Introduction
Cancer incidence in 2040 will increase by 47% compared to 2020. Lung cancer is the leading cause of death from malignant neoplasms in both men and women. Therefore, diagnosis and treatment of early lung cancer through screening programs and prediction of recurrence are essential for lung cancer mortality reduction [1, 2]. Surgery is the primary treatment method for non-small cell lung cancer [3]. However, the disease recurs in about 30% of patients during the first five years. The recurrence-free period lasts on average 15.7 months [4]. According to National Cancer Comprehensive Network (NCCN) standards, adjuvant chemotherapy is recommended for stage IB–IIIA lung cancer. There is no need to prescribe cytotoxic therapy in each case. It is necessary for the presence of risk factors such as tumor size greater than 4 cm, vascular invasion, poorly differentiated tumor, marginal resection, unknown status of regional lymph nodes, and visceral pleural lesions. Mandatory chemoradiation therapy is recommended for patients with N2 [5].
In the daily routine practice at the preoperative stage, one should collect the patient’s history of bad habits, chronic diseases, and social status, and additionally analyze the histology of the tumor and its differentiation. Immunohistochemistry and molecular biology for the presence of the most common mutations (epidermal growth factor receptor – EGFR, ALK, B-RAF, and ROS1) should be performed after surgical treatment [6].
There are a few methods for identifying factors related to recurrence following surgery. The first are clinical parameters and TNM (T: tumor size, N: metastases in regional lymph nodes, M: distant metastases) classification. Furthermore, the physical examination is very important, because a lower performance status and the presence of symptoms are unfavorable prognostic factors for disease-free survival [7]. An extensive pathological investigation is also essential because histological differentiation, vessel invasion, lymphatic permeation, and pleural invasion have all been reported to be poor prognostic factors for disease-free survival [8]. The other method for predicting recurrence uses molecular biological techniques. Lung cancer is a highly aggressive disease that includes different histological subtypes with distinct clinicopathological and molecular features. Therefore, the establishment of biological markers is necessary to predict disease recurrence. Immunohistochemistry is now widely used to determine the prognosis in patients with breast cancer [9], lung cancer [10], lymphoma [11], and many other types of tumors.
Material and methods
Study design
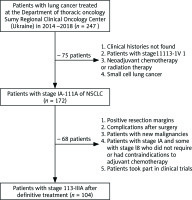
There were 104 patients selected for performing the retrospective study. They received definitive treatment according to NCCN standards [5]. The treatment was carried out in the Sumy Regional Clinical Oncology Center (Ukraine) during 2014–2018. Exclusion criteria were neoadjuvant chemotherapy or radiation therapy, positive resection margins, and the appearance of a new malignancy. The study group did not include patients with stage IA and some with stage IB who did not require or had contraindications to adjuvant chemotherapy. Patients with grade 3 or more complications after surgery, such as air leakage, atelectasis, pneumonia, chylothorax, arrhythmia, bronchopleural fistula, and cerebral infarction, were excluded from the study (Fig. 1).
Data on age, sex, smoking status, category T, category N, primary tumor size, stage of disease, lymphovascular invasion, histological variant, histological differentiation, marital status, and Eastern Cooperative Oncology Group performance score were taken from the medical records. Immunohistochemistry was performed using tumor tissue obtained from postoperative material. According to age at the time of diagnosis, patients were divided into two groups: ≤ 60 years and > 60 years. Disease stages, categories T and N, were determined according to the eighth edition of the TNM classification [12]. According to the histology and histological differentiation, patient distribution was performed based on World Health Organization data on the classification of lung tumors (2015) [13].
Histology
Paraffin blocks of lung cancer were taken from the archives of the Sumy Regional Clinical Oncology Center. Serial sections with a thickness of 4–5 µm were stained with Mayer’s hematoxylin and eosin.
Immunohistochemistry
Serial sections with a thickness of 4 µm were applied to the SuperFrost adhesive slides (Thermo Scientific, USA). The deparaffined sections were subjected to antigen unmasking by heat treatment in 0.1 M citrate buffer (pH 6.0) at 95–98°C. The Ul-traVision Quanto Detection System HRP Polymer (Thermo Scientific, USA) was used to visualize the immunohistochemical (IHC) reaction. Endogenous peroxidase activity was blocked by 3% hydrogen peroxide. Ultra V Block was used to prevent non-specific reaction and background staining. The reaction was amplified with Primary Antibodies Amplifier Quanto. Diaminobenzidine was used as a chromogen. The nuclei were counter-stained with Mayer’s hematoxylin. Primary antibodies against PD-L1 (Master Diagnostica, clone Cal-10, dilution 1 : 50), EGFR (Thermo Scientific, clone EP38Y, dilution 1 : 100), p53 (Master Diagnostica, clone SP5, dilution 1 : 50), Ki-67 (Thermo Scientific, clone SP6, dilution 1 : 200) and ALK (Abcam, clone ab37807, dilution 1 : 100) were used. At least six different fields of view (FOV) with a diameter of 1 mm were analyzed for each sample. The immunohistochemical results were presented as the mean number of positive cells per field of view.
In cases of positive staining for PD-L1, a clear membrane response was detected in tumor cells with a complete or partial cytoplasmic environment of any intensity (Fig. 1). In our calculations, we did not consider a positive reaction in the immune cells of the tumor microenvironment (lymphocytes, macrophages). We also ignored the cytoplasmic response to PD-L1 in tumor cells.
Epidermal growth factor receptor-immunoreactivity was scored based on membranous and/or cytoplasmic staining (Fig. 1), as follows: 0, no staining or faint staining in < 10% of tumor cells; 1+, weak staining in 10% of tumor cells; 2+, moderate staining in 10% of tumor cells; 3+, strong staining in 10% of tumor cells [14].
A positive result of ALK staining of lung tumor tissue samples was positive cytoplasmic staining of tumor cells, often in the form of lumps inside the cytoplasm and/or with a membrane component (Fig. 1). The classical H-score scale with a graduation of 0–3 with the multiplication of the percentage of positive-stained cells (range 0–300) was used for evaluation. A positive reaction was considered as 200–300 (+3) [15].
The p53 protein was found in the nuclei of lung tumor cells and was more often combined with poorly differentiated tumors with pronounced manifestations of cellular atypia (Fig. 1). Evaluation of p53 was performed by counting the percentage of stained cells in 6 different FOV with a diameter of 1 mm. Ki-67 also had nuclear localization in the case of positive staining of tumor cells (Fig. 1). The evaluation of immunohistochemistry results was similar to that for p53 protein.
Follow-up of patients after surgical treatment and adjuvant chemotherapy
Patients were monitored according to local practice. The first computed tomography was performed one month after surgery. Subsequently, this examination was repeated every three months for the first three years, then every six months for two years. After five years after the operation, computed tomography was performed once a year. Magnetic resonance imaging of the brain, ultrasound of the neck, abdominal cavity, pelvis, and bone scans were performed as needed in case of suspicion of recurrence of the disease.
In case of recurrence of the disease, all metastases were divided into two groups: locoregional and distant. Locoregional metastases appeared in resection margins and mediastinal lymph nodes on the affected side. All other metastases were considered distant.
We calculated the recurrence-free survival of patients. The recurrence-free period was considered from diagnosis to the registration of radiological disease progression.
Ethics committee
The study was performed retrospectively. We adhered to the Declaration of Helsinki and received the approval of the Local Ethics Commission of the Sumy Regional Clinical Oncology Center.
This research has been performed with the financial support of grants from the Ministry of Education and Science of Ukraine No. 0112U100471 “Condition of mineralized tissues using new composites with Ag+ and Cu2+ nanoparticles”.
Statistical analysis
Cox regression was used to model the survival of patients with lung cancer. The main advantage of this model is that it allows one to work with categorical and censored data. In general, the model can be presented as follows:
h (t) = h0 (t) * exp (β1 * ×1 + β2 * ×2… + βp * ×p), where h0 (t) is the base risk; β1,…, βp-coefficients; ×1, ×2,…, ×p are independent variables (so-called predictors). The coefficients β1,…, and βp describe the degree of influence of each variable on the probability of occurrence of the event. Thus, when the variable xp increases by 1, the risk increases exp (βp) times. This is provided that the value of other predictors has not changed.
The standard data model was created in Excel, and the analytical model was created in the software environment IBM SPSS Statistics 27. All calculations were also made in the software environment SPSS. The risk factors were significant when the two-sided p-value was less than 0.05. The Kaplan-Meier method was used to estimate the recurrence-free survival of patients.
Results
Study design
Baseline characteristics of patients and the expression level of IHC markers are presented in Table 1.
Table 1
Characteristics of patients and levels of expression of immunohistochemical markers
Histology
All lung cancer tissue samples belonged to non-small-cell lung carcinoma. According to histological types, 62 cases of squamous cell carcinoma, 34 cases of adenocarcinoma, and 8 cases of large cell carcinoma and adenosquamous were detected in the study group.
Squamous cell lung cancer is represented by solid areas and layers of squamous epithelium with cellular atypia: polymorphism of cells and nuclei, hyperchromatic nuclei, and pathological mitoses. A characteristic feature of differentiated forms of squamous cell carcinoma was keratin in the form of rounded horny formations called “cancer pearls”. Squamous cell carcinoma of low differentiation was characterized by the diffuse arrangement of tumor cells without the formation of close connections between cells, the presence of areas of necrosis, and pronounced atypia of monomorphic tumor cells.
Adenocarcinoma was characterized by atypical glandular structures that infiltrated interstitial lung tissue (Fig. 2). Tumor cells made up of glandular and pseudoglandular structures showed signs of cellular atypia. Some tumors had a papillary pattern and contained mucin.
Fig. 2
Histology and immunohistochemistry of lung cancer tissue
ALK – immunohistochemical (IHC) expression of ALK, EGFR – IHC expression of epidermal growth factor receptor, H&E – hematoxylin-eosin staining, Ki-67 – IHC expression of Ki-67, PD-L1 – IHC expression of PD-L1, p53 – IHC expression of p53, The figures in the inserts correspond to the enlarged area of this sample. Magnification is indicated in the lower left corner of the image as a marker.
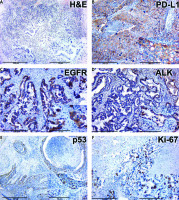
Large cell lung cancer consisted of large polygonal cells with vacuolated nuclei and distinct cellular atypia. Adenosquamous cancer contained signs of squamous cell carcinoma and adenocarcinoma.
Follow-up of patients after surgical treatment and adjuvant chemotherapy
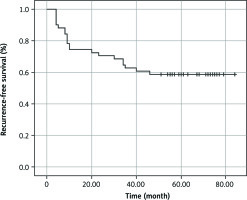
Among the 104 patients included in the study, 42 had stage IB disease. They all received adjuvant chemotherapy because they had one or more risk factors, such as a primary tumor larger than 4 cm, lymphovascular invasion, poor tumor differentiation, or visceral pleural lesions. Stage II and III non-small cell lung cancer were diagnosed in 46 and 16 patients, respectively. They also received adjuvant chemotherapy. Among all patients, 14 had N2, so according to NCCN standards, they received chemoradiation therapy. During the follow-up period, recurrence of the disease occurred in 42 (40.4%) patients, and 38 (90.5%) of them died. Another 4 (9.5%) people died of other causes. The median recurrence-free survival was 56.3 months, range 4–84.0 months (95% CI = 46.866–65.683). The recurrence-free survival rate was 58.8% (Fig. 3).
Fig. 3
Kaplan-Meier curve showing recurrence-free survival in patients after definitive treatment of IB-IIIA non-small-cell lung carcinoma
Loco-regional metastases were recorded in 10 (23.8%) patients; 9 (21.5%) had distant metastases to the lungs, 7 (16.7%) to the kidneys, 4 (9.5%) in the bones, 4 (9.5%) in the lymph nodes of the neck, 4 (9.5%) in the liver, and 4 (9.5%) in the brain.
Disease recurrence and death from the underlying condition occurred in 14 patients with N2. This group of patients most often had lung metastases and local recurrences – 4 (28.7%) and 3 (21.4%). Metastases to the brain occurred in 2 (14.3%) persons, to the bones in 2 (14.3%). Another 1 (7.1%) patient had metastases in the neck lymph nodes, 1 (7.1%) in the liver, and 1 (7.1%) in the kidneys.
Statistical analysis
Univariate Cox regression analyses revealed that sex, category T, category N, histological differentiation, and smoking status were significantly associated with disease recurrence. Multivariable Cox regression analyses identified category T, histological differentiation, and smoking status as independent predictors of disease recurrence (Table 2).
Table 2
Univariate and multivariable analyses of clinical-pathological factors and immunohistochemical markers associated with recurrence-free survival
Table 3 presents the recurrence rate of lung cancer with identified independent predictors.
Table 3
Incidence of lung cancer recurrence in patients with identified independent predictors
Discussion
According to GLOBOCAN, the survival rate of patients with lung cancer five years after diagnosis is 10–20%, although, in some countries, the rates are much higher: Israel – 27%, Japan – 33%. These data apply to patients with all stages of the malignant process [16]. Data from the National Cancer Registry show that in Ukraine, the overall 5-year survival of lung cancer patients is close to 25%, which is a tolerable result [17].
The leading cause of death is recurrence of the disease. According to our study, recurrence of the disease after definitive treatment was reported in 40.4% of people, while only 3.8% of patients died from other causes.
The cohort of patients we studied had stage IB–IIIA non-small cell lung cancer. 84.6% of patients had stages I and II, which is entirely consistent with the data of the national cancer registry and our previous study. In contrast to the all-Ukrainian data, early detection of lung cancer (stage I–II) in the Sumy region is much higher (40.23% vs. 22.4%, respectively). This situation leads to decreased detection frequency of advanced stages – 22.1% vs. 35.15%. Apparently, due to the early detection of lung cancer in the population of the Sumy region, the average 1-year survival is better. At the same time, mortality up to 1 year is much lower than the Ukrainian average (46.2% vs. 55.7%) [18].
The use of Cox regression with univariant and multivariable analysis allowed us to identify independent predictors of disease recurrence – category T, histological differentiation, and smoking status – in a study group of 104 people. Each of these factors is confirmed in the scientific literature and our research.
In 2017, the Ontario Evidence-Based Cancer Care Program provided updated guidelines for treating surgically resected stage II–IIIA non-small cell lung cancer patients. Adjuvant chemotherapy has significantly improved overall survival in patients with stage II–IIIA disease [19]. Already in 2021, such a pattern was observed for the IB stage. Therefore, in the case of a primary tumor size ≥ 4 cm, adjuvant chemotherapy is recommended [20].
During our study, one of the critical predictors of recurrence of non-small cell lung cancer was category T, which we determined according to the eighth edition of the TNM classification. Category T does not depend only on the size of the primary tumor. Its formation is influenced by the spread of the primary tumor to structures such as the tracheal bifurcation, parietal and visceral pleura, chest, diaphragmatic nerve, pericardium, diaphragm, mediastinum, heart, large vessels, trachea, recurrent laryngeal nerve, esophagus, and vertebral body and the appearance of individual tumor nodes in the same lobe or an adjacent lobe of the lung. According to the TNM classification, category T is one of the prognostic factors associated with the primary tumor [12].
Some scientists have compared the survival of patients in whom tumors belonged to one category T. The first group included the patients according to the size of the primary tumor, and the second group included the patients according to the spread to neighboring structures. It was found that the overall survival of patients in both groups was almost the same. That is why it is essential to evaluate the T category, not just the size of the primary tumor [21]. Category T is recognized as an independent factor in the prognosis of cancer recurrence of the gallbladder, vulva, thyroid, and breast [22–25].
Smoking is the leading cause of lung cancer and another independent predictor of relapse. The risk of developing a malignant lung tumor in smokers is 20 times higher than in non-smokers [26].
Researchers have found that the risk of lifelong lung cancer in women who smoke is 11.6%, and among men it is 17.2%. The rates are much lower among those who have never smoked – 1.4% and 1.3%, respectively [27]. Passive smoking is also dangerous. According to some data, smoke inhaled from the air is even more harmful [28].
At the same time, smoking cessation helps to restore the bronchial mucosa. Patients who smoke at the time of diagnosis have a worse prognosis than those who quit the habit a few years ago. People who have never smoked have better treatment outcomes than smokers [29].
According to our study, recurrence of the disease was recorded in 43.9% of patients who smoked and among 27.3% who had never smoked. This situation reflects global trends. At the time of lung cancer diagnosis, about 86% of patients quit smoking. But after a month, most of them return to the harmful habit. Within the first year after definitive lung cancer treatment, 60% of patients return to the habit. Instead, people who have abstained from smoking for six months continue to have a healthy lifestyle [30].
One of the most significant studies of this problem was conducted by Rachel et al. between 2010 and 2016 on 1,124 people who received radical treatment for non-small cell lung cancer. Thus, the 1-year survival rate among smokers was 77%, among former smokers 60%, and among current smokers 57%. After adjusting for disease, age, ECOG, and gender, the former and current smokers were more likely to die within one year. The risk of death was 25% lower among those who quit smoking [31]. Among the contingent of people who managed to refrain from smoking for more than a year, married women with a high level of education predominated [32].
Histological differentiation is an independent predictor of lung cancer recurrence. This risk factor may indicate the aggressiveness of cancer, sensitivity to drug and radiation therapy, and susceptibility to metastasis and recurrence. Lung cancer tumor aggressiveness and natural growth pattern play a role in prognosis [33].
The relationship between patient survival and the histological differentiation of the tumor was studied for squamous cell carcinoma of the cervix [34]. Poorly differentiated tumors have been significantly associated with older age, larger tumor size, and lymph node metastasis. In multivariable analysis, poorly differentiated tumors are associated with low patient survival compared to moderate- and well-differentiated tumors.
The significance of histological differentiation as an independent risk factor for lung cancer recurrence was proved in a large cohort study between 1992 and 2007. Patients with stage I lung cancer with a low degree of tumor differentiation, vascular invasion, or visceral pleura have lower 5-year recurrence-free survival rates than patients with stage II lung cancer but without risk factors (63% vs. 83%, respectively) [35].
Our study traces the relationship between the histological differentiation of the tumor and the incidence of lung cancer recurrence. Thus, recurrence occurred in 20% of patients with well-differentiated tumors, 26.4% with moderately differentiated tumors, and 92.3% with poorly differentiated tumors. 75% of patients had well- and moderately differentiated tumors in the study cohort.
Immunohistochemistry played one of the leading roles in determination of the individual biological properties of tumors. However, our research and the results of research by other scientists show that only a combination of several markers can predict the prognosis of the disease. We consider this to be one of the reasons why the model did not help predict disease recurrence. A literature review suggested that every single biomarker cannot be an independent predictor of relapse-free or overall survival. Nevertheless, correlation with the category TNM has often been observed [36].
Folescu et al. found that patients with high Ki-67 expression were more likely to have advanced stages and a low degree of histopathological gradation of the tumor. The same study revealed that co-overexpression of Ki-67 and EGFR increases the metastatic potential of the tumor. These factors lead to low recurrence-free and overall survival. Due to the small selection, the results could not be statistically significant but could justify more detailed studies with a large cohort of patients [37].
Woodard et al. discovered that high PD-L1 expression is associated with low overall survival in patients with lung adenocarcinomas. At the same time, PD-L1 has no significant effect on disease-free recurrence [38].
In their study, Scagliotti et al. observed an association between overexpression of Ki-67, p53, and a high risk of recurrence in patients with lung cancer. It is worth noting that a high level of expression of a single biomarker did not allow a poor prognosis to be suspected [39, 40].
Conclusions
Category T, histological differentiation, and smoking status are independent predictors of recurrence in patients with surgically resected non-small-cell lung cancer. Many investigations have studied the influence of biomarkers on disease prognosis in patients with lung cancer. However, it is essential to highlight that most information was collected in small geographical areas involving small cohorts of patients. As a result, research data may differ significantly. As we can see, most scientists believe that only a combination of several markers can predict the prognosis of the disease. We consider that the main reasons biological markers (PD-L1, Ki67, p53, EGFR, and ALK) did not help the model predict the disease recurrence was a small cohort of patients and their consideration as separate predictors. At the same time, for statistical reliability, it is necessary to conduct a study on a larger cohort of patients and compare the mutual influence of several biomarkers.