Summary
Assessments of predictive biomarkers in the management of atrial fibrillation recurrence after radiofrequency catheter ablation (RFCA) has been grown as an interesting topic. N-terminal pro-B-type natriuretic peptide (NT-proBNP) is considered a possible marker for artial fibrosis. This was a single-center research of 326 patients with nonvalvular AF and preserved systolic function after enduring an initial RFCA. These patients suffered AF recurrence had considerably greater pre-ablation NT-proBNP levels. A high pre-ablation NT-proBNP level was powerful to predict AF recurrence after RFCA.
Introduction
Radiofrequency catheter ablation (RFCA) for rhythm control is a successful healing choice for individuals with atrial fibrillation (AF) [1]. However, a substantial relapse rate after RFCA remains a persistent problem [2]. Because the ablation techniques have been developed over time, the relapse rate might be partially due to the complicated pathological mechanisms. Although potential factors, including atrial fibrosis and remodeling, myocardial injury markers, and inflammatory factors, have been verified to predict post-ablation recurrence [3–5], parameters that can simply create the stratification of patients who will sustain a better rhythm outcome are urgently needed in clinical practice. There has been increasing interest in identifying the predictive significance of pre-ablation blood biomarkers linked to fibrosis that could predict post-ablation recurrence because blood detection is very inexpensive, quick, and commonly accepted in daily check-ups [6].
N-terminal pro-B-type natriuretic peptide (NT-proBNP) is a neurohormone that myocytes release in reaction to abnormal haemodynamic circumstances, and it is also linked to fibrosis of the heart [7]. The atrium may undergo a reverse remodeling process when successful AF ablation and NT-proBNP levels can drop after sinus rhythm is restored [8]. Meanwhile, it is inconsistent whether increased natriuretic peptides are real predictors of AF recurrence at certain time points [9].
Aim
We conducted this research study to demonstrate the predictive significance of NT-proBNP as a follow-up biomarker for recurrence of RFCA patients.
Material and methods
Study population
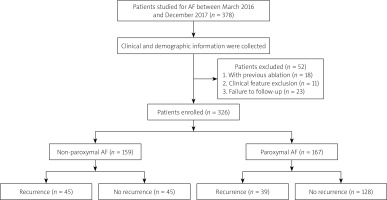
A total of 326 patients with nonvalvular AF, who underwent a primary RFCA between March 2018 and December 2019, were retrospectively analysed. Patients aged less than 20 years or over 85 years, patients with heart failure that was not under the control, or a low left ventricular ejection fraction < 50%, renal insufficiency, valvular diseases, congenital heart disease, hyperthyroidism, malignancies, or current inflammation were excluded from consideration. Likewise, patients with missing/incomplete data were also excluded. At the final stage of this research, paroxysmal and non-paroxysmal patients were categorized into 2 groups, respectively: individuals who had a constant sinus rhythm during continuation (the “no recurrence group”) and individuals who experienced a recurrence of AF (the “recurrence group”). The participants’ demographic and clinical characteristics containing sex, age, body mass index (BMI), prior histories, evaluated scores of HAS-BLED and CHA2DS2-VASc, prescribed medications, blood tests, echocardiographic parameters, ablation procedure, complications, and follow-up information were compared. The flowchart of subject inclusion is summarized in Figure 1. The Chinese PLA General Hospital’s Human Ethics Review Committee approved the study. All individuals gave informed consent for their ablation operation and the retrospective use of their clinical information. The protocols followed the “Declaration of Helsinki” and the competent committee on human experimentation’s ethical guidelines.
Definitions
Paroxysmal AF was described as AF that ended unexpectedly or was cardioverted within 7 days, persistent AF was described as AF that continued for more than 7 days and was treated with medications or direct current cardioversion, long-lasting persistent AF was described as AF that continued more than one year, and permanent AF was described as AF that was unsuccessful or indicated for rhythm reversal methods, such as ablation and cardioversion [10]. Non-paroxysmal AF was composed of persistent AF, long-lasting persistent AF, and permanent AF. The recurrence of AF was described as any atrial tachyarrhythmia (AF/atrial tachyarrhythmia/atrial flutter) lasting at least 30 s and being recorded by ECG or 24-hour Holter monitoring after the 3-month “blanking phase” after extirpation. HFpEF was defined as heart failure with left ventricle ejection fraction (LVEF) ≥ 50% [11]. Participants with estimated glomerular filtration rate (eGFR) < 60 ml/min/1.73 m2 were considered as renal insufficiency [12]. We calculated eGFR using the Chinese version of the modification of diet in renal disease equation as follows [13]: eGFR (ml/min/1.73 m2) = 175 × standard creatinine (mg/dl) – 1.234 × age (years) − 0.179 × 0.79 (if female).
Measurement of NT-pro-BNP
Blood samples from the periphery were obtained from every individual who was fasting on the 2nd morning after being admitted to the hospital. Within 1 h after collection, all samples were submitted to our hospital’s central laboratory. For 15 min, blood samples were centrifuged at 3500 RPM to extract plasma samples, and the NT-proBNP level was estimated using a chemiluminescent immunoassay (Roche Diagnostics, Mannheim, Germany).
Radiofrequency catheter ablation
Before beginning the process, all participants underwent transoesophageal echocardiography (TEE) to exclude any thrombus in the left atrium (LA), and multidetector computed tomography (MDCT) scans for better knowledge of their pulmonary vein (PV) and LA anatomy. No antiarrhythmic medicines were used for a minimum of 5 half-lives prior to ablation. Each participant received anticoagulation treatment for one month before the surgery and 3 months after that. The ablation process was carried out under analgesia with intravenous fentanyl. Due to a transseptal puncture, intravenous heparin (100 IU/kg) was given to keep the stimulated clotting time between 300 and 350 s. The PentaRay catheter (Biosense Webster, USA) was applied to reassemble the (3D) 3-dimensional structure of the PV and LA trunks with the CARTO 3 mapping method (Biosense Webster, United States), by which the LA and PV anatomy was mapped and incorporated into the MDCT scan image. A point-by-point irrigated radiofrequency ablation directed by electroanatomic mapping was performed on each PV antrum. At 43°C, a limit temperature with a maximum power of 35 W and irrigation rate of 17 ml/min for 20–30 s was to be supplied. During ablation of the posterior wall, the RFCA power was reduced to 25 W to reduce the risk of damaging surrounding structures. The procedure aimed to isolate the 4 pulmonary veins in both directions. Additional non-pulmonary vein ablation was left to the operator’s discretion, containing linear ablation (LINE, e.g. LA roof, LA floor, superior vena cava, cavotricuspid isthmus or mitral isthmus), and/or complex fractionated atrial electrography (CFAE). If the AF continued after the ablation, direct biphasic current shocks of 100–150 J were utilized to maintain the sinus pulse. Heparin was employed in the procedure once again.
Clinical follow-up
Anti-arrhythmic drugs (AAD) and anticoagulation therapy were given orally for at least 3 months after the procedure. Outpatient hospital evaluation with 12-lead electrocardiograms (ECG) and 24-hour Holter was conducted every 3 months during the first year and then every 6 months for a total of 24 months. External event recorders such as 24-hour Holter monitoring, and ambulatory electrocardiograms were performed whenever patients experienced symptoms, for example, vertigo, fatigue, palpitations, or nausea. Individuals were invited to email us their ECG and 24-hour Holter through WeChat if their outpatient follow-up was cancelled, and phone interviews were organized. Analyses of ECGs were achieved by skilled electrophysiologists, and the rhythm was depicted along the following classification: sinus rhythm, AF, and other (i.e. pacemaker and atrial flutter). Assessment of therapeutic follow-up data was performed by researchers blinded to values of NT-proBNP.
Statistical analysis
Continuous variables were stated as the mean ± median or SD (interquartile range) and linked using the independent Mann-Whitney U test or sample t-test based on the normality test results. The percentages were expressed for categorical variables in percent (%) and compared using the χ2 test or Fisher exact test. Correlation analyses between levels of NT-proBNP and other variables were conducted using Spearman’s rank test. The capacity of NT-proBNP to discriminate in predicting AF recurrence after RFCA was assessed by the receiver operating characteristic (ROC)-derived (AUC) area under the curve. Kaplan-Meier (KM) survival curves were used to estimate the cumulative recurrence rate of AF. Univariate regression was used to analyse the unadjusted HR, and variables having a p-value less than 0.20 were then used in a multivariate study to find independent predictors of recurrence of AF. The statistical significance of a 2-sided p-value of 0.05 was found. SPSS software version 23.0 was used for all statistical analyses.
Results
Baseline characteristics
Statistical studies were conducted on 326 patients. Paroxysmal AF was seen in 167 (51.2%) patients and non-paroxysmal AF in 159 (48.8%) patients. Eighty-four (25.8%) patients developed AF throughout the follow-up observation period (average continuation of 14 (interquartile range (IQR): 12–16) months). Their baseline features are listed in Table I. We summarized the application ratio of the ablation strategies in the recurrence and non-recurrence group as follows: CPVI only (31.1% vs. 39.5% in non-paroxysmal AF and 84.4% vs. 87.1% in paroxysmal AF), CPVI + LINE (44.5% vs. 38.6% in non-paroxysmal AF and 10.2% vs. 13.2% in paroxysmal AF), CPVI + CFAE (4.4% vs. 2.3% in non-paroxysmal AF and 2.6% vs. 0.8% in paroxysmal AF), and CPVI + LINE + CFAE (20.0% vs. 19.3% in non-paroxysmal AF and 2.6% vs. 2.5% in paroxysmal AF). Individuals in the recurrence group exhibited significantly greater NT-proBNP levels (all p < 0.001) and larger left diameters (all p = 0.01). In both paroxysmal and non-paroxysmal AF, no additional statistically significant imbalance was observed between recurrence and non-recurrence groups in terms of age, BMI, HTN, HFpEF, previous stroke/TIA, eGFR, LVD, LVEF, and medications such as types of anticoagulant.
Table I
AF recurrence and baseline features
| Characteristics | Non-paroxysmal AF | Paroxysmal AF | ||||||
|---|---|---|---|---|---|---|---|---|
| Total (n = 159) | Recurrence (n = 45) | No recurrence (n = 114) | P-value | Total (n = 167) | Recurrence (n = 39) | No recurrence (n = 128) | P-value | |
| Male, n (%) | 119 (74.8) | 32 (71.1) | 87 (76.3) | 0.49 | 117 (70.1) | 27 (69.2) | 90 (70.3) | 0.89 |
| Age [years] | 59.42 ±10.34 | 59.82 ±9.37 | 59.25 ±10.74 | 0.76 | 60.54 ±11.21 | 60.44 ±11.73 | 60.57 ±11.09 | 0.94 |
| BMI [kg/m2] | 25.92 ±2.98 | 26.07 ±3.04 | 25.13 ±2.61 | 0.43 | 25.58 ±2.91 | 25.08 ±3.04 | 25.73 ±2.86 | 0.23 |
| Smoking, n (%) | 57 (35.8) | 17 (37.8) | 40 (35.1) | 0.75 | 53 (31.7) | 13 (33.3) | 40 (31.3) | 0.16 |
| Drinking, n (%) | 21 (13.2) | 7 (15.6) | 14 (12.3) | 0.58 | 17 (10.2) | 5 (12.8) | 12 (9.4) | 0.53 |
| Hypertension, n (%) | 98 (61.6) | 29 (64.4.) | 72 (63.2) | 0.88 | 93 (55.7) | 23 (58.9) | 70 (54.6) | 0.64 |
| Diabetes mellitus, n (%) | 24 (15.1) | 8 (17.8) | 17 (17.9) | 0.66 | 23 (13.8) | 6 (15.4) | 17 (13.3) | 0.74 |
| Hyperlipidaemia, n (%) | 41 (25.8) | 12 (26.7) | 29 (25.4) | 0.87 | 40 (23.9) | 10 (25.6) | 30 (23.4) | 0.78 |
| CHD, n (%) | 34 (20.4) | 8 (17.8) | 26 (22.8) | 0.49 | 36 (21.6) | 9 (23.1) | 27 (21.1) | 0.79 |
| HFpEF, n (%) | 9 (5.7) | 3 (8.9) | 6 (5.3) | 0.70 | 7 (4.2) | 3 (7.7) | 4 (3.1) | 0.36 |
| Stroke/TIA, n (%) | 17 (10.7) | 6 (13.3) | 11 (0.96) | 0.50 | 17 (10.2) | 5 (12.8) | 12 (9.3) | 0.53 |
| OSAS, n (%) | 15 (9.4) | 5 (11.1) | 10 (8.38) | 0.59 | 16 (9.6) | 4 (10.3) | 12 (9.4) | 0.87 |
| eGFR [ml/min/1.73 m2] | 91 (79–103) | 91 (80–102) | 91 (79–101) | 0.61 | 93 (81–104) | 93 (81–105) | 93 (80–104) | 0.97 |
| Vascular disease, n (%) | 7 (4.3) | 2 (4.4) | 5 (4.2) | 0.62 | 8 (4.8) | 2 (5.1) | 6 (4.7) | 0.59 |
| CHA2DS2-VASc score | 2 [1–3] | 2 [1–3] | 2 [1–3] | 0.23 | 1 [1–3] | 2 [1–3] | 1 [1–3] | 0.32 |
| HAS-BLED score | 1 [1–2] | 1 [1–3] | 1 [0–2] | 0.44 | 1 [0–2] | 1 [0–2] | 1 [0–2] | 0.24 |
| NT-proBNP [pg/ml] | 234.3 [130.4–605.5] | 389.4 [180.4–661.1] | 141.7 [98.6–402.4] | 0.000** | 134.6 [65.8–345.6] | 348.0 [175.1–517.5] | 99.4 [54.3–269.7] | < 0.001** |
| Echocardiographic data: | ||||||||
| LAD [mm] | 37 [33–41] | 40 [35–42] | 36 [33–40] | 0.01** | 36 [34–40] | 38 [35–42] | 36 [33–39] | 0.01** |
| LVD [mm] | 45 [42–48] | 45 [42–48] | 45 [42–48] | 0.82 | 45 [43–47] | 45 [42–46] | 45 [43–48] | 0.10 |
| LVEF (%) | 55 [50–58] | 55 [51–58] | 55 [51–58] | 0.17 | 55 [52–58] | 55 [50–58] | 56 [52–58] | 0.27 |
| Medications, n (%): | ||||||||
| NOAC | 111 (71.1) | 33 (73.3) | 78 (68.4) | 0.21 | 113 (67.6) | 26 (66.6) | 87 (67.9) | 0.10 |
| Amiodarone | 103 (64.80) | 28 (64.4) | 75 (63.0) | 0.92 | 105 (62.9) | 25 (64.1) | 80 (62.5) | 0.86 |
| Propafenone | 30 (18.9) | 10 (22.2) | 20 (17.5) | 0.50 | 32 (19.2) | 8 (20.8) | 24 (18.8) | 0.81 |
| β-blockers | 58 (36.5) | 17 (37.7) | 41 (36.0) | 0.83 | 58 (34.7) | 14 (35.9) | 44 (34.4) | 0.86 |
| ACEI/ARB | 44 (27.7) | 13 (28.8) | 31 (27.2) | 0.54 | 44 (26.3) | 11 (28.2) | 33 (25.8) | 0.86 |
| Sacubitril–valsartan | 10 (6.3) | 3 (6.7) | 7 (6.1) | 9 (4.2) | 2 (5.2) | 7 (5.5) | ||
| Statins | 77 (48.4) | 22 (48.9) | 55 (48.2) | 0.94 | 75 (44.9) | 18 (46.2) | 57 (44.5) | 0.59 |
| Antiplatelet, n (%) | 8 (5.0) | 3 (6.7) | 5 (4.4) | 0.69 | 8 (4.8) | 2 (5.1) | 6 (4.7) | 0.92 |
| Ablation procedures, n (%): | ||||||||
| CPVI only | 59 (37.1) | 14 (31.1) | 45 (39.5) | 0.96 | 141 (84.4) | 34 (87.1) | 107 (83.5) | 0.67 |
| CPVI + LINE | 64 (40.3) | 20 (44.5) | 44 (38.6) | 0.49 | 20 (12.0) | 3 (10.2) | 17 (13.2) | 0.41 |
| CPVI + CFAE | 5 (3.1) | 2 (4.4) | 3 (2.3) | 0.62 | 2 (1.2) | 1 (2.6) | 1 (0.8) | 0.42 |
| CPVI + LINE + CFAE | 31 (19.5) | 9 (20.0) | 22 (19.3) | 0.58 | 4 (2.4) | 1 (2.6) | 3 (2.5) | 0.92 |
| Periprocedural complications, n (%): | ||||||||
| Cardiac tamponade | 1 (0.6) | 0 (0) | 1 (0.8) | 0.36 | 2 (1.2) | 1 (2.6) | 1 (0.8) | 0.42 |
| Perioperative stroke | 1 (0.6) | 1 (2.2) | 0 (0) | 0.28 | 1 (0.6) | 0 (0.0) | 1 (0.8) | 1.0 |
| Pseudoaneurysm | 1 (0.6) | 0 (0.0) | 1 (0.8) | 1.0 | 1 (0.5) | 0 (0.0) | 1 (0.8) | 1.0 |
| Long-term stroke | 1 (0.6) | 1 (2.2) | 0 (0.0) | 0.28 | 1 (0.5) | 1 (2.6) | 0 (0.0) | 0.23 |
BMI – body mass index, CHD – coronary heart disease, HFpEF – heart failure with preserved ejection fraction, TIA – transient ischaemic attack, OSAS – obstructive sleep apnoea syndrome, eGFR – estimated glomerular filtration rate, CHA2DS2-VASc – congestive heart failure (CHF), hypertension (HT), age ≥ 75 years [doubled], diabetes mellitus (DM), stroke [doubled], vascular disease, age 65–74 years, sex category [female], HAS-BLED – elderly, alcohol use, history of impaired kidney or hepatic performance, history of hypertension, stroke history, history of major bleeding, the usage of medications predispose to bleeding, an international normalized ratio that is labile, LVEF – left ventricular ejection fraction, NT-proBNP – N-terminal pro-brain natriuretic peptide, ARB – angiotensin receptor blocker, LAD – left atrial diameter, LVD – left ventricular diameter, ACEI – angiotensin-converting enzyme inhibitors, NOAC – new oral anticoagulants, CPVI – circumferential pulmonary vein, LINE – left atrial roof, floor, superior vena cava, cavotricuspid isthmus, or mitral isthmus, CFAE – complex fractionated atrial electrography.
Correlations between NT-proBNP and baseline characteristics
Results from Spearman’s rank correlation analysis (Table II) revealed that NT-proBNP was positively linked with age (r = 0.25, p < 0.001), non-paroxysmal AF (r = 0.32, p = 0.003), CHA2DS2-VASc score (r = 0.14, p = 0.03), HAS-BLED score (r = 0.12, p = 0.02), and LAD (r = 0.19, p = 0.01). NT-proBNP was negatively associated with male sex (r = –0.18, p = 0.003) and LVEF (r = –0.21, p < 0.001).
Table II
Correlation between NT-proBNP and patients’ other characteristics.
| Variables | Correlation coefficient (r) | P-value |
|---|---|---|
| Age | 0.25 | 0.000** |
| Gender (male) | –0.18 | 0.003** |
| BMI | 0.04 | 0.51 |
| Smoking | 0.02 | 0.75 |
| Non-paroxysmal AF | 0.32 | 0.003** |
| Hypertension | 0.04 | 0.48 |
| Diabetes mellitus | 0.02 | 0.72 |
| CHD | 0.08 | 0.13 |
| CHA2DS2-VASc score | 0.14 | 0.03* |
| HAS-BLED score | 0.12 | 0.02* |
| LAD | 0.19 | 0.01** |
| LVD | 0.09 | 0.14 |
| LVEF | –0.21 | 0.000** |
| ACEI/ARB | –0.03 | 0.64 |
AF – atrial fibrillation, CHA2DS2-VASc – Congestive heart failure (CHF), Hypertension (HT), Age ≥ 75 years (doubled), Diabetes mellitus (DM), Stroke (doubled), Vascular disease, Age (65-74 years), Sex category (female), HASBLED – history of hypertension, elderly, history of stroke, history of impaired renal or liver performance, alcohol use, medication use predisposing to bleeding, history of major bleeding, Labile international normalized ratio, LAD – left atrial diameter, LVD – left ventricular diameter, LVEF – left ventricular ejection fraction, NT-proBNP – Nterminal pro-brain natriuretic peptide.
Baseline NT-proBNP predictive value
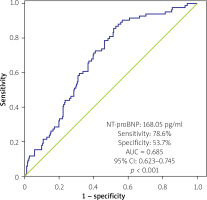
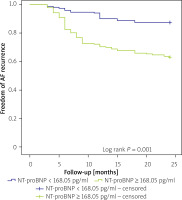
The baseline NT-proBNP cut-off point to forecast AF recurrence was 168.05 pg/ml, according to the results of the ROC curve analysis (sensitivity: 78.6%, specificity: 53.7%). Figure 2 showed the AUC value of 0.65 (95% CI: 0.62–0.74, p < 0.0010. For a cut-off value ≥ 168.05, the 326 individuals were separated into 2 subgroups. The high NT-proBNP group presented a considerably shorter event-free time than the low NT-proBNP group (18.4 vs. 22.2 months, log-rank p = 0.001).
Recurrence of AF in multivariate and univariate analysis
Table III showed the outcomes from multivariate and univariate Cox proportional hazards regression examination for AF recurrence. NT-proBNP ≥ 168.05 pg/ml, LAD, and LVEF were significantly related to AF recurrence in the univariate regression examination. However, in the multivariate regression investigation, forward stepwise analysis adjusted by age, BMI, NT-proBNP ≥ 168.05 pg/ml, LAD and LVEF, NT-proBNP ≥ 168.05 pg/ml (HR = 2.89, 95% CI: 1.71–4.90, p < 0.001), and LAD (HR = 1.05, 95% CI: 1.01–1.09, p = 0.010) were independent predictors for post-ablation AF recurrence AF after RFCA (Figure 3).
Table III
Recurrence of AF in multivariate and univariate analysis
| Variables | Univariate analysis | Multivariate analysis | ||||
|---|---|---|---|---|---|---|
| HR | 95% CI | P-value | HR | 95% CI | P-value | |
| Age [years] | 1.01 | 0.98–1.02 | 0.91 | |||
| Sex (male) | 1.22 | 0.76–1.96 | 0.40 | |||
| BMI [kg/m2] | 1.05 | 0.98–1.12 | 0.11 | |||
| Smoking | 1.06 | 0.68–1.65 | 0.78 | |||
| Paroxysmal | 1.13 | 0.73–1.73 | 0.57 | |||
| Non-paroxysmal | 1.07 | 0.61–1.87 | 0.80 | |||
| Diabetes mellitus, n (%) | 1.23 | 0.71–2.12 | 0.45 | |||
| CHD, n (%) | 1.37 | 0.82–2.29 | 0.22 | |||
| NT-proBNP ≥ 168.05 pg/ml | 3.39 | 2.03–5.65 | 0.000** | 2.89 | 1.71–4.90 | 0.000** |
| LAD [mm] | 1.07 | 1.033–1.11 | 0.000** | 1.05 | 1.01–1.09 | 0.010* |
| LVD [mm] | 0.98 | 0.93–1.04 | 0.65 | |||
| LVEF (%) | 0.94 | 0.90–0.99 | 0.03* | |||
| Additional ablation | 1.33 | 0.83–2.14 | 0.23 | |||
Discussion
This study assayed the predictive significance of pre-procedural plasma NT-proBNP level in AF recurrence in 326 patients who had their initial RFCA and noticed the following: (1) 45 (28.3%) patients in paroxysmal AF while 39 (23.4%) patients in paroxysmal AF agonized AF recurrence throughout the median continuation duration of 14 (IQR: 12–16) months. (2) Baseline NT-proBNP was considerably elevated in individuals with AF recurrence compared to those who persisted in sinus rhythm. (3) A high pre-ablation level of NT-proBNP ≥ 168.05 pg/ml was a prognostic factor of recurrence of AF following RFCA.
Catheter ablation has become an indispensable treatment option for AF. Unfortunately, AF recurrence frequently outweighs the effectiveness of ablation. Identification of the predictors, including a blood/serum biomarker of AF recurrence following RFCA, is therefore critical for sensing high-risk patients and can help physicians to make patients aware of the risk-benefit evaluation, assist the manipulator in selecting the most appropriate healing approach, and implement individualized management plans [14]. NT-proBNP is an inactive prohormone, has a prolonged half-life, increased concentrations, and is more detectable. According to prior research, NT-proBNP is predominantly produced in an atrium in AF patients and there is a direct connection between elevated NT-proBNP levels and AF load [15]. The Cardiovascular Health Study has shown that elevated NT-proBNP is a marker of substantial risk for atrial fibrillation in a community-based population of older adults [16]. However, attempts at determining a value for the prediction of baseline NT-proBNP for AF recurrence after RFCA have yielded contradictory results [8, 17, 18]. This might be related to the fact that: (1) overestimation of effect sizes is caused by a scarcity of standardized measuring units and an inability to distinguish natriuretic peptides [18] and (2) NT-proBNP would be influenced by several mechanisms containing age, gender, renal function, LA, and LV stretch [19]. A study by Lau et al. demonstrated that women had a greater baseline level of NT-proBNP [20]. We detected that baseline NT-proBNP was positively correlated with age, non-paroxysmal AF, and LAD, and it was negatively correlated with male gender and LVEF. The above findings in our research are concordant with the reported studies [15, 19–22].
Previous studies have demonstrated that structural remodeling in atrial fibrosis is the risk factor for the occurrence and persistence of AF. Fibrosis is caused by a rise in defective extracellular matrix deposition, leading to atrial conduction abnormalities and AF [23]. As for recurrent arrhythmia after ablation, increased NT-proBNP might be an independent predictor of AF recurrence [24, 25]. In the current literature, we put variables into the multivariable proportional hazards regression model, a higher baseline NT-proBNP level (≥ 168.05 pg/ml) were independent predictors for AF recurrence. We speculated an explanation as follows: Firstly, an atrial stretch that is followed by an increased central volume loading caused by a lack of atrial contraction and an abnormally accelerated ventricular rhythm, leading to LA enlargement and stimulating the secretion of natriuretic peptides [26]. BNP elevation is linked to a greater left atrial size, which may increase the likelihood of AF recurrence after ablation due to atrial fibrosis and remodeling [27]. Secondly, a high baseline NT-proBNP level might be the consequence of poor cardiac function in the absence of inflammation, which may cause AF due to elevated myocardium fibrosis and lowered conduction velocity [28, 29]. Thirdly, elevated plasma BNP and its expression of a gene in heart tissues may reduce resting sympathetic activity while increasing vagal activity via the cGMP route. Lastly, it contributed to the advancement and preservation of AF [30, 31]. NT-proBNP is often easier to detect by biomedical tests than changes in heart structure observed by imaging investigations, considering the advantage of slower fluctuations. To predict AF recurrence following RFCA, baseline NT-proBNP levels could be employed as a more sensitive measure than cardiac structural alterations.
An amusing discovery in our study is that there was no significant difference in the recurrence ratio between patients with non-paroxysmal and paroxysmal AFs (28.3% vs. 23.4%, p = 0.31), which is concordant with the Substrate and Trigger Ablation for Reduction of Atrial Fibrillation Trial Part II (STAR AF II) [32]. This may be due to our performance of additional ablation procedures, if necessary, and non-advanced atrial substrate in our non-paroxysmal AFs.
Limitations of the research investigation: Some restrictions to our investigation would be deliberated: (1) Our first limitation was that this was a retrospective study performed at a single location using a small sample size. (2) The heart rhythm and rate at the time of baseline NT-proBNP detection was not recorded, even though most subjects were asymptomatic. (3) Although we did take account of the individuals with HFpEF, but we did rule out the possibility of HFrEF and renal insufficiency, which can also lead to an increase in NT-proBNP levels. (4) The recurrence rates may have been underestimated due to the ECG and intermittent Holter monitoring.
Conclusions
The present findings suggest that a greater baseline NT-proBNP level might predict AF recurrence in individuals receiving RFCA. This discovery may help improve the stratification of candidates for atrial fibrillation ablation in clinical practice. Considering the limitations described above, further well-designed, larger, long-term follow-up, and multi-centre prospective studies are required to verify these results.