Introduction
The majority of malignant tumours that cause bile duct obstruction, particularly primary hepatobiliary, pancreatic or papillary cancer, as well as advanced gastrointestinal cancer, are inoperable at the time of diagnosis. Malignant biliary obstruction results in pruritus, pain, jaundice, and cholangitis, often decreasing the quality of life. Furthermore, as these tumours usually occur in the elderly population, the advanced surgery required for radical management is often impossible. The main objective in these patients is the alleviation of symptoms and amelioration of the quality of life. Therefore, effective drainage of bile is crucial in these cases. When treating patients with obstructive jaundice by drainage methods, the usual order of methods is endoscopic retrograde cholangiopancreatography (ERCP), then endoscopic ultrasound-guided biliary drainage (EUS-BD) and only then percutaneous transhepatic biliary drainage (PTBD) [1-7].
Percutaneous transhepatic biliary drainage is effective and successfully leads to biliary decompression, with easy monitoring and catheter manipulation in the case of tube dysfunction or drainage problems. However, cholangitis, bile peritonitis due to bile leakage, bleeding, pneumothorax, liver abscesses, patient discomfort related to the catheter, and body fluid loss with electrolyte imbalance are also possible [3, 5].
Currently, there is insufficient evidence to support or refute routine biliary drainage (BD) for patients with advanced malignant biliary obstruction [5, 8]. Some reports revealed that BD was effective for treating symptoms [9, 10]. On the other hand, in some cases liver function did not improve after BD and the prognosis was poor [10, 11]. In the absence of a strong body of evidence, selection of patients for PTBD depends on the clinician’s or hospital’s preferences. So, in our hospital, PTBD is performed in cases of biliary obstruction after failed ERCP. We are often in doubt as to whether to recommend PTBD or opt for best supportive care for patients with biliary obstruction with advanced inoperable malignant tumours. We reviewed our experiences with PTBD in patients with advanced inoperable malignancy and analysed factors that affected clinical outcomes after procedures, with the objective to differentiate between patients who would or would not receive benefits from PTBD.
Material and methods
A retrospective analysis was performed on all patients who underwent PTBD at our hospital due to inoperable, malignant, symptomatic, biliary obstruction. Cases were identified by searching the hospital database of all patients with the diagnosis of malignant biliary obstruction during the study period. Patients were enrolled if managed using PTBD for obstructive jaundice due to advanced inoperable malignant disease confirmed by clinical and imaging findings. An uncorrectable coagulation disorder was the only absolute contraindication for treatment. Written informed consent was obtained from all patients before PTBD. Board approval was obtained by the hospital’s Scientific and Ethics Committee.
Baseline pre-procedural laboratory tests including blood count, serum bilirubin, renal function and coagulation profile (international normalized ratio – INR, prothrombin time, partial thromboplastin time) were performed. Any clotting disorder was corrected appropriately. Computed tomography and/or sonography evaluation of the lesion and the dilatation status of the biliary tree were performed prior to the procedure.
PTBD was performed under ultrasound guidance with careful localization of the biliary tree and proper selection of the entry site. The procedure was performed using local anaesthesia. No conscious sedation was applied. The route for BD depended on the location and available ‘window’ for optimal access to the biliary tree. The access site (right or left lobe puncture) was chosen according to the pre-procedural imaging. Generally, the most commonly used method was the trocar technique using an 8F multisidehole catheter (Boston Scientific, Boston, USA), guiding it into the bile duct (Fig. 1). If the introduction of an 8F catheter into the bile duct was difficult or impossible due to the position or the diameter of the bile duct, a modified trocar method using a multisidehole 5F catheter (Boston Scientific, Boston, USA) was used instead. With this technique, a 14 gauge needle was first introduced into the bile duct. Subsequently, a 5 F catheter was introduced through the needle into the bile duct. After introducing the catheter and the establishment of BD through the catheter, the needle was removed (Fig. 2). The catheter was then secured to the skin and connected to a bag attached to the skin for continuous PTBD. In cases which required introduction of more than one catheter in order to improve BD, additional catheters were introduced by the same method in other selected bile ducts. If a catheter problem occurred, it was resolved by repositioning and flushing of the catheter or by introducing a new one.
Fig. 1
Percutaneous transhepatic biliary drainage performed using an 8F catheter. A) 8F catheter introduced into the bile duct. B) Checking for catheter patency by injecting contrast through the catheter into bile ducts during follow-up cholangiography
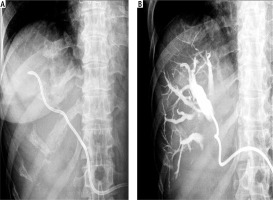
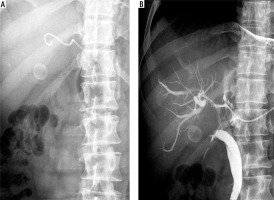
Fig. 2
Percutaneous transhepatic biliary drainage performed using a 5F catheter. A) 5F catheter introduced into the bile duct. B) Checking for catheter patency by injecting contrast through the catheter into bile ducts during follow-up cholangiography
The number and size of the catheters were determined individually for each patient by the clinicians and interventional ultrasonographists. Their decisions were based upon clinical considerations, the size and location of the bile duct and its relationship to the surrounding vascular structures. Colour Doppler sonography was performed prior to intervention in order to determine the catheter pathway through the liver tissue towards the targeted bile duct, avoiding vascular structure damage. In general, colour Doppler sonography was not used during the intervention as it was more difficult to follow the catheter tip passing through the liver tissue. Adequate drainage is usually confirmed by a steady decline in serum bilirubin concentrations. Catheter patency was checked by injecting contrast and observing its passage through the catheter (Figs. 1, 2).
The study’s primary endpoints were significant bilirubin level decrease and survival rates. Secondary endpoints included periprocedural major and minor complication rates, primary patency rates (defined as the patency of the deployed catheters without any additional intervention), and secondary patency rates (defined as the catheter patency following an additional re-intervention due to clinical relapse of the symptoms). Catheters were considered patent in cases in which no increase in serum bilirubin levels or dilatation of the intrahepatic ducts was noted. Catheter patency was defined as the duration from the insertion of the catheter until the date of failure. Catheter failure was defined as persistent or recurrent jaundice. Complications were divided into major and minor according to the standards of the Society of Interventional Radiology [12].
Statistical analysis was performed using MedCalc software version 15, Mariakerke, Belgium. Descriptive statistics were presented as means ± SD and medians (IQ ranges) for normal and skewed data distributions, respectively. Discrete variables were presented as proportions. The Wilcoxon signed-rank test was used to test the skewed distributed paired samples. Patients’ survival rates were calculated using the log-rank test and a Cox proportional hazard regression model was built to assess the risk factors for mortality. P values with < 0.05 taken as the level of statistical significance were reported.
Results
There were 108 patients with malignant biliary obstruction who were admitted to our hospital and who underwent PTBD during the study period. All patients suffered from advanced inoperable malignant disease and were considered unsuitable for surgery due to tumour extension and/or poor physical conditions. Pancreatic carcinoma (n = 34), cholangiocarcinoma (n = 20), and hepatocellular carcinoma (n = 20) were the most frequent underlying malignant diseases (in about 70% of cases). The post-procedural, median (IQR) hospitalization period was 9 (4-17) days. Patient baseline demographic, clinical and laboratory data are reported in Table 1.
Table 1
Baseline characteristics of 108 patients before percutaneous transhepatic biliary drainage
Percutaneous transhepatic biliary drainage was technically successful and bile ducts were successfully drained in all 108 patients. Follow-up cholangiography 3-5 days after catheter placement showed adequate decompression of the biliary ducts in all patients. Procedural details and outcomes are analytically reported in Table 2. Follow-up was done in all patients and serum bilirubin levels were recorded prior to and following the intervention. The median (IQR) serum bilirubin level, which was 282 (171-376) µmol/l before drainage, decreased significantly, to 80 (56-144) µmol/l, 15 days after catheter placement (p < 0.001). The pre- and post-procedure decrease in bilirubin levels was statistically significant (p < 0.001).
Table 2
Procedural details and outcomes
By the end of the study, 100 patients had succumbed to various conditions and 8 patients were still alive. Patient survival ranged from 3 to 597 days and the median (IQR) survival time following PTBD was 168 days (90-302). The 1, 3, 6, 12 and 18-month survival rates were 96.3%, 75.9%, 48.1%, 8.3% and 1.9%, respectively (Table 2, Fig. 3). Multivariate analysis revealed that liver metastases and alkaline phosphatase (ALP) were significantly associated with mortality (Table 3).
Fig. 3
Survival rates of patients with inoperable, malignant biliary obstruction undergoing percutaneous biliary drainage for obstructive jaundice
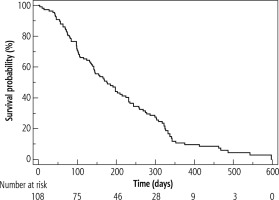
Table 3
Risk factors for mortality in patients with inoperable, malignant biliary obstruction undergoing percutaneous biliary drainage for obstructive jaundice
The overall complication rate was 9.3% (10/108 patients). Major complications occurred in 6 patients (5.6%). Two patients had acute cholecystitis and they underwent percutaneous cholecystostomy. Two patients had sepsis and cholangitis, which was resolved within 7 days by antibiotic treatment alone. One patient suffered from pancreatitis and received conservative therapy. The remaining patient exhibited symptoms of pneumonia and pleural infection which were successfully treated by conservative therapy. Procedure-related minor complications were observed in 4/108 patients (3.7%) with self-limiting haemorrhagic cholangitis (1 patients) and self-limiting haemobilia (3 patients) which were successfully managed using conservative antibiotic therapy and completely resolved within 3 days without transfusion.
A total of 4 patients succumbed to their conditions within 30 days of receiving PTBD. However, they were not included in the data for major complications as the cause of mortality since 2 patients succumbed to invasive primary tumour progression, one patient died due to heart failure and the fourth patient died 3 days after the procedure due to aspiration. Therefore, none are classified as drainage procedure-related complications.
Discussion
We reported our long-term experience (15 years) in PTBD of 108 patients with obstructive jaundice due to advanced inoperable malignant disease. Considering that our study population represented a seriously ill subset of patients, the median survival time after the complete period of follow-up, the 1, 3, 6, 12 and 18-month survival rates and the overall complication rates (Table 2) confirmed PTBD as safe and effective management of obstructive jaundice in patients with advanced inoperable malignant disease who are unsuitable for surgery due to tumour extension and/or poor physical conditions.
Percutaneous transhepatic biliary drainage is an established and well-reported method used to relieve jaundice with clinical success rates ranging between 75% and 98% in various reports [10, 13-16]. The advanced inoperable malignant disease has been identified as an independent poor prognostic factor and predictor of increased mortality following PTBD [10, 14, 17]. In our series, the clinical success rate was achieved and liver dysfunction due to obstructive jaundice improved after PTBD in 82.4% of cases. Considering the clinical success rate, the median survival time, and the 1, 3, 6, 12 and 18-month survival rates (Table 2), our results are comparable to previously reported results involving patients with less advanced, ECOG I-IV, disease [10, 14, 16].
Our results suggest (Table 3) that patients with obstructive jaundice, with the presence of liver metastases and elevation in ALP as the poor prognostic factors, could not receive survival benefit from PTBD even though they achieved an initial improvement of their hepatic dysfunction after PTBD. However, patient age, gender, serum bili-rubin, aminotransferases, γ-glutamyltransferase (GGT), CEA, albumin levels prior to intervention, and obstruction levels were generally not found to be associated with mortality (Table 3). Several studies showed that the presence of liver metastases was accompanied by a lower success rate because of poor liver reserve and advanced systemic disease [10, 16, 18]. In Kasuga et al.’s [10] study poor performance status, presence of ascites and high level of CA19-9 were independently associated with a poor prognosis. However, the elevated level of ALP was not associated with a poor prognosis in this study. The complication rate of PTBD in previous studies ranges between 7.8% and 42% and the 30-day mortality rate ranges between 0.9% and 19.8% [16, 18-20]. The overall complica-tion rate (9.3%) and in-hospital mortality rate (3.7%) in our series are comparable to or better than those reported in previous studies.
In our study, PTBD was technically successful in all patients. The majority of the procedures were performed with a single right-sided access, following the pre-procedural imaging assessment (Table 2). However, when lesions involved both hepatic ducts we decided to drain both hepatic lobes: using two stents in 16/108 (14.8%), and three stents in 4/108 (3.7%) patients, with the intention to improve palliation outcomes (Table 2).During the follow-up period biliary reobstruction due to catheter occlusion (mainly caused by tumour ingrowth, with or without tumour overgrowth) occurred in 25 (23.1%) cases. Considering the median primary and secondary patency, the overall biliary reobstruction rates and the overall complication rates (Table 2), our results are comparable to previously reported results [10, 14, 16]. Moreover, PTBD was not interrupted due to catheter problems in any of the patients and usually lasted until the end of the patient’s life. PTBD was discontinued earlier in only a few patients, because it was estimated that there was a terminal liver failure due to which PTBD became ineffective.
Several authors consider that endoscopic stenting of biliary obstruction should be the preferred palliative modality because it is less invasive compared to surgery and more comfortable compared to the percutaneous approach. However, there are patients in whom ERCP fails because of unsuccessful biliary cannulation (especially in patients with obstructions close to or above the bile duct bifurcation), or inaccessible papilla due to duodenal stenosis caused by tumour invasion. In those cases, some authors consider that EUS-BD should be performed as a more physiological and more comfortable alternative to PTBD [3, 4]. The major advantage of the endoscopic approach is that it creates a permanent biliary-gastrointestinal tract with no spillage of bile content, in contrast to PTBD, thereby causing beneficial effects on restoring nutritional status and immune function and reducing endotoxemia [3, 4, 11]. However, stent malfunction due to its clogging after EUS-BD is not uncommon. In the case of catheter dysfunction and drainage problems the monitoring and catheter manipulation are very difficult or impossible by endoscope, unlike with the PTBD approach. Moreover, the results of several studies (2 meta-analyses) indicate that PTBD had a higher therapeutic success rate than EUS-BD in the treatment of malignancy-induced biliary obstruction [21-27].
There are certain limitations to our study such as the lack of a predefined clinical and imaging follow-up due to the retrospective design without a control group and with a series of patients who were treated over a long period of time, as well as the inability to obtain detailed data regarding the patients’ post-procedural quality of life using dedicated questionnaires. Nevertheless, we believe that our study has significance because it presents our long term experience in the treatment of a critically ill subset of patients with obstructive jaundice caused by advanced inoperable malignant disease who are unsuitable for surgery due to tumour extension and/or poor physical conditions, and for whom PTBD provides practically the only available treatment.
In conclusion, PTBD is a safe and effective way to relieve jaundice caused by advanced inoperable malignant disease in cases considered unsuitable for surgery due to tumour extension and/or poor physical conditions. Our results suggest that in patients with obstructive jaundice the presence of liver metastases and/or elevation in ALP level as poor prognostic factors were associated with poor survival. Therefore, careful patient selection is necessary when introducing PTBD in order to avoid invasive procedures in patients with a poor prognosis.






