Practice variability among healthcare providers can increase the risk of human error and is associated with adverse events [1]. For example, it has been demonstrated that different colour-coding schemes for syringe labels between institutions once contributed to increased risk of morbidity and mortality with drug administration [2]. Therefore, to decrease that variability, standardized colour-coding of syringe labels was introduced in the USA in 1994, in Australia and New Zealand in 1996, in Canada in 1999, and in the UK in 2003 [3]. Since then, evidence-based studies have demonstrated that standardized colour-coding of syringe labels results in a decreased incidence of drug swap errors [4, 5]. Nevertheless, drug administration errors continue to be reported and have not been eliminated. One possible contributor to the continued observation of drug errors may be related to practice variability with other aspects of the drug label, such as location, orientation, written details, and legibility. We, therefore, sought to describe the practice variability with drug labels among a cohort of anaesthesia residents in an academic medical centre.
METHODS
Setting
The protocol for this study was determined to be exempt in accordance with 45 CFR 46.104(d) by the Tufts Health Sciences Institutional Review Board (STUDY00000614). The study was performed at an urban tertiary care academic medical centre with an anaesthesia residency program.
Data collection
In a controlled, lecture-style environment, residents were instructed to prepare a syringe of a drug. Each participant received one 10-mL syringe (Becton Dickinson, Franklin Lakes, NJ), a blunt needle, and a pen. Participants were also given a roll of the atracurium label (FSI Label Company, Holland, MI) on which the name “atracurium” was pre-printed but other details (e.g. concentration) were blank. Residents were asked to label the syringe as they usually do in the real perioperative environment. Once the task was completed, all syringes were collected and assessed for label placement, compliance with the Joint Commission on Accreditation of Healthcare Organizations (JCAHO) standards (which includes writing the medication name, concentration, date, time, and initials of the provider preparing the syringe), and legibility.
Outcome measures
The primary outcomes included:
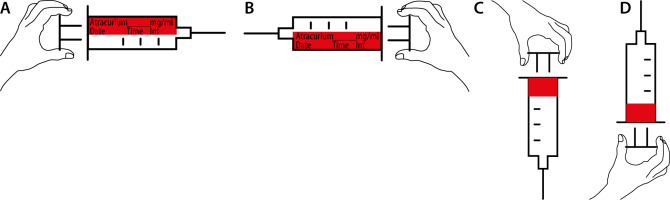
Location of the label (longitudinal vs. circumferential), where longitudinal was defined as a label placed across the length of the syringe, while circumferential was defined as a label wrapped around the base of the syringe (see Figure 1 for examples).
Orientation (right-hand vs. left-hand orientation), where right-hand orientation was defined as a longitudinal label on which the text was upright when the plunger was held with the right hand, and left-hand orientation was defined as a longitudinal label on which the text was upright when the plunger was held with the left hand (see Figure 1 for examples). For consistent terminology, we arbitrarily defined circumferential labels as being of right-hand orientation when the text was upright with the needle pointing upwards, and as being of left-hand orientation when the text was upright with the needle pointing downwards.
Secondary outcomes included:
Compliance with JCAHO labelling standards, defined as including drug name, concentration, provider initials, and date and time of preparation.
Legibility of the drug labels. To assess legibility, we asked 3 non-clinical staff to determine as either legible or not each of the JCAHO labelling components. The total responses were tallied, and in the case that the responses were not unanimous, we used the response of 2 out of the 3 as the final result, in keeping with prior methodology [6]. In the case of syringes with more than one label, all labels were assessed for legibility. For missing label components (e.g. no concentration listed), legibility was not assessed. If the labels had preprinted names and were thus always legible, staff were instructed that if the label name was partially covered or the label was ripped in a way where only part of the name was visible, then it should be considered illegible.
RESULTS
A total of 32 anaesthesia residents who attended the lecture also participated in the study, placing 48 labels on 32 syringes. A total of 11 (34%) syringes had a single longitudinally placed label, 6 (19%) had a single circumferential label, and 15 (47%) had both longitudinally and circumferentially placed labels. Of the syringes with longitudinally placed labels, 17 (63%) were placed in the right-hand orientation, 9 (33%) in the left-hand orientation, and 1 (4%) had 2 labels placed in both orientations. Of the syringes with circumferentially placed labels, 17 (81%) were placed in the right-hand orientation and 4 (19%) in the left-hand orientation. Table 1 describes compliance with JCAHO labelling standards and legibility.
TABLE 1
Label documentation
DISCUSSION
In this study, we observed a great deal of practice variability with syringe labelling among anaesthesia residents. We found that about a third of participants used a single longitudinal label, about a fifth used a single circumferential label, and nearly half used labels in both locations on a single syringe. We also found significant variability with the orientation of the label on the syringes, with most participants placing labels in the right-hand orientation. Compliance with JCAHO standards for label information was higher with longitudinally placed labels compared to circumferentially placed labels, and legibility was generally high.
The finding that there was no single clearly acceptable location for placement of the drug labels demonstrates that there is significant practice variability with drug labelling among our residents despite a standardized colour-coding scheme. While our results cannot be used to demonstrate a decreased risk of error with the use of one approach over another, advantages and disadvantages with circumferentially placing labels have been described. Several articles suggest that circumferential labels may decrease the “search time” for specific syringes because circumferential labels can be seen regardless of how the syringe is oriented, while longitudinal labels may not be visible if the label is facing away from the provider [7, 8]. At the same time, one of the disadvantages of circumferentially affixed labels is that the drug name may not be legible without rotation [9], which can be particularly problematic when using barcode-based medication safety systems [10].
While there was less practice variability with regards to the orientation of the syringe label, because most labels were placed in the right-hand orientation, there was still a significant proportion that were placed in the left-hand orientation. We did not ask our participants about handedness, but if our residents represent a sample similar to the general public, then about 10% of our residents would be left-handed [11]. The finding that more than 10% of labels were placed with the left-hand orientation suggests this is less likely due to handedness, and probably represents a practice variability that could contribute to confusion and adverse drug events.
We observed significantly lower practice variability with compliance with JCAHO standards and legibility, because label information was nearly 100% compliant with longitudinally placed labels and legibility was also nearly 100%, despite anecdotal evidence suggesting that labels are commonly not compliant and illegible. Our finding might be due to the Hawthorne effect [12] because the residents knew they were participating in an activity related to proper drug labelling. More realistic compliance might be that which was observed with the circumferentially placed labels, in which only about a third of labels were compliant. Because anaesthesia providers commonly use labels with pre-printed drug names but have to handwrite the concentration, date, time, and initials, it is not uncommon for providers to skip this additional step as a strategy to improve efficiency and decrease the cognitive load. However, incomplete labelling can contribute to drug error, and therefore other strategies to improve efficiency (e.g. using label printers, which have been shown to increase compliance with JCAHO standards [13]) may be more appropriate.
CONCLUSIONS
We observed a significant amount of practice variability among our residents with different aspects of drug labelling. The risk of drug error is most likely to be highest in cases where a handover occurs [14], because different anaesthesia providers have their own approach to labelling that might conflict with that of the other providers. Standardizing orientation and location of syringe labels should be considered as a quality and safety initiative. Using prefilled syringes results in less cognitive complexity with drug administration [15], and one of the features of prefilled syringes is that the labels are all oriented and placed in the same way each time by the manufacturer. Such further standardization of syringe labels should be considered by major medical societies, especially because the increased cognitive load is associated with decreased clinical performance and can contribute to drug error.