Summary
Transcatheter aortic valve replacement (TAVR) appears to be safe when performed with a concomitant mitral valve regurgitation (MR) and is associated with lower post-procedural stroke risk. Further studies are needed to confirm these findings and to define the best treatment option when there is concomitant severe aortic stenosis and significant MR in non-high surgical risk patients. Furthermore, whether MR improvement post-TAVR depends on the type of the MR, primary versus secondary, needs to be further evaluated.
Introduction
Aortic stenosis (AS) is the most common valvular disease in elderly people [1], and its prevalence is increasing up to 9.8% in the 80–89-year old population [2]. Transcatheter aortic valve replacement (TAVR) has become the recommended treatment for severe symptomatic AS patients with high or prohibitive surgical risk [3, 4].
Mitral regurgitation (MR) is the most common type of moderate or severe heart valve disease among U.S. adults older than 55 years of age. Its prevalence increases further with age as well [1]. Irrespective of the cause and mechanism, moderate or severe MR is associated with cardiac dysfunction and increased mortality [1, 5]. Frequently, patients with severe aortic stenosis have concomitant MR; therefore, the impact of MR on patients undergoing TAVR is important [6].
Aim
This study was conducted to compare in-hospital outcomes of concomitant MR in patients undergoing TAVR.
Material and methods
Data source
The National Readmissions Data (NRD) is part of the Healthcare Cost and Utilization Project (HCUP) databases which developed through a Federal-State-Industry partnership and is sponsored by the Agency for Healthcare Research and Quality (AHRQ). The HCUP databases include the largest collection of de-identified longitudinal hospital care data in the United States from 35 states, with all-payer and encounter-level information. The NRD is a unique data subset designed to support various types of analyses including readmission rates with safeguards to protect the privacy of individual patients, physicians, and hospitals. It contains discharges from all hospitals provided by HCUP Partners from over 20 states. It contains more than a hundred clinical and nonclinical variables for each hospital stay, including a unique record identifier that can be used to combine data elements from different NRD files, a verified patient linkage number for linking hospital visits for the same patient across hospitals, International Classification of Diseases, Ninth Revision, Clinical Modification (ICD-9-CM) codes for principal and secondary procedures and diagnoses (including comorbidities and complications), age, gender, and others [7].
Study cohort
ICD-9-CM codes of TAVR were used to search the principal or secondary procedure fields to identify discharges who underwent TAVR during the index hospitalization. Subsequently, ICD-9-CM codes of MR were used to identify the TAVR subgroup with MR (TAVR-MR). MR included isolated MR and combined MR with mitral stenosis/aortic valve disorder regardless of the cause. Unspecified mitral valve disorders were excluded. The NRD excludes rehabilitation and long-term acute care hospitals because of inconsistent data capture from these facilities and their special patient population that has longer stays. It also excludes discharges from patients with missed age, missed or questionable linkage numbers or from hospitals with more than 50% of their discharges excluded because of these criteria, because patients treated at these hospitals may not be reliably tracked over time.
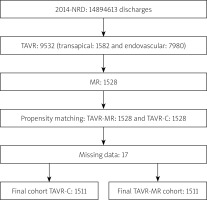
The patients’ demographics, baseline characteristics, comorbidities and the post-procedural complications were extracted using ICD-9-CM codes as well. Because the HCUP databases do not have a present-on-admission indicator, which could limit the ability to distinguish comorbid conditions from the post-procedural complications, we used the recommended algorithms developed by AHRQ to identify both the comorbid conditions and the post-procedural complications from secondary diagnoses’ fields. Comorbid conditions were identified using the Medicare diagnosis related groups and secondary diagnosis codes of comorbidity indexes such as Deyo’s modification of Charlson’s comorbidity index and the Elixhauser comorbidity index, which are specific to comorbid conditions. The post-procedural complications were identified using the patient’s safety indicators version 4.4, March 2012, which were established by AHRQ to monitor preventable adverse events during hospitalization, and using the secondary codes that are specific to the post-procedural complications (Tables I, II) [8–11]. We followed all HCUP recommendations and standards to use HCUP databases highlighted by Khera et al. (Figure 1) [12].
Table I
International Classification of Diseases, Ninth Revision, Clinical Modification (ICD-9-CM) codes used to extract comorbid conditions from the National Readmissions Data (NRD) database
Table II
ICD-9-CM codes used to extract post-procedural complications from the NRD database
Study outcomes
Study outcomes included in-hospital all-cause mortality, in-hospital post-procedural complications of stroke, acute myocardial infarction (AMI), bleeding, mechanical complications of prosthetic valve, vascular complications (VC), need for new permanent pacemaker (PPM) implantation and 30-day readmission rates. The 2014 NRD reports in-hospital all-cause deaths during the index hospitalization. Based on the ICD-9 definition, post-procedural stroke was defined as a new post-procedural neurological deficit(s) or transient ischemic attacks. AMI included new post-procedural subendocardial or transmural myocardial infarction. Bleeding included post-procedural bleeding, anemia, or blood or blood product transfusion. Mechanical complications of the prosthetic valve included paravalvular leak, valve malposition, embolization, dislodgement or malfunction. Vascular complications included accidental puncture or injury of blood vessels or retroperitoneum, arteriovenous fistula formation, VC requiring surgery, and other VC. Permanent pacemaker defined as new post-procedural insertion of permanent pacemaker device excluding defibrillators, and temporary and previously inserted pacemaker (Table II).
The 30-day readmission rate was calculated based on HCUP recommendations. We identified all-cause (including planned admission, injury, trauma, etc.) all readmissions (including first and subsequent admissions) within the specified period (i.e. 30 days) to any hospital within the same state (as cross-state readmissions cannot be tracked by the NRD dataset). Transfers were not considered readmissions. We excluded TAVR patients who died during the index hospitalization, age less than 18 years, missing LOS or if the TAVR procedure was performed in the month of December as the readmission rate cannot be calculated in 2 different years [13].
Statistical analysis
Statistical Analysis System (SAS) software 9.4 (TS1M4, SAS Institute Inc, Cary, North Carolina) was used for data extraction, propensity score matching and statistical analysis. Statistical analysis was performed on the unweighted (i.e. actual) number of discharges rather than the weighted (i.e. the national estimates) discharges, which is roughly equal to 2.2 times the unweighted numbers [7]. Logistic regression was used to create the propensity score, based on basic demographics and baseline characteristics (Table III) for a one-to-one parallel, balanced propensity score matching model using a caliper of 0.001. The McNemar test was used to compare paired categorical variables of the baseline characteristics and outcomes while the paired-samples t-test was used to compare continuous variables. Multivariable logistic and linear regression models were used to identify predictors of post-procedural stroke in the TAVR-MR by calculating adjusted odds ratios (OR) and 95% confidence intervals (95% CI) for baseline characteristics and common comorbidities (Table III) [14, 15]. A p-value of < 0.05 was used for statistical significance.
Table III
Variables used in univariable and multivariable logistic and linear regression models for propensity matching and adjusted analysis to determine predictors of core post-TAVR stroke
Results
The 2014 NRD has 14.9 million unweighted (36 million weighted) discharges; there were 9,532 unweighted TAVR, 1,528 of which had a diagnosis of MR. After excluding missing data, each group included 1,511 patients (Figure 1). Average age was 81.7 years and 49.3% were male. Both groups were comparable in terms of basic demographics, baseline characteristics, anticoagulation status, TAVR access site and common comorbidities. Coronary artery disease, hypertension, hyperlipidemia, heart failure and atrial fibrillation were the most common comorbidities. Combined MR and mitral stenosis was extremely rare, 0.3% of TAVR-MR. More than 80% of TAVRs were performed via an endovascular approach, and less than 20% were transapical. The two groups were comparable in terms of number of active diagnoses and chronic conditions documented in the chart during indexed hospitalization (Table IV).
Table IV
Demographics, baseline characteristics and comorbidities of transcatheter aortic valve replacement with mitral regurgitation (TAVR-MR) group and control (TAVR-C) group with p-values for each variable. A p-value < 0.05 indicates a statistically significant difference between groups
| Parameter | TAVR-MR (%) | TAVR-C (%) | P-value |
|---|---|---|---|
| Number of patients | 1511 | 1511 | – |
| Mean age [years] | 82.0 | 81.4 | 0.08 |
| Female | 50.6 | 50.8 | 0.88 |
| Transapical TAVR | 16.3 | 17.8 | 0.26 |
| Endovascular TAVR | 84.1 | 82.3 | 0.19 |
| Mitral stenosis | 0.3 | 0.3 | 1.00 |
| Chronic coronary artery disease | 69.2 | 69.4 | 0.87 |
| Chronic kidney disease | 40.3 | 39.9 | 0.82 |
| Hypertension | 79.4 | 78.6 | 0.59 |
| Diabetes mellitus | 37.5 | 38.2 | 0.71 |
| Hyperlipidemia | 63.7 | 62.1 | 0.38 |
| Heart failure (systolic or diastolic) | 81.6 | 80.6 | 0.46 |
| Systolic heart failure | 35.9 | 36.1 | 0.93 |
| Atrial fibrillation | 50.8 | 52.5 | 0.35 |
| Atrial flutter | 6.3 | 7.1 | 0.38 |
| Long-term anticoagulation | 18.5 | 19.3 | 0.57 |
| Abnormal coagulation profile* | 1.1 | 1.1 | 0.86 |
| Peripheral vascular disease | 30.6 | 30.6 | 0.97 |
| Carotid artery stenosis | 7.9 | 8.3 | 0.69 |
| Chronic obstructive pulmonary disease | 25.9 | 26.3 | 0.80 |
| Mean number of diagnoses in chart | 19.6 | 19.6 | 0.59 |
| Mean number of chronic conditions | 10.1 | 10.1 | 0.71 |
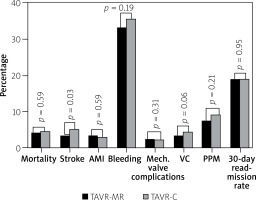
TAVR-MR was associated with lower risk of post-procedural stroke than TAVR-C (3.5% vs. 5.2%, p = 0.03). There was no significant difference between groups in terms of in-hospital all-cause mortality (4.1% vs. 4.5%, p = 0.59), AMI (3.2% vs. 2.9%, p = 0.59), bleeding (33.4% vs. 35.6%, p = 0.19), mechanical complications of prosthetic valve (2.5% vs. 1.9%, p = 0.31), VC (3.2% vs. 4.4%, p = 0.06), PPM (7.9% vs. 9.1%, p = 0.21) or 30-day readmission rates (19.0% vs. 19.1%, p = 0.95) (Table V, Figure 2). Multivariable regression models for the TAVR-MR group showed that the number of active diagnoses was the only predictor of post-procedural stroke (OR = 1.081, 95% CI: 1.010–1.56, p = 0.02) (Table III).
Table V
Study outcomes results with p-values for each outcome. A p-value < 0.05 indicates a statistically significant outcome
Discussion
This study shows that TAVR-MR was associated with lower risk of in-hospital post-procedural stroke in comparison to a propensity-matched TAVR-C. There was no significant difference between the two groups in terms of in-hospital mortality or other post-procedural complications, or 30-day readmission rates.
The incidence of post-TAVR stroke was estimated to be 5.4% at 30 days in the Placement of AoRtic TraNscathetER Valves (PARTNER) trial, which was comparable to our control group of 5.2%, and associated with worse outcomes and increased mortality [16, 17]. Post-TAVR stroke is classified into early (up to 2 days post-procedure), delayed (2 to 30 days) or late (30 days post-TAVR).
Coexisting moderate to severe MR in patients undergoing TAVR is common, and has been reported in 19–48% [17–20]. The prevalence of MR in TAVR patients in this cohort was 15.6%. Despite the fact that the presence of MR was not evaluated as a risk factor for post-TAVR stroke in a subgroup analysis of the PARTNER trial [20], there are existing data in the literature linking mitral regurgitation to a lower post-TAVR stroke with incidence of 1.5–3.3% [6, 20].
Kleiman et al. noted that 20.1% of patients who did not have a stroke within the first ten days post-TAVR had more than moderate MR [21]. Additionally, Little et al. had no reported major stroke between 30-day and one-year post-TAVR in a population of 53 patients with severe MR [22]. This may suggest a probable stroke-protective effect with coexistence of MR. Furthermore, some studies have suggested that the incidence of post-TAVR stroke could be inversely related to MR severity [23]. Our study revealed post-TAVR stroke during the index hospitalization (i.e. which represents early and delayed post-TAVR stokes) in 3.5% of TAVR-MR.
Although MR has been described as an independent predictor of mortality in many heart diseases, it has been linked with lower stroke risk in atrial fibrillation patients [24]. Evidence suggests a reduced thromboembolic event rate in MR versus mitral stenosis patients with or without cardiac arrhythmias [25]. The prevalence of left atrial thrombus in patients undergoing transthoracic or transesophageal echocardiography seems to have an inverse relation to the severity of mitral regurgitation, and it has been hypothesized that mitral regurgitant jet reduces blood stasis and hypercoagulable status in the left atrium of patients with atrial fibrillation [24, 26, 27]. The same hypothesis might be applicable to TAVR-MR patients even with the absence of atrial fibrillation, probably by decreasing the blood stasis in the straining left ventricle, which is facing a very high afterload secondary to severe AS. Similarly, aortic regurgitation has been associated with lower post-TAVR stroke [20], which further supports the probable stroke-protective effect of regurgitant valve disorders by preventing blood stagnation.
The relief of aortic valve obstruction by TAVR, and subsequently left ventricle afterload reduction, has been associated with an improvement in MR severity. The vast majority of MR in TAVR patients is functional MR, and improvement was noted in 47% of those with severe MR and 35% of those with moderate MR at 1 year [28]. Furthermore, the improvement in MR was associated with improvement in quality of life [22]. While the literature reports improvement in the majority of patients with functional MR who undergo surgical aortic valve replacement, it suggests similar or even worse post-surgical echocardiographic findings in patients with concomitant primary MR. However, there are no data to support such a correlation in TAVR patients with concomitant primary mitral valve disease.
There are conflicting data in the literature on the effect of concomitant severe MR on TAVR mortality. In our study there was no significant difference in in-hospital mortality between the two groups, which is consistent with the current literature. This could be explained by the left ventricular remodeling after TAVR that occurs in MR patients [29, 30]. Conversely, higher in-hospital mortality with moderate and severe MR patients undergoing TAVR has also been reported [6, 23].
The current study has important clinical implications. It represents real-world experience and outcomes in the United States and it adds more evidence to the current literature suggesting that TAVR is safe when there is a concomitant MR which might have a stroke-protective effect. The current guidelines recommend mitral valve repair or replacement for moderate or severe MR during cardiac surgery for other reasons [3]. This study raises the question of incorporating MR in the available operative risk assessment scoring systems when evaluating the stroke and overall risk. This is especially important because of the known added risk, higher mortality, and higher incidence of major complications when performing combined aortic and mitral valve surgery compared to isolated aortic valve surgery [31].
Limitations
This is a retrospective study. Although the two groups were comparable in baseline characteristics, heterogeneity and other confounders remain a concern in any large database. This could include the presence of other stroke risk factors such as cerebral vascular disease. The NRD database does not provide information about cross-state readmissions, TAVR systems used nor their type/generation. It also does not provide information regarding the severity, cause, type of MR (i.e. primary versus secondary), postprocedural MR reduction, or the nature or severity of stroke. Adjudication to these could not be performed. Furthermore, outcomes were defined based on ICD-9-CM codes rather than Valve Academic Research Consortium (VARC) criteria.
Conclusions
Transcatheter aortic valve replacement appears to be safe when performed with a concomitant MR and is associated with lower post-procedural stroke risk. Further studies are needed to confirm these findings and to define the best treatment option when there is concomitant severe AS and significant MR in non-high surgical risk patients. Furthermore, whether post-TAVR MR improvement depends on the type of the MR, primary versus secondary, needs to be further evaluated.