Introduction
Colorectal cancer (CRC) is widely diagnosed with cancer, accounting for over 9% of all forms of cancer, and the world’s fourth-largest cause of cancer deaths. It poses a significant concern for public health and is a major global cause of morbidity and mortality [1]. While CRC is a disease in the developed world, the rates of CRC in developing countries are increasing [2], which indicate an increased occurrence of CRC risk factors, such as unhealthy diets, obesity, and smoking [3]. CRC has a heterogeneous structure, with a small fraction of the tumours responding to chemotherapy. A molecular understanding is required of the CRC in oncology [4]. A previous study assessed the convenience of patient-derived tumour xenografts as a tool in cancer diagnosis [5].
The rate of CRC incidence has prompted the search for novel treatments furthermore markers cause early detection and the evaluation and monitoring of the prognosis of the drug therapy of choice. MicroRNAs (miRNAs) consider family of the latest research biomarkers. These RNA molecules modulate the other gene expressions and are approximately 20–22 nucleotides long. The strong correlation between tumourigenesis and miRNA expression has led to a substantial amount of research investigating their potential use as biomarkers and therapeutics for cancer. The maintenance of cancer, angiogenesis, and epithelial-mesenchymal transformation are also related to altered miRNA levels, all of which lead to malignancy [6].
MiR-224 and miR-200a are prognostic tumour markers of CRC. MiR-224 activates signalling of Wnt/β-catenin and directs the nuclear translocation of β-catenin in CRC [7]. Furthermore, an phenotype and bad prognosis have been significant with high levels of miR-224 expression, and miR-224 overexpression stimulated in vitro proliferation of CRC cells [8]. Additionally, miR-224 supports CRC metastasis. In CRC, miR-224 expressions have prognostic value for the survival of CRC patients, alone or with its targets [9]. MiR-200a one of miR-200 families present on chromosome 1, and it is overexpressed in mesenchymal-epithelial transition in CRC. Furthermore, a relation between the miR-200a family and permanence in CRC patients has been confirmed. Thus, miR-200a expression as in surgical samples may be helpful to design treatment. Further studies are necessary to assess the effectiveness of these miRNAs on the CRC treatment [10].
Chemotherapy is an adjuvant treatment for unresectable CRC, to prevent postoperative recurrence, and for systemic chemotherapy. Fluorouracil, oxaliplatin, tegafur, uracil-tegafur, and doxifluridine are anticancer drugs that have been approved for CRC treatment [11]. Oxaliplatin, a chemotherapeutic platinum-based drug, is used for the treatment of CRC. Platinum-based drugs increase cytotoxicity through oxidative stress, leading to the direct and indirect production of reactive oxygen species (ROS) [12]. Furthermore, its use causes significant gastrointestinal side effects, resulting in dosage limitation and therapy discontinuation [13]. Apoptosis is among the recognized pathways of oxaliplatin [14].
Numerous pharmaceutical agents originate from natural products, and it is fair to assume that there are additional agents in nature that remain undiscovered [15]. Phytic acid (PA) is an omnipresent, natural antitumour substance that is particularly abundant in cereals and legumes in the plant kingdom. PA and its lower phosphorylated forms present in much smaller amounts in most mammalian cells, where they are essential for critical cellular functions. During inositol polyphosphate metabolism, mammalian cells take up exogenous PA by feeding or they develop it. PA is involved in development, inflammation, and neurotransmission regulation [16].
Extracellular PA may be taken up and metabolized by mammalian cells, and the rate and trend of PA metabolism by cancer cells vary according to the cell type [17]. PA has shown antitumour activity in the experimental models of tumourigenesis, which is a fascinating property of PA [18]. The main aim of our study was the first-time determination of miR-224 and miR-200a in CRC. We also evaluated the possible antitumour and protective effects of PA in CRC treatment.
Material and methods
Chemicals
PA powder (PA sodium salt hydrate) and 1,2-dimethylhydrazine (1,2-DMH) were purchased from Sigma-Aldrich. Oxaliplatin was purchased from Sanofi-Aventis Deutschland GmbH (Frankfurt, Germany). Other chemicals were obtained following a quality control examination from commercial sources and had an analytical grade.
Animals
We purchased 36 male rats (150 g) from the Animal Care Unit of Vacsera. The rats under controlled experimental conditions were housed in polyethylene cages (temperature, 25°C ± 1°C; humidity, 50% ± 5%; and light/dark cycle). Food and water were available ad libitum. Every 3 days the animals were weighed, and their activity was tracked on a regular basis [19]. The Animal Care and Use Committee of the Faculty of Pharmacy at Damanhur University approved all experimental protocols (Ref. 1647ag).
Design and sampling
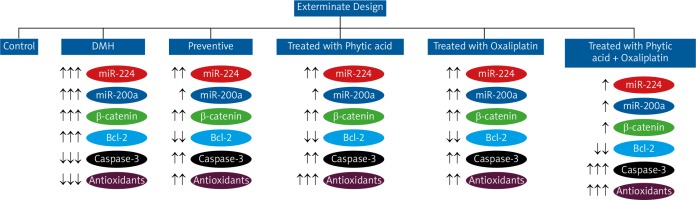
After 2 weeks of acclimatization, we zdivided the rats into 6 groups containing 6 rats each. Group I rats (control) were injected with 0.9% normal saline throughout the experiment. Group II rats (positive control) were subcutaneously injected with 1,2-DMH (20 mg/kg) twice weekly for 10 weeks [20]. Group III rats (preventive) were treated orally for 12 weeks with PA (2 weeks before induction with 1,2-DMH, as in group II, followed by 10 weeks of oral treatment with 0.5% w/v PA daily) [21]. Group IV rats (treated with PA) were treated orally with PA (0.5% w/v daily, 8 weeks) after induction with 1,2-DMH, as in group II. Group V rats (treated with oxaliplatin) were treated with intraperitoneal injections (ip) of oxaliplatin (5 mg/kg) after induction with 1,2-DMH, as in group II [22]. Group VI rats (treated with oxaliplatin + PA) were treated with a combination of a single weekly dose of oxaliplatin for 3 weeks (5 mg/kg ip) and oral PA (0.5% w/v) daily for 8 weeks after induction with 1,2-DMH, as in group II. All rats were sacrificed after 18 weeks (Fig. 1).
The rats fasted overnight at the end of the experiment and were anaesthetized by diethyl ether and sacrificed by cervical dislocation. Colorectal tissue was applied for histopathological analysis, and the remainder was homogenized in 0.1 M Tris-HCl buffer (pH 7.4) and centrifuged. The resultant supernatant was used for molecular and biochemical studies.
Blood samples (4 mL) were taken, centrifuged at 5000 rpm, and the serum was collected. Reverse-transcription-polymerase chain reaction (RT-PCR) was used to monitor the miR-224, miR-200a, β-catenin, and β-actin expressions. ELISA was performed to determine the expression of Bcl-2 and caspase-3 activity in colorectal tissues. The total protein content of the colorectal homogenate was estimated by Bradford reagent [23]. The samples were stored at –80°C until use.
RNA preparation and reverse-transcription-polymerase chain reaction
Total tissue RNA was isolated using a Total RNA Omega Bio-tek Kit. Then, RT-PCR was performed by an RT-PCR Master Mix Kit (122020-1109; (BIORON GmbH) and Taq Master (Jena Bioscience), which produces a high RNA yield. The cDNA strand was synthesized according to the instruction manual of the Revert Aid TM cDNA synthesis kit. PCR was completed by a thermal cycler (BOECO, BYQ6041R-158; Hamburg, Germany). The PCR conditions were as follows: pre-denaturing for 5 min at 94°C, 30 cycles of denaturing at 94°C for 30 sec, annealing at the primer-specific temperature for 30 sec, and extension at 72°C for 1 min.
Reference sequences obtained from the NCBI were applied to design primers for miR-224, miR-200a, β-catenin, and β-actin. The primer sequences and melting temperatures are illustrated in Table 1.
Table 1
Primers sequences used for the reverse-transcription-polymerase chain reaction technique
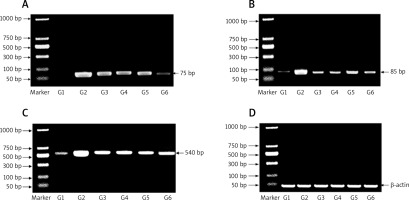
The products of PCR were electrophoresed on 1% of agarose gel using TAE buffer. A DNA ladder of 50–1000 kbp was used (Jena Bioscience). The bands were detected using a transilluminator and then photographed.
Determination of Bcl-2 in colorectal tissues using ELISA
We used a Bcl-2 Human SimpleStep ELISA Kit (ab202411; Abcam) for measurement of Bcl-2 protein quantitatively in the extracts of rat tissue. All reagents, working standards, and samples were prepared. The colorectal homogenates were typically prepared with phosphate-buffered saline (PBS). Then, 50 µL of sample or standard were added to the appropriate wells of the ELISA plate. After adding 50 µL of the antibody to each well, the plates were incubated for 1 h. Next, each well washed with 350 µL of 1× Wash Buffer 3 times, and 100 µL of 3,3’,5,5’-tetramethylbenzidine substrate was added to each well and incubated for 10 min. Finally, 100 µL of stop solution was added, and the absorbance was read at 450 nm. The Bcl-2 concentration (ng/mL) was calculated by using a typical standard curve.
Determination of caspase-3 levels
We used ELISA Kit of Caspase-3 (MBS262914; MyBioSource, San Diego, USA) for the quantitative determination of caspase-3 in the rat tissue homogenates. Briefly, the tissue homogenate was prepared using tissue slices washed in 0.01 M PBS. A tissue protein extraction reagent was added, mixed in ice water (1 g: 10 mL), and centrifuged for 10 min at 5000 rpm, and supernatant was removed for use. Each supernatant sample was added to corresponding wells (100 µL/well) of the ELISA plate and incubated at 37°C for 90 min. Biotinylated rat caspase-3 antibody (100 µL/well) was added and incubated at 37°C for 60 min. Then, the enzyme-conjugate was prepared, and 100 µL was added and incubated at 37°C for 30 min to each well. Finally, 100 µL of a coloured reagent was added to wells and incubated at 37°C for 15 min. The absorbance was recorded at 450 nm within 10 min using a reader (1603029; BioTek Instruments, USA).
Estimation of tissue total antioxidants
We used an assay Kit of Total Antioxidant Capacity (ab65329; Abcam) to determine the total tissue antioxidant levels. Briefly, the small kit vials were centrifuged. Then, the sample was centrifuged at 4°C for 5 min. All reagents were equilibrated to room temperature prior to use. Cu2+ working solution was freshly prepared before adding 100 µL to all standard and sample wells. The solution was mixed, and the plate was protected from light and incubated at room temperature for 90 min on an orbital shaker. The absorbance at 570 nm was measured using a microplate reader. Trolox was used to standardize the antioxidant results by measuring the other antioxidants in Trolox equivalents. The concentration of total antioxidant capacity (TAC) or Trolox (nmol/mg) was calculated as Ts/SV * D, where Ts = TAC amount in the sample, Sv = sample volume added to the sample well, and D = sample dilution factor.
Histopathological examinations
Fresh colorectal tissues were washed with 0.9% saline solution and fixed in buffered formalin. Then, the formalin-fixed tissue samples were transferred to 70% ethanol and embedded in paraffin. Finally, paraffin block sections (0.5 µm thick) were double stained with haematoxylin and eosin (HE) and visualized using an optical microscope.
Data analysis
The data were statistically analysed using Graph Pad Prism 4.0 software (GraphPad Software, Inc., San Diego, CA). Values were reported as means ± standard deviation (SD), and p-values < 0.05 were considered statistically significant. Multiple comparisons were performed using one-way analysis of variance (ANOVA) followed by Tukey’s multiple comparison post hoc test.
Results
Expression of miR-224, miR-200a, and β-catenin by reverse-transcription-polymerase chain reaction
Figure 2 shows the mRNA expression of miR-224, miR-200a, and β-catenin expression as determined by RT-PCR for all experimental groups. The expression of miR-224, miR-200a, and β-catenin was increased in the carcinogenic group but significantly decreased in the other groups (group 3: prophylactic, group 4: treated with PA, group 5: treated with oxaliplatin, group 6: treated with oxaliplatin + PA). β-actin was used as the housekeeping gene in the RT-PCR assays.
Estimation of Bcl-2, Caspase-3, and total antioxidants in colorectal tissues by ELISA
Table 2 illustrates the Bcl-2, Caspase-3, and total antioxidants levels in all experimental groups. Bcl-2 levels were markedly increased in group II but reduced significantly in other treated groups (group 3: prophylactic, group 4: treated with PA, group 5: treated with oxaliplatin, group 6: treated with oxaliplatin + PA). Caspase-3 and total antioxidants were significantly reduced in group II but increased gradually in other groups.
Table 2
Levels of Bcl-2, Caspase-3, and total antioxidants in all experimental groups
[i] Data were expressed as mean ± SD of Bcl-2: B-cell lymphoma-2, Caspase-3, and total antioxidants. * – values differ significantly from group I (control group) (p < 0.05), # – values differ significantly from group II (positive control) (p < 0.05). Statistical analysis was determined by one-way analysis of variance (ANOVA) followed by Tukey’s test as a multiple comparison post-ANOVA test
Histopathological results
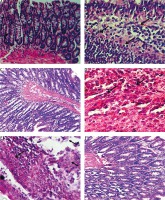
We examined HE-stained slides to evaluate changes in the colorectal cells in all rat groups. Normal architecture and histological structure were observed in the colorectal section of rats in the control group (Fig. 3 A). On the other hand, the positive control group showed proliferation of the epithelial layer tumour cells infiltrating the submucosa (Fig. 3 B), with distorted, swollen nuclei and tightly packed glands causing invasive adenoma. The tumour cells were polyhedral with hyperchromatic nuclei. The preventive group showed a more or less natural histological appearance, with some villi and goblet cells with simple columnar epithelium (Fig. 3 C). Additionally, the regeneration of villi and epithelial linings were observed in response to PA treatment as well as reduced penetration of cancer cells into the connective tissue (Fig. 3 D). The oxaliplatin group showed colorectal necrosis and strong mast cell infiltration (Fig. 3 E). The group treated with PA and oxaliplatin showed normal morphology with some inflammation (Fig. 3 F).
Fig. 3
Histopathological analysis showing the effect of phytic acid (PA) alone or in combination with oxaliplatin. A – normal architecture and histological structure are shown in the colorectal section of rats in the control group, B – positive control group showing proliferation of epithelial layer tumour cells infiltrating the submucosa, C – preventive group showing some villi and goblet cells with simple columnar epithelium, D – regeneration of villi and epithelial linings and reduced penetration of cancer cells into the connective tissue following PA treatment, E – oxaliplatin group showing colorectal necrosis and strong mast cell infiltration (arrows), F – normal morphology and some inflammation in the experimental group treated with PA and oxaliplatin. Slides were stained using haematoxylin and eosin (×40)
Discussion
Colorectal cancer is a common cancer type in many countries. Because it has few symptoms, it is usually discovered in the final stages [24]. Although CRC has many tumour markers, its diagnosis remains problematic. miRNAs are small, non-coding RNAs that act as suppressors of many cancer types. miR-224 was found to play a primary role in several types of cancer and may be prognostically and biologically important in CRC [25].
Our study determined the presence of miR-224 and miR-200a in CRC. We found that miR-224 expression markedly increased in CRC and enhanced its prognosis. Furthermore, our results confirmed that miR-224 and miR-200a were significantly increased in rats with CRC. Moreover, the levels of miR-224 and miR-200a were markedly up-regulated in the carcinogenic group (untreated CRC) compared with the groups treated with PA alone or in combination with oxaliplatin. MiR-224 levels consistently increase with cancer severity, and miR-224 promotes CRC metastasis of in vitro and in vivo by regulating SMAD4. Furthermore, miR-224 levels may have a prognostic role in the survival of patients with CRC [9].
Similarly, we found that miR-200a expression was increased with cancer progression. Both miR-224 miR-200a have a diagnostic and prognostic effect on CRC by reducing inflammation and exerting antitumour effects through different mechanisms. In our study, apoptosis was evaluated by determining the levels of Bcl-2 and caspase-3 expression.
β-catenin plays a role in regulating cell-cell adhesion and is a crucial component of the Wnt pathway. In our study, β-catenin overexpression was associated with CRC. We determined the expression of β-catenin in colorectal tissue in rats in different experimental groups using RT-PCR. β-catenin expression levels were increased in CRC and decreased in the groups treated with PA alone or in combination with oxaliplatin. The expression of β-catenin in azoxymethane-induced rat colon tumours relative to adjacent healthy colon mucosa was found to be pronounced by immunostaining and immunoblotting in both colon adenoma and carcinoma tissues as well as in the cytoplasm and nucleus of the cancer cells. Furthermore, antibody binding in healthy colon epithelial cells was restricted to membranes at intercellular borders [26].
Oxaliplatin is a chemotherapeutic agent used as the drug of choice in CRC treatment. It stimulates cytotoxicity by increasing the levels of ROS and oxidative stress. Because oxaliplatin has severe gastrointestinal side effects, the use of natural products, such as PA, reduces the dose of oxaliplatin as well as its side effects and cytotoxicity. The apoptosis pathway is the main mechanism of action for oxaliplatin as a chemotherapeutic drug.
PA is a natural product that is present in grains and nuts. Our study found that PA might inhibit CRC and play a vital role in the chemoprevention of CRC. Furthermore, PA treatment could reduce the expression of tumour markers such as miR-224 and miR-200a. Our findings also suggested that PA had a synergistic action with oxaliplatin in CRC treatment. In our study, we attempted to identify some of the pathways involved in the effect of PA as a preventive and therapeutic agent for CRC. It has been shown that PA has a potent chemopreventive and antitumour action against CRC. The development and progression of CRC include defects of the intestinal mucosal barrier and inflammation processes. The effect of PA on the intestinal mucosal barrier and proinflammatory cytokines was investigated, and PA was found to have beneficial effects on the intestinal mucosal barrier and reduce serum proinflammatory cytokine levels [27].
Our study found that the Bcl-2 level increased with the incidence of CRC and decreased when rats were treated with PA alone or in combination with oxaliplatin. Bcl-2 is an apoptotic regulator that suppresses apoptosis in several cell types (antiapoptotic agent). As such, it is a crucial protein involved in cell control and is critically related to a number of cancers through mutation or damage to the structure or function of the Bcl-2 proteins. The fusion protein’s unregulated expression induces an unusually high amount of Bcl-2, disrupting apoptosis regulation and leading to unregulated cell growth. The functional interface of Bcl-2/p53 represents a central regulator of apoptosis that can be triggered in colorectal carcinoma cells by targeting Bcl-2 [28].
Apoptosis is an important pathway that clarifies cancer incidence and its regulation. We investigated the expression of caspase-3 as an apoptotic agent. The levels of caspase-3 were markedly reduced in CRC induced with 1,2-DMH and significantly increased in rats treated with PA alone or in combination with oxaliplatin. These findings showed that PA interacted with the apoptosis pathway in the treatment of CRC in rats.
Besides its anticancer properties, PA is also of nutritional value. The anticancer activity of PA against hepatocellular carcinoma cells has been investigated using expression studies of the apoptosis-regulating caspase-3 genes by RT-PCR and DNA fragmentation assays to assess apoptotic activity. Hepatocellular carcinoma cells treated with PA showed caspase-3 up-regulation and Bcl-2 down-regulation, and apoptosis was evident by the presence of fragmented genomic DNA in PA-treated cells. Furthermore, a reducing power assay showed that PA had antioxidant activity. PA had biological activity, stimulated apoptosis in cancer cells, and showed growth inhibition [29].
Our study found that PA is useful because of its possible protective role against CRC in rats. PA treatment alone or in combination with oxaliplatin increased total antioxidant levels and decreased oxidative stress in rats with CRC. Our results correlated with a previous study showing that PA acted as an antioxidant against the harmful effects of generated toxins and played an important role in reducing oxidative stress levels. Because these antioxidants work together, a change in any of them can disturb the balance and trigger cell damage that leads to cancer [30].
The ROS levels were increased in 1,2-DMH-induced CRC rats, resulting in an imbalance between the generation and removal of ROS and leading to oxidative stress-related malignancy. Antioxidants are reducing agents that become oxidized and prevent the production of free radicals. In some cancer cases, the level of antioxidants produced is insufficient to totally overcome the free radicals. Free radicals may be generated by the action of 1,2-DMH in CRC models [31]. Our histopathological examinations confirmed our biochemical and molecular findings. The histopathological analysis of CRC cells showed logical evidence of the potential effect of PA alone or in combination with oxaliplatin against CRC.
Conclusions
Our study revealed that PA possessed efficient anticarcinogenic properties against 1,2-DMH-induced CRC in rats when used as a preventive agent or as a treatment agent alone or in combination with oxaliplatin. PA inhibits CRC in rats through pathways of apoptosis, cell proliferation, and antioxidants.