The treatment of severe infections in intensive therapy units continues to be a challenge. This is because establishing optimum dosing regimens of antimicrobials still poses some difficulties, particularly for patients in septic shock. Septic patients are at risk of having subtherapeutic concentrations of antibiotics in the blood with a standard dosing regimen, owing to an increased volume of distribution and variable renal or hepatic clearance. Another cause of excessive decrease in antibiotic concentrations in blood may be instituting continuous renal replacement therapy (CRRT). CRRT may have a significant effect on the clearance of antimicrobials, in some cases considerably accelerating their elimination. The rate of this process depends on the surface area of the filter used, the kind of membrane, the intensity of the procedure, and the type of anticoagulation [1–4]. The first studies on pharmacokinetics of antibiotics during CRRT were conducted with filters of a relatively small surface area (< 1 m2) and with low intensity of the procedure (dialysate/substitute flow of 1–2 L h-1). At present, CRRT procedures are performed with filters of a larger surface area (min. 1.5 m2), and, if septic shock is the case, with a much greater intensity, hence the need to reassess the pharmacokinetics of antibiotics in critically ill patients during CRRT regarding their clinical efficacy.
The efficacy of antimicrobial drugs depends on whether the target pharmacokinetic/pharmacodynamic (PK/PD) indices are attained. Fluoroquinolones efficacy depends on the ratio of 24-hour area under the curve (AUC) to minimum inhibitory concentration (MIC) (AUC0–24/MIC). The target for ciprofloxacin is the ratio of AUC0–24/MIC of 30–50 for G(+) infections, over 125 for G(-) infections, and in the case of bloodstream infection ≥ 250 [5–9]. Ciprofloxacin efficacy has been also proved in Pseudomonas aureginosa infections when the peak drug concentration in blood exceeds MIC eight-fold (Cmax/MIC > 8–10) [10]. There is a scarcity of pharmacokinetic studies on ciprofloxacin dosing during CRRT, and these focus primarily on standard dosing (Table 1). In recent years there have been reports stressing the need to use higher doses of this antibiotic, particularly in obese patients; hence, pharmacokinetics must be assessed to determine the clinical efficacy with such dosing [14, 17].
TABLE 1
Collected data for ciprofloxacin used in critically ill patients in intensive care unit receiving continuous renal replacement therapy (CRRT)
| Date of publication | Author reference | n | CRRT technique | Dialysis /UF rate | Filter type surface | Ciprofloxacin dose | PK/PD target | PK/PD target attainment MIC 0.25 (µg mL-1) | PK/PD target attainment MIC 0.5 (µg mL-1) | PK/PD target attainment MIC 1 (µg mL-1) |
|---|---|---|---|---|---|---|---|---|---|---|
| 2001 | Wallis et al. [11] | 6 | CVVHDF | Dialysate 1 L h-1 and predilution 2 L h-1 | AN69HF NS | 200 mg t.i.d | AUC0–24/MIC > 125 | 33% patients | 66% patients | NA |
| 2001 | Malone et al. [12] | 10 | CVVH/CVVHDF | CVVH: UF 9–21 mL min-1 CVVHDF: dialysate 0.8–1 L h-1 UF 14–24 mL min-1 | AN69HF 0.6 m2 | 400 mg daily – 400 mg b.i.d. | AUC0–24/MIC > 125 | 100% patients | 60% patients | 10% patients |
| 2002 | Bellmann et al. [13] | 2 | CVVH | NA | Baxter PSHF 70 0.71 m2 | 400 mg b.i.d. | AUC0–24/MIC ≥ 100 | 50% patients | 0% patients | NA |
| 2010 | Utrup et al. [14] | 1 | CVVHDF | Dialysate 1000 mL h-1 UF 1250–1500 mL h-1 | AN69HF 0.9 m2 | 800 mg b.i.d. | AUC0–24/MIC > 125 and Cmax/MIC > 10 | 100% patients (185 kg) | 100% patients (185 kg) | 100% patients (185 kg) |
| 2011 | Spooner et al. [15] | 7 | CVVHDF | 1–2 L h-1 dialysate and 2 L h-1 UF | AN69HF NS | 400 mg b.i.d. | AUC0–24/MIC > 100 | NA | 71% patients | NA |
| 2014 | Scheer et al. [10] | 20 | CVVHD/ VVHDF | CVVHD: dialysate 2–2.2 L h-1 CVVHDF: dialysate 1.3–2.6 L h-1 UF 100–450 mL h-1 | Ultraflux AV 1000 1.8 m2 | 200 mg b.i.d. | AUC0–24/MIC ≥ 125 and Cmax/MIC ≥ 10 | NA | 45% patients | NA |
| 2015 | Shotwell et al. [16] | 14 | CVVHD | 25 mL kg-1 h-1 | AN60 0.9m2 | 400 mg daily – 400 mg b.i.d. | AUC0–24/MIC>100 | NA | NA | 57% patients |
| 2016 | Roger et al. [1] | NA | CVVHF/CVVHDF | CVVHF: 30 mL kg-1 h-1 CVVHDF: 15 mL kg-1 h-1 + 15 mL kg-1 h-1 | Aquamax AF19 1.9 m2 | 400 mg b.i.d. – 400 mg t.i.d. | AUC0–24/MIC ≥ 125 | 100% patients (90 kg) | 0% patients (90 kg) | NA |
[i] AUC0–24 – area under the concentration-time curve over 24 hours, b.i.d. – twice a day, Cmax – maximum plasma concentration, CVVH – continuous veno-venous haemofiltration, CVVHD – continuous veno-venous haemodialysis, CVVHDF – continuous veno-venous haemodiafiltration, UF – ultrafiltration, MIC – minimum inhibitory concentration, NA – not available, NS – not specified, PK/PD – pharmacokinetic/pharmacodynamic, t.i.d. – three times a day
The aim of our study was to assess the pharmacokinetics and the probability of achieving PK/PD target for ciprofloxacin dosed 400 mg every eight hours intravenously during CRRT using a filter with large surface area (1.8 m2) and greater intensity.
METHODS
Patients
The study obtained the approval from the Bioethics Committee at the Regional Chamber of Physicians and Dentists in Olsztyn, and written informed consent was obtained from all subjects or from a legal surrogate. The eligibility criteria were met by 20 patients on CRRT (continuous veno-venous haemofiltration – CVVH, or continuous veno-venous haemodialysis – CVVHD) and receiving ciprofloxacin. Blood samples were collected from 18 patients during ciprofloxacin treatment. Two patients were excluded from the study because CRRT had to be discontinued in their case (cannula dysfunction, the need to transport a patient to perform a surgical procedure).
Antimicrobial treatment
The patients were considered eligible to be treated with ciprofloxacin (Ciprofloxacin Kabi, Fresenius Kabi, Germany) by the attending physician on the basis of patients’ culture results or as an empirical therapy. Ciprofloxacin was administered every eight hours in the dose of 400 mg (regardless of body weight) as a solution of 2 mg mL-1. The drug was administered in a one-hour infusion using a high-precision infusion pump (Fresenius Vial – Le Grand Chemin, Brezins, France). Continuous ECG monitoring was used in every patient to assess QT interval and detect arrhythmias.
In four patients the study was conducted during the administration of the first drug dose, while in 14 patients it was performed during the administration of subsequent doses.
Continuous renal replacement therapy
Patients were declared eligible for CRRT by the attending physician because of renal failure or overhydration. The choice of therapy between CVVH and CVVHD was also made by the attending physician, who took into consideration mainly the type of anticoagulation thought to be optimal for a particular patient. In the case of heparin anticoagulation CVVH was used, while CVVHD was used with citrate anticoagulation (CVVH for this method of anticoagulation was not available to the researcher). In all the patient groups CRRT was performed using a Multifiltrate machine (Fresenius Medical Care, Germany) using a filter with 1.8 m2 surface area (AV 1000 polysulfone membrane, Fresenius Medical Care, Germany), through a double lumen cannula inserted into the femoral vein or internal jugular vein. The data on intensity of CRRT are presented in Table 2.
TABLE 2
Demographic characteristics of the patients
Sample collection
Blood samples (2 mL) were collected to heparin tubes (Improvacuter, Shanghai International Holding Corp. GmbH, Hamburg, Germany) from the arterial line of the extracorporeal CRRT circuit before (time 0) and 30, 60, 75, 90, 120, 180, 240, and 480 minutes after the initiation of ciprofloxacin infusion. The blood samples were kept in an ice bath for up to 1 h and centrifuged at 3000 × γ for 10 min. The plasma was then frozen and stored at –80oC until it was subject to chromatographic analysis.
Ciprofloxacin determination by high-performance liquid chromatography
The ciprofloxacin instrumental analysis was performed using high performance liquid chromatography (HPLC) methods previously described by Onichimowski et al. [4]. The extraction procedure of ciprofloxacin from plasma samples was carried out according to Ziółkowski et al. 2014, and the calibration range was 0.1–10.0 μg mL-1 [18]. Because of changes in extraction procedure (type of matrix and ciprofloxacin instead of enrofloxacin) the method was revalidated in terms of linearity, precision, accuracy, and total recovery according to FDA (2018) [19].
Pharmacokinetic analysis
The pharmacokinetic analysis was performed using non-compartmental analysis with a commercial software program ThothPro™ (Gdańsk, Poland). The method of pharmacokinetic analysis determined the following: AUC from 0 to 8 h (AUC0→8) according to the linear trapezoidal rule; area under the first moment of the curve from 0 to 8 h (AUMC0→8); mean residence time (MRT); the slope of elimination phase (kel); half-life in the elimination phase (t1/2kel); apparent volume of distribution (Vd); volume of distribution in steady state (Vss); and total body clearance (ClB). The maximum concentration (Cmax) and the time of Cmax (tmax) were determined directly from the raw data.
Efficacy assessment
The measure adopted to assess the efficacy of the antibiotic therapy was the proportion of patients for whom pre-defined PK/PD indices were achieved. This corresponds to the probability of target attainment. AUC0–24 for the drug was calculated by multiplying the AUC0–8 obtained from its pharmacokinetic profile by three.
Statistical analysis
For all the patients the statistical analysis assessed the correlation between Cmax, Vss, and CLB and the albumin level, the day of antibiotic therapy, and the SOFA score on the day of the study. Also, the correlation of the pharmacokinetic parameters studied with the drug dose, and the calculated per kg body weight was assessed. The correlation was considered strong when r > 0.7. (GraphPad Prism 3.1; Graphpad Software, San Diego, CA, USA).
RESULTS
Antibiotic infusions were well tolerated by all the patients. No adverse effects associated with drug administration were observed.
The concentration-time profile for ciprofloxacin in particular patients is presented in Figure 1. The demographic characteristics of the patients, mode of CRRT, and intensity of procedure are presented in Table 2. The values of selected pharmacokinetic parameters of ciprofloxacin in individual critically ill patients are presented in Table 3.
FIGURE 1
Ciprofloxacin concentrations in critically ill patients treated intravenously with the drug at the dose of 400 mg per patient during continuous renwal replacement therapy
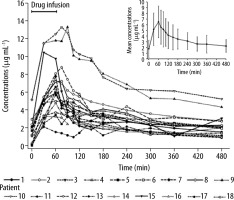
TABLE 3
The values of selected pharmacokinetic parameters of ciprofloxacin in individual critically ill patients after one-hour infusion at a dose of 400 mg per patient
[i] kel – elimination rate constant, t1/2kel – half-life in elimination phase, tmax– time of maximum concentration, Cmax – maximum plasma concentration, AUC0→8 – area under the concentration vs. time curve from 0 to 8 h, AUMC0→8 – area under the first moment of curve from 0 to 8 h, ClB – total body clearance, MRT – mean residence time, Vd – apparent volume of distribution, Vss – volume of distribution in steady state
There was considerable inter-individual variability observed in pharmacokinetic parameters for ciprofloxacin.
No correlation of pharmacokinetic parameters (Cmax, Vss, and ClB) was found with clinical parameters, i.e. albumin level, day of antibiotic therapy, and SOFA score on the day of the study. Likewise, there was no correlation found between the parameters studied and the dose of antibiotic as calculated per kg body weight.
The efficacy of the antibiotic dosage regimen used in our study in attaining PK/PD indices defined as effective in relation to particular MIC values is presented in Table 4.
TABLE 4
Percent of patients in whom PK/PD target was achieved for ciprofloxacin and particular MIC values
DISCUSSION
Although several papers so far have discussed the influence of CRRT on ciprofloxacin clearance, opinions on the fully effective dosing regimen remain divided (Table 1). Moreover, the results of the studies presented in the aforementioned papers are in some part becoming out-of-date due to the fact that CRRT techniques used in intensive therapy units have been changing, and these changes may result in increasing ciprofloxacin clearance.
In our study the values of pharmacokinetic parameters varied between the patients. A similar large inter-individual variability was also observed by other authors [2, 6, 10, 11]. Ciprofloxacin is a drug that is mainly excreted by the kidneys, but intestinal and biliary routes as well as metabolic pathways are also involved in its active elimination. The role of extrarenal mechanisms may be of much greater importance in patients with renal dysfunction, which, when CRRT is used, may result in large inter-individual differences in the drug concentrations in the blood [3]. The above-mentioned mechanisms may be responsible for the fact that drug concentrations in the blood of some patients may be significantly lower than the mean value in a given population, and thus may not reach therapeutic levels, particularly with CRRT implemented in patients with residual renal function [3]. Using fluoroquinolones is associated with fast growth of resistance, not only to drugs from this class but also other antimicrobials. Therefore, their appropriate use is crucial [21]. According to many papers, a ciprofloxacin dosage of 200 mg b.i.d. did not result in optimum PK/PD indices [10, 13]. In the paper by Scheer et al. discussing pharmacokinetics of ciprofloxacin used in the dose of 400 mg per day in 20 septic patients treated with CVVHD or CVVHDF, only in nine patients did AUC/MIC0–24 exceed 125 [10]. Many papers define the dosage of ciprofloxacin in the dose of 400 mg i.v. b.i.d. as sufficient [6, 9]. However, such conclusions often refer to low MIC values (e.g. 0.125 µg mL-1 in Zelenitski’s paper) or to the majority and not to the total population of patients in the study (Forrest) [6, 9]. In turn, Utup et al., in order to achieve AUC0–24/MIC ≥ 125 in a 160 kg patient with Enterobacter aerogenes infection and instituted CVVHDF, needed a dose of 1600 mg/day [12]. Lack of therapeutic success in a small proportion of patients causes a rise in antibiotic resistance on a personal and population basis. This is why, in the authors’ opinion, dosing algorithms should be considered effective when the proportion of patients attaining PK/PD target reaches or is close to 100%. In order to sustain the efficacy of fluoroquinolones they should be used in maximum doses approved or with therapeutic drug monitoring [1]. It is of note that in critically ill patients presenting with renal failure the role of extrarenal clearance may significantly increase. The addition of yet another route of drug elimination in the form of CRRT may cause a considerable decrease in the drug concentration in blood. If this is the case, the standard dosing regimen recommended by EUCAST (400 mg b.i.d.) may prove insufficient. Thus, the high doses recommended by EUCAST (400 mg t.i.d.) should be used in the treatment of all infections in critically ill patients undergoing CRRT [22]. In a study by Michiel Haeseker, in which ciprofloxacin concentrations were monitored in 80 patients with residual renal function receiving the drug in the dose of 400 mg b.i.d., 21% and 75% patients failed to reach the target AUC0–24/MIC ≥ 125 for MIC 0.25 and 0.5 mg L-1, respectively [11]. A computer-simulated increase in a daily dose from 800 mg to 1200 mg included in the paper decreased these percentages to 1% and 37%, respectively. Perreiter et al. investigated dosing regimens for ciprofloxacin in a tertiary acute care medical centre. Only in four patients out of 62 (6.54%) receiving the drug in the dose of 800 mg per day did the AUC0–24/MIC ratio exceed the value of 250 for MIC of the isolated pathogens [23]. Khachman et al. [8] assessed the concentrations of ciprofloxacin in 102 ICU patients using the value of fAUC0–24/MIC (where f is the free drug concentration) ≥ 90 (equivalent to AUC0–24/MIC ≥ 125) as an index for predicting clinical efficacy. The study demonstrated lack of clinical efficacy for the daily dose of 800 mg at MIC of only 0.25 mg L-1 and for the daily dose of 1200 mg at MIC of 0.5 mg L-1. In a paper by Roger et al., aiming to assess the population pharmacokinetics of ciprofloxacin during CRRT in 11 patients using Monte Carlo simulation in a population of patients with body mass of 90 kg, the AUC0–24/MIC exceeded 125 in 100% of patients for MIC = 0.25, for both the doses of 800 and 1200 mg/day. However, in the group with body weight of 140 kg the above-mentioned PK/PD target was achieved for the dose of 800 mg only for MIC = 0.125 mg L-1. Both dosing regimens were ineffective for MIC 0.5 mg L-1 regardless of body weight [1]. In our study, with the dose of 1200 mg per day used, 100% patients during CRRT achieved the parameter of AUC0–24/MIC ≥ 125 for MIC = 0.25, as in the paper referred to above; nevertheless, the more challenging target, i.e. the ratio of AUC0–24/MIC ≥ 250, was achieved only for MIC = 0.125. For MIC = 0.5 for G(-) bacteria the target PK/PD values were reached only in 66.66% of patients (Table 4). In the paper by Spooner et al., in turn, in all the critically ill patients in the study (n = 11) treated with CVVHD, with a ciprofloxacin dosage regimen of 800 mg per day, the AUC0–24/MIC ratio exceeded 125 for MIC = 0.5 mg L-1 [16]. The higher efficacy of the dosing regimen demonstrated by Spooner in comparison to that achieved in our study or Roger’s study may result from a significant difference in the surface area of the filter used during CRRT (0.55, 1.8, and 1.9 m2, respectively).
CONCLUSIONS
During the last decade, together with an increase in CRRT intensity and the surface area of the filter, there has been a rise in the effectiveness of antibiotic elimination in the critically ill. The intention is to maximally increase the proportion of patients in whom clinical efficacy, expressed as achieving the PK/PD target, is reached. In an attempt to bring it as close to 100% as possible, it seems reasonable that during CRRT high doses of ciprofloxacin should be used, i.e. 400 mg intravenously t.i.d., as recommended by EUCAST, particularly in obese patients. Such management, apart from maximally increasing the probability of therapeutic success, may reduce the risk of inducing antibiotic resistance in intensive therapy units.




