of ganglia located anteriorly and laterally to the aorta at the level of Th12 to L2 [3] vertebrae, which transmit the nociceptive input through splanchnic nerves and Th5-Th12 spinal nerve roots, and further through spinothalamic tracts to higher centres in the central nervous system [4].
The pancreatic ductal carcinoma, which constitutes up to 95% of pancreatic cancers, has one of the highest incidences of perineural invasion among cancers (80–100%) [5], subsequently generating visceral pain with neuropathic component [6, 7]. Although relatively effective, the appropriate use of analgesics and co-analgesics according to the European Society for Medical Oncology guidelines [8] is not always sufficient for optimal pain control [5] and is often associated with adverse effects related mostly to opioids [9]. Persistent pain not only significantly affects patients’ quality of life (QoL) [10] but also induces functional and structural changes at the peripheral, spinal, and supraspinal level of the nervous system, further facilitating and exacerbating the cancer-related pain through peripheral and central sensitization [11, 12].
According to the review by Vissers et al. [13], celiac plexus block (CPB), a transient interruption of pain transmission through local anaesthetic injection, or celiac plexus neurolysis (CPN), a prolonged interruption through chemical ablation with alcohol [14], were found to significantly reduce pain intensity and opioid consumption compared to systemic analgesic therapy. However, these invasive methods do not remove the need for opioids and do not significantly improve the patients’ QoL [15]. In this case report, we explain the methodology and present the outcomes of the first patient in Europe to be treated with non-invasive celiac plexus radiosurgery, a novel approach towards combating visceral pain in patients with advanced pancreatic cancer.
Patient medical history
A 61-year-old male patient with advanced unresectable pancreatic cancer was referred by an anaesthesiologist to the radiotherapy (RT) department due to severe, debilitating pain, despite appropriate pharmacotherapy, and contraindications to CPB/CPN. The patient had had a pancreatic adenocarcinoma diagnosed 2 years earlier due to significant weight loss and abdominal pain. The tumour was found to be unresectable due to mesenteric vein and artery involvement. Gastroenteroanastomosis and cholecystoenterostomy were performed due to biliary and gastrointestinal obstruction, and the patient was referred for systemic treatment. The patient has been receiving chemotherapy consisting of FOLFOX, FOLFIRI, and once again FOLFIRI (ongoing) with 4- and 3-month breaks in between. Additionally, irreversible electroporation with calcium ions and chemotherapy, a novel modality of treatment in advanced pancreatic carcinoma [16], was performed less than a year after the primary diagnosis. At the time of registration, the patient had marker progression (CA 19 – 9 = 2143 U/mL), and a mildly enhancing lesion in the head of the pancreas described as a minor residual tumour on computed tomography (CT). The patient had never received RT before.
Baseline
The patient reported severe and stabbing pain, spreading from the upper abdomen to the lower back in a belt-like distribution. The general condition was modest, with an Eastern Cooperative Oncology Group (ECOG) performance status of 2, and palliative performance scale (PPSv2) [17] of 70%. The initial brief pain inventory – short form questionnaire (BPI-SF) yielded an average 6/10 pain intensity, which ranged from 3/10 to 8/10, despite proper use of analgesics. The remaining BPI-SF parameters (Table 1) and functional assessment of cancer therapy – hepatobiliary cancer questionnaire confirmed a significant impact of pain on QoL. At the time of registration, the patient was receiving a substantial amount of analgesics, including 75 µg/h transdermal fentanyl patch every 3 days, 20 mg immediate-release oral morphine twice a day along with an average of four doses of 100 µg intranasal fentanyl daily, 1g of metamizole thrice a day, and 150 mg of pregabalin twice a day. Due to the exceptionally long medical history of advanced, unresectable pancreatic cancer, low disease volume on CT, relatively young age, and no history of prior RT, we decided to initiate the treatment process with further diagnostics and performed a positron emission tomography computed tomography (PET-CT) scan to assess the possibility of radical radiosurgery or radio-chemotherapy. Unfortunately, the PET-CT scan revealed significant disease advancement, a metabolically active 6 x 4.8 cm mass in the head of the pancreas, and a metastatic lymph node adjacent to the descending duodenum. The patient was subsequently consulted regarding ‘Celiac Plexus Radiosurgery for Pain Management’, and, having fulfilled all inclusion and exclusion criteria, he was enrolled in the clinical trial. To account for variability in pain reporting, the patient had the assessment repeated before RT, which took place 2 weeks after the baseline assessment. The results were comparable to the initial assessment, as presented in Table 1.
Table 1
Analgesic consumption and quality of life-related brief pain inventory – short form parameters
Treatment preparation and delivery
The RT planning procedures started with the preparation of an individual positioning device (vacuum mattress – Fig. 1 A), followed by a respiratory-gated deep inspiration breath hold (DIBH) treatment planning CT. The RT planning series are performed with the use of the aforementioned positioning device, to provide the best reproducibility of the patient’s anatomy during the treatment. The DIBH technique reduces the impact of internal organs’ respiratory movement. The patient is asked to hold their breath multiple times during irradiation, and the respiratory cycle is supervised through external infrared optics monitoring a small plastic cube located on the patients’ chest, which can be seen in Figure 1 B.
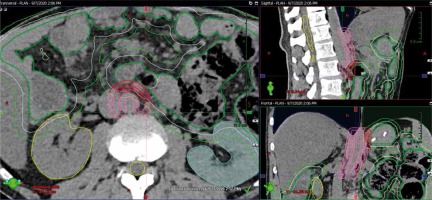
The target volume for celiac plexus radiosurgery is defined anatomically as a 5 mm wide anterolateral aspect of the abdominal aorta at the level of Th12-L2 (Fig. 2). Due to the palliative intent of the treatment, the safety of RT delivery is our highest priority, and all of the organs at risk (OAR) are carefully contoured including the distal part of the oesophagus, stomach, bowel (including duodenal sub-volume), spinal cord, liver, and kidneys. To account for bowel movement, additional 0.5 and 1 cm margins are added, and derivate OARs are taken into account during RT planning.
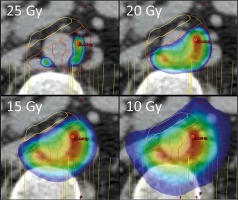
The dose prescribed to the celiac plexus was a single fraction of 25 Gy. However, the dose distribution was significantly different from the usual uniform delivery of a single dose to the whole planning treatment volume (PTV). To account for the significant radiosensitivity of the bowel adjacent to the celiac plexus, 4 separate volumes were defined and prescribed between 25 and 10 Gy (Fig. 3). This novel approach towards RT planning allows us to achieve delivery of a very high ablative dose to a relatively large volume (26.9 cc), and yet maintain high safety of the treatment.
Fig. 3
Dose distribution in the target volume, illustrating the use of dose-painting to avoid overdosing the small bowel. Colours closer to red represent a higher dose
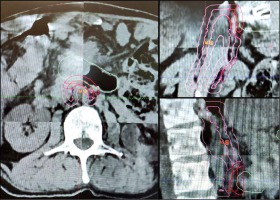
On the day of the treatment, 2 hours before RT, the patient received 8 mg of ondansetron, 8 mg of dexamethasone, and 20 mg of omeprazole orally. The initial patient’s positioning was performed using two 2D kV images, followed by a respiratory-gated cone-beam CT (CBCT). The patient’s position was aligned to the RT planning CT (Fig. 4), and irradiation was initiated. The whole therapeutic session took about 30 minutes, including positioning.
Results
Immediately after treatment, the patient experienced transient pain exacerbation of up to 8 points on an 11-point numerical rating scale (NRS), mild diarrhoea, and fatigue. The adverse effects peaked on the second day and slowly decreased. At 1 week, the patient reported persistent fatigue and mild diarrhoea, but the pain was reduced to an average NRS of 4. By the second week, the adverse effects subsided. At 3 weeks, the pain was optimally controlled (Table 1) using only a 75 µg/h transdermal fentanyl patch, and the patient was no longer using short-acting analgesics, neuropathic agents, or metamizole. The total oral morphine equivalent dose (MED) consumption of analgesics dropped by 37% to a MED of 180 mg, and performance status improved to 1 and 90% on the ECOG and PPSv2 scale, respectively. We advised further reduction of opioids, and at 6 weeks the patient was using a 50 µg/h transdermal fentanyl patch, with no significant cancer-related pain, and little effect of pain on QoL aspects (Table 1). The patient, however, reported episodes of breakthrough pain every few days, which required an application of a single dose of 100 µg of intranasal fentanyl, up to a total MED of 136 mg. He also noticed episodes of heartburn and discomfort in the right hip, which were probably unrelated to cancer. The clinical situation remained stable until the control visit at 3 months after the treatment, when the patient reported a partial relapse of symptoms and increased pain intensity up to 4/10, which required a subsequent increase of analgesic dose and use of short-acting opioids.
The patient was eventually disqualified from further systemic treatment due to deteriorating blood tests, and at 5 months after treatment a CT revealed significant disease progression. The progression led to significant pain and functional status decline. The last available information regarding the patient was obtained at 22 weeks after treatment, through a phone call with the family. The patient was spending about 23.5 hours a day in bed, had reduced fluid and food intake, and was receiving a dose of analgesics comparable to the pre-treatment period.
Discussion
To our best knowledge, in this article, we present the case report of the first patient treated for pain with celiac plexus radiosurgery in Europe. However; this method of treatment was developed at the Sheba Medical Centre, Israel. The initial proof-of-concept trial, which included 21 patients treated with either 5 x 9 Gy (9.5%) or a single fraction of 25 Gy (90.5%), resulted in pain reduction in all cases, including 7 (33.3%) patients reporting complete pain relief. The median baseline pain of 6/10 was reduced to 2.3/10 at 3 weeks, and 1.8/10 at 6 weeks (p < 0.0005) [18]. Moreover, the treatment provided a QoL improvement, including a statistically significant decrease in all domains of pain-interference [19]. As a consequence of positive results, the currently ongoing international phase II trial was initiated [20].
Celiac plexus radiosurgery can be applied both as an upfront palliative procedure or in patients refractory to CPD/CPN [21]. Considering the lack of necessity for hospitalization, the treatment can be completed within 1–2 weeks of the initial registration visit, and its availability remains uncompromised by the in-patient restrictions related to the COVID-19 pandemic.
The safety of celiac plexus radiosurgery is yet to be established in the context of re-irradiation, and for now, the previous RT to the upper abdomen is considered to be a contraindication. Nonetheless, the protocol allows for the simultaneous treatment of the primary tumour at the primary doctors’ discretion, usually to a single dose of 10–15 Gy.
Fractionated palliative RT has previously been reported to cause pain relief in advanced pancreatic cancer patients [22–25], but the data is of relatively low quality due to the retrospective nature of the reports, heterogenous study groups, and significant variability in applied RT fractionation schemes. Nevertheless, such treatment remains an option in patients who are not candidates for CPR.