Introduction
Invasive coronary angiography (CA) is the traditional imaging modality for evaluation of the coronaries and guidance of percutaneous coronary intervention (PCI) during treatment of ST-segment elevation myocardial infarction (STEMI) [1]. However, CA provides just a two-dimensional luminography of the coronaries, without additional insights into plaque morphology, extent of atherosclerosis, thrombus burden and mechanisms of vessel occlusion [2].
The use of optical coherence tomography (OCT) delivers high-resolution images that accurately depict vessel and lesion characteristics, including the presence of thrombus, provides incremental assistance in stent implantation and minimizes stent-related issues [2]. OCT may also reveal the pathophysiology of arterial thrombosis, and optimize therapeutic options during PCI, especially in young patients where mechanisms of STEMI may vary [3].
Though plaque rupture remains the most common etiology of myocardial infarction (MI), in patients younger than 40–45 years unique syndromes such as plaque erosion, coronary microvascular dysfunction, spontaneous coronary artery dissection (SCAD), myocarditis, coronary embolism, hypercoagulable state and coronary spasm (drug related or not) are prevalent [3]. Consequently, intracoronary imaging is essential to clarify the underlying mechanisms when treating young MI patients, since stenting might not be the holy grail in this subgroup. Thrombus aspiration, though not routinely advocated by the guidelines, is required in selected patients for the recovery of coronary flow during primary PCI [1]. Previous reports have highlighted that in selected young patients presenting with STEMI, lone thrombus aspiration without balloon angioplasty or stenting is feasible and is associated with favorable short and long-term outcomes [4]. Dedicated thrombectomy catheters have been developed, but one size does not fit all anatomies, with some catheters being too small to accommodate a large thrombotic burden [5].
Aim
We report the case of a young patient who presented with anterior STEMI and underwent thrombectomy of a large free-floating thrombus through a “home-made” aspiration device, assembled from a guiding extension catheter and a guide catheter. OCT showed no signs of plaque rupture, allowing deferral of stent implantation.
Case report
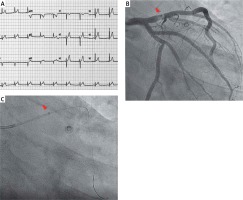
A 38-year-old man, with positive family history for coronary artery disease, presented with acute chest pain. Electrocardiography revealed hyperacute T waves in leads I, aVL, V2- > V6, corresponding with the diagnosis of an anterolateral STEMI (Figure 1 A). He was loaded with 250 mg of aspirin, 180 mg of ticagrelor and underwent emergency CA, via the right radial artery approach. CA revealed a subtotal occlusion of the proximal segment of the left anterior descending artery (LAD) with the presence of a mobile thrombus (Figure 1 B arrow) whereas the left main stem, circumflex and right coronary artery were normal. In view of the free-floating thrombus in the proximal segment of the LAD we decided to proceed with thrombus aspiration. After administration of 7500 IU of unfractionated heparin via a 6-French 3.5 extra back-up (EBU, Medtronic) guiding catheter, we tried to perform thrombosuction through a standard 6-French aspiration catheter, without success. We hypothesized that the small inner diameter (1.1 mm) of the standard aspiration catheter (Medtronic, Export) was not large enough to aspirate the large thrombus burden accumulated in the LAD.
Figure 1
A – ECG showing anterolateral STEMI. B – Angiography revealed the presence of thrombus (arrow) in the proximal LAD. C – Guide extension catheter (arrow) through the guiding catheter at the proximal LAD
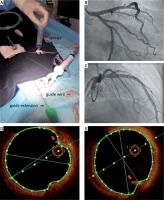
Taking into consideration the large diameter of the vessel at the lesion site (between 3.5 and 4.00 mm), the proximal location of the thrombus and the large thrombus burden, we decided to construct a wider diameter “home-made” aspiration device by using a 6-French guiding extension catheter, which has an inner diameter of 1.45 mm and would theoretically offer a greater aspiration potential. Specifically, a 6-French Guidezilla II guide extension catheter (Boston Scientific) was advanced through the EBU guiding catheter at the proximal LAD (Figure 1 C, arrow). Successful thrombectomy was performed through the “guide extension-guiding catheter” assembly via a 20 cc luer-lock vacuum aspiration syringe attached to the manifold by a three-way stopcock (Figure 2 A). Prior to any further manipulation, vigorous back-bleeding through the Y-connector was performed, to eliminate any chance of air or thrombus embolism. Intracoronary injection of tirofiban followed, to ensure adequate resolution of thrombus microparticles, resulting in an excellent angiographic result (Figures 2 B, C). OCT illustrated normal lumen area with presence of minimal thrombus remnants (Figures 2 D, E, arrow), intima thickening and fibroatheroma (Figure 2 D, asterisk) without any signs of plaque rupture at the lesion site, allowing deferral of stent implantation. Control CA after 3 days revealed a perfect result.
Figure 2
A – Guide extension-guiding catheter assembly: arrows indicate the luer-lock syringe attached to manifold, guidewire and guide extension catheter. B, C – Perfect angiographic result after thrombus aspiration. D, E – OCT illustrating presence of minimal thrombus remnants (D, E arrow), intima thickening and fibroatheroma (D, asterisk) without any signs of plaque rupture
The patient was discharged in good condition with lifelong aspirin and ticagrelor for 1 year. At 3-month follow-up he was asymptomatic with a normal echocardiogram. Further investigation excluded paradoxical emboli but revealed a positive lupus anticoagulant, for which rheumatologists advised lifelong use of aspirin.
Discussion
Restoring coronary blood flow is of utmost importance in the treatment of STEMI during primary PCI. Impaired flow and decreased coronary perfusion is related to reperfusion injury, causing arrhythmias, suboptimal microvascular circulation, contractile abnormalities, permanent myocardial dysfunction and fatal events [6, 7]. Thrombus aspiration has been proposed as an adjunct to primary PCI to further improve epicardial and myocardial perfusion by the prevention of distal embolization of thrombotic material and plaque debris [1]. Despite the fact that thrombus aspiration can improve coronary blood flow and resolve ST-segment elevation, it is not routinely advocated by the guidelines (Class IIIA) [1].
The two landmark TASTE and TOTAL trials, which investigated the role of routine manual thrombus aspiration versus conventional PCI, showed no benefit in clinical outcomes of routine aspiration overall or in the high thrombotic risk subgroup [8–10]. Additionally, increased risk of stroke was noted in the TOTAL trial [9, 10]. Similarly, in a meta-analysis of 17 trials, aspiration thrombectomy was not shown to be of benefit in reducing the risk of death or reinfarction [11]. In the high-thrombus burden subgroup, the trend towards reduced cardiovascular death and increased stroke/transient ischemic attack (TIA) calls for further research and use of improved thrombus aspiration technologies in this high-risk subgroup [12].
However, in patients with heavy thrombotic burden, thrombus aspiration should be considered. Several randomized trials have demonstrated the effectiveness and safety of manual thrombus aspiration during primary PCI. The majority of studies in the literature conclude that thrombectomy improves TIMI flow and provides rapid resolution of ST segment deviation. The TAPAS and EXPIRA trials, along with a few meta-analyses, reported that thrombectomy improves the long-term clinical outcome of reducing cardiac death and major adverse cardiovascular events (MACE). However, these studies showed no statistical significance for the prediction of MACE [13, 14]. The EXPIRA trial illustrated a reduction of the infarct area in cardiac magnetic resonance imaging, after thrombectomy [14]. A survival benefit among patients with STEMI was suggested by the TAPAS trial [13].
In addition, STEMI is an uncommon entity in young adults and its incidence depends on the cut-off age used [15]. Most studies have used an age cut-off of 40–45 years to define young patients with STEMI [3]. It has been reported that < 1% of patients with STEMI are ≤ 35 years [15]. Young STEMI patients differ from older patients in the risk factor profile and in the extent of atheromatic plaque coronary burden. In particular, young coronary patients are characterized by a higher proportion of heavy smoking, a lower proportion of hypertension and diabetes mellitus and a relatively high proportion (15–20%) of angiographically “normal” coronary arteries [15]. Apart from the classical mechanism of plaque rupture, MI in young individuals can be attributed to plaque erosion, coronary microvascular dysfunction, SCAD, myocarditis, coronary embolism, hypercoagulable state and coronary spasm related or not to drug use (e.g. cocaine) [3]. In particular, the presence of antiphospholipid antibodies, such as the positive lupus anticoagulant detected in our patient, is a known risk factor for arterial and vein thrombosis by causing a hypercoagulable state [3]. Therefore, diverse underlying mechanisms may be present in young patients with MI, requiring tailored interventional and drug therapy. The use of OCT is essential to reveal the underlying pathophysiology, and optimize therapeutic options [2]. In our patient OCT showed no signs of plaque rupture, allowing deferral of stent implantation after adequate thrombus aspiration.
From a practical point of view, coronary diameter varies between individuals, and commercially available thrombectomy catheters may not be suitable for certain anatomies to achieve sufficient thrombus aspiration (Table I A). Guiding extension catheters have a larger inner diameter and can offer a greater aspiration potential (Table I B). The assembly and use of a home-made aspiration catheter has already been described above. Of note, this technique should only be used if the coronary artery is large enough to accommodate the assembly, since the guide extension catheter can dissect the coronaries and only when the guiding catheter has perfectly engaged the coronary artery to avoid dislocation of thrombus to the systematic circulation during manipulations.
Table I
Characteristics of commonly used thrombus aspiration (A) and guide extension catheters (B)
Conclusions
Diverse underlying mechanisms may be present in young patients with MI, requiring a case-by-case approach. The use of intracoronary imaging is essential to elucidate the diagnosis and optimize therapeutic options. In selected young patients with MI, OCT-guided lone thrombus aspiration without balloon angioplasty or stenting, either by conventional means or home-made aspiration catheters, can be a feasible and effective therapeutic approach.







