Dear Editor,
This letter discusses precautions regarding the waterproof structure of the oximetry central venous catheter (CVC) shaft and the risk of blood reflux and leakage that can occur when the catheter shaft is cut.
To assess oxygen supply-demand balance [1] and haemodynamics [2], the Swan-Ganz pulmonary artery catheter has been used for perioperative and postoperative management and treatment of critically ill patients for a half a century. In current clinical practice, haemodynamic parameters such as stroke volume (SV) and cardiac output (CO), as well as central venous oxygen saturation (ScvO2), can be measured continuously using a combination of FloTrac Sensor (Edwards Lifesciences Japan Ltd., Tokyo, Japan) [3], Edwards PreSep Oximetry Catheter (Edwards Lifesciences Japan Ltd., Tokyo, Japan) [4], and EV1000 Clinical Platform (Edwards Lifesciences Japan Ltd., Tokyo, Japan) or Vigileo Monitor (Edwards Lifesciences Japan Ltd., Tokyo, Japan) [5, 6]. These methods are less invasive than the Swan-Ganz pulmonary artery catheter, and the changes in the parameter values can be used as an index for perioperative management in both cardiovascular and non-cardiovascular surgeries. In addition, they can be used for the treatment of critically ill patients in the intensive care unit, enabling proactive determination of an appropriate therapy [7]. Compared to intermittent sampling and traditional vital signs alone, continuous ScvO2 monitoring is a more sensitive indicator of tissue perfusion because it reveals the true adequacy of tissue oxygenation, enabling early detection and assessment of clinical response to intervention [7, 8].
We report the case of a 39-year-old male patient (height 171 cm; body mass 60 kg) with tricuspid atresia and ventricular septal defect, who underwent a Fontan conversion operation with an extra cardiac conduit-total cavopulmonary connection (TCPC) [9, 10], after an atriopulmonary connection (APC) Fontan operation 30 years ago, due to a history of pulmonary embolism and arrhythmia [11, 12]. Because the use of a Swan-Ganz pulmonary artery catheter was impossible due to Fontan circulation, the perioperative anaesthesia management was performed using a combination of FloTrac Sensor, Edwards PreSep Oximetry Catheter, and EV1000 Clinical Platform. An Edwards PreSep Oximetry Catheter was inserted 13 cm via the real-time ultrasound-guided right supraclavicular approach [13]. The tip of the Edwards PreSep Oximetry Catheter was approximately 2 cm deeper than the carina level on a chest X-ray taken during anaesthesia induction to check CVC depth, which is a routine practice at our institution for Glenn and Fontan operations. Therefore, it was pulled out 2 cm, considering that the planned surgery is a Fontan conversion operation, and affixed to the skin at 11 cm. After completion of thoracotomy and adhesion release around the heart and great vessels, the patient was connected to a cardiopulmonary bypass. When the APC site on the pulmonary artery was incised to connect a 22 mm Goa-Tex extracardiac TCPC conduit, the tip of the Edwards PreSep Oximetry Catheter was found to be so deep that it remained in the right atrium used for the APC. Because the opening of the proximal lumen of the Edwards PreSep Oximetry Catheter is approximately 7 cm from the catheter tip (Figure 1), which is much more proximal than a normal CVC, withdrawing the Edwards PreSep Oximetry Catheter was considered unsafe because of the risk of extravascular deviation of the proximal lumen opening. Therefore, the surgeon cut the catheter tip from the surgical field, even though it was possible that the ScvO2 value would no longer be measurable. However, the EV1000 Clinical Platform displayed the ScvO2 value for nearly three hours thereafter with realistic values, showing fluctuations between 82% and 92% as before. After around three hours, the EV1000 Clinical Platform gave an alarm and the ScvO2 value disappeared suddenly. The anaesthesiologist checked the optical module connector part of the Edwards PreSep Oximetry Catheter (Figure 2) and found that it was wet with blood that had flowed back. Therefore, a normal CVC was newly inserted in the right internal jugular vein, and the Edwards PreSep Oximetry Catheter was removed to reduce infection risk after the surgery.
FIGURE 1
Tip of Edwards PreSep Oximetry Catheter. The opening of the proximal lumen is located at approximately 7 cm from the catheter tip
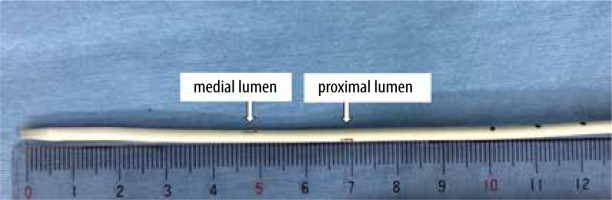
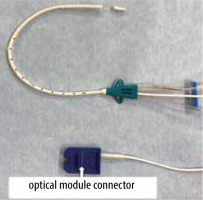
FIGURE 2
An Edwards PreSep Oximetry Catheter with the catheter tip cut off. The optical fibres are seen at the cutting stump of the catheter shaft. The optical module connector became wet with blood that had flowed back from the cutting stump of the catheter shaft
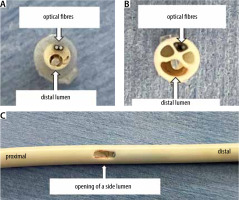
Figures 3A and 3B show the cut tip of the Edwards PreSep Oximetry Catheter. The catheter shaft has four lumens: three lumens for infusions and drugs, and a fourth lumen for two optical fibres for ScvO2 measurement. The drug reaches only the distal lumen at the catheter tip because the medial and proximal lumens are occluded on the distal side of the opening in the catheter shaft (Figure 3C). As Figure 3B shows, the lumen for the ScvO2 optical fibres is hollow and not waterproof. Therefore, the blood flowed back from the cutting stump of the catheter shaft via the lumen for the ScvO2 optical fibres and reached the optical module connector. As a result, the optical module connector became wet, causing the ScvO2 value to be displayed no longer. Nonetheless, it is still unclear how the ScvO2 value could be displayed for several hours after the catheter tip was cut, and whether the shown ScvO2 value was accurate; therefore, we contacted the manufacturer, Edwards Lifesciences Japan Ltd. (Tokyo, Japan), and asked them. The near-infrared rays are transmitted through an optical fibre in the catheter shaft and irradiated onto blood. The reflected light is transmitted to the optical module through another optical fibre, and then the ScvO2 value is calculated by spectrophotometry. Therefore, the ScvO2 value can be measured even when the catheter shaft is cut. However, the lumen for the ScvO2 optical fibres is waterproof only at the CVC tip and not in the catheter shaft.
FIGURE 3
The cut tip and the opening of a side (middle or proximal) lumen on the catheter shaft of the Edwards PreSep Oximetry Catheter. A) Catheter tip seen from the tip side. The distal lumen is open, and two optical fibres are located at the catheter tip. B) Catheter tip seen from the shaft stump. It has three lumens for infusions and drugs, and another lumen for two optical fibres for the ScvO2 measurement, which is not waterproof. C) The opening of a side lumen on the catheter shaft, which is occluded on the distal side with some blue material
The CVC tip is made of soft material and has a tapering structure so as not to damage the inner wall of the heart and blood vessels. When the CVC is inserted too deep, its tip may obstruct the operative procedure or cause harm at the anastomosis in cardiac surgery, or it may lead to life-threatening complications such as arrhythmia or pericardial tamponade [14, 15]. This may lead to withdrawal of the CVC or cutting of the CVC tip, after considering the disadvantages of each alternative. A similar problem can arise when using the Edwards PediaSat Oximetry Catheter for paediatric patients [16] because it has a similar structure to the Edwards PreSep Oximetry Catheter.
Written informed consent was obtained from the patient for publication of this paper and accompanying image.




