Introduction
Gastroesophageal varices are detected in approximately 50% of patients with liver cirrhosis. Their presence correlates with the severity of liver disease. Although only 40% of Child-Pugh class A patients have varices, about 85% of Child-Pugh class C patients have varices [1, 2]. Variceal hemorrhage occurs at a yearly rate of 5-15%, and the 6-week mortality after hemorrhage is about 20% [3, 4]. In general, variceal bleeding ceases spontaneously in 40-50% of patients, but the incidence of early re-bleeding ranges between 30% and 40% within the first 6 weeks after the attack. Around 40% of all re-bleeding episodes occur within the first 5 days [5, 6]. Gastric varices (GVs) bleed less frequently than esophageal varices (EVs) and are responsible for 10-30% of all variceal hemorrhages [7]. However, gastric variceal bleeding tends to be more severe than esophageal with a greater need for blood transfusion and a higher mortality [8-10].
Gastric varices are frequently associated with EVs, either as an isolated GV or as an extension of an EV. Extension on the lesser curvature is known as gastroesophageal varices type 1 (GOV1), and extension into the fundus is known as gastroesophageal varices type 2 (GOV2) [8]. The 2-year incidence of bleeding of GOV2 is > 50% [8]. The incidence of bleeding may be associated with the severity of liver disease or risky signs of bleeding over the varices [11]. Primary prophylaxis of EVs is well established with esophageal band ligation (EBL) for high-risk EVs with or without β-blockers [12]. In patients with GOV2 and high-risk EVs, it is not known whether EBL without intervention for gastric extension can increase the chance of bleeding from GVs and PHG.
The management of GVs has not been studied as well as EVs, and both evaluation and treatment remain controversial [13]. Cyanoacrylate injection for rapid control of variceal bleeding was originally reported by Soehendra et al. [14]. This method has been suggested for treatment of GV bleeding by the American Society for Gastrointestinal Endoscopy [15], the Baveno consensus [12], and the National Institute for Health and Clinical Excellence guideline [16]. Whether or not cyanoacrylate can be used as a primary prophylaxis for GVs has not been established. The Baveno VI criteria recommended that more studies be conducted in this area [12]. The study aim was to evaluate the efficacy of cyanoacrylate injection for primary prophylaxis of GOV2 associated with a high risk of bleeding EVs before EBL.
Material and methods
This randomized parallel controlled trial included 75 patients with liver cirrhosis and bleeding or high risk of bleeding EVs associated with GOV2, which had not previously bled or been treated by injection. The patients were enrolled consecutively from the Gastroenterology Department of Benha University Hospital and Ahmed Maher Teaching Hospital from November, 2018 to December, 2019. This study was approved by the Benha Faculty of Medicine Institutional Review Board and Ethics Committee (approval number: PACTR202008491799224).
Bleeding EVs were identified by the presence of an esophageal bleeding spurter, a recent clot over the bleeding site, oozing from the varices, or the white nipple sign. High-risk EVs were identified as grades II-IV according to De Franchis (2005) [17] or the presence of signs of impending rupture or red color signs according to Beppu et al. (1981) [18]. Red color signs refer to dilated small vessels or microtelangiectasia on the variceal surface and are subdivided into cherry red spots, red wale markings, hemocystic spots, or diffuse redness.
Patients with upper gastrointestinal bleeding were vitally stabilized before being asked if they were willing to enroll in the study if they were eligible after the endoscopic examination. Eligible patients who were screened for EVs were asked to enroll in the study. Patients who refused to be enrolled in the study, had active or previous bleeding from GVs, had expected impending bleeding from GVs, and who had any contraindication to β-blockers (bronchial asthma, bronchospasm, severe peripheral arterial disease, including Raynaud’s syndrome, slow heart rate, or uncontrolled heart failure), were already on β-blockers before presentation, or had hepatic encephalopathy were also excluded.
The patients were previously randomized by a computer program to 3 groups by block randomization (n = 25 each). The random allocation remained concealed from the investigator in an opaque envelope until endoscopic examination was performed and the patient was confirmed to be eligible for the study, after which the envelope was opened to allocate the patient.
In group A, the GVs were injected with cyanoacrylate mixed 1 : 1 with Lipidol. The number of ampoules was decided according to the varix size with an attempt to solidify the varix in one session. The EVs were banded in the same session. The number of bands was decided according to the number, grade, and extension of the varices. The patients were started on carvedilol 48 hours after the procedure or successful initial hemostasis if they came in with active esophageal variceal bleeding. Carvedilol was given in the form of an oral tablet with an initial dose of 6.25 mg daily, which was titrated according to heart rate and blood pressure each week. The target was a 25% decrease in heart rate from baseline provided that the systolic blood pressure did not decrease below 90 mmHg and heart rate did not decrease below 60.
In group B, patients underwent EBL and started carvedilol without GV injection. In group C, patients underwent EBL only.
During upper GI endoscopic examination, the grade and risk of EVs were reported according to Beppu et al. and De Franchis [17, 18]. The GV extension size was measured according to an endoscopic retrograde cholangiopancreatography cannula calibrated for each 0.5 cm. Large GVs were defined as those ≥ 2 cm. PHG was reported as none, mild, or severe according to Carpinelli et al. (1997) [19].
Follow-up endoscopy appointments were set each month from the index endoscopy or until the end point. The end point was identified as eradication of EVs in all groups, with complete obturation of GVs in group A, upper GI bleeding, or death. During follow-up endoscopy, testing for complete obturation of GVs and cyanoacrylate injection if indicated was performed in group A. The size of GVs was measured in all groups, and signs of impending rupture or not were reported. The grades of EVs and PHG, if identified, were also reported. Follow-up was performed clinically or by phone contact with patients who refused to continue for endoscopic follow-up or those who did not show up so that we could determine the incidence of bleeding or mortality. Follow-up was continued for ≥ 24 weeks after enrollment of the last patient.
All patients included in this study also were subjected to full history taking, clinical examination, and laboratory investigations, including complete blood cell count, liver profile tests, prothrombin time and activity determined by the international normalized ratio, and serum creatinine. The Model for End-Stage Liver Disease (MELD) score and Child-Pugh classification according to the modified Child-Pugh criteria [20] were calculated. Abdominal ultrasonography was also performed and reported.
Statistical methods
Sample size was calculated on the basis of the estimated incidence rate of bleeding in GOV2 as 55%. A minimum sample size of 21 patients was needed in each group to detect a change from 55% to 20% with a confidence of 90% and 80% power of the study. Comparisons among the 3 treatment groups were made by performing the chi square test for dichotomous or categorical variables and the ANOVA test for continuous variables. Non-parametric tests were used when indicated. Outcomes were identified as bleeding, death, and change in GVs, EVs, or PHG. Test variables were considered as pre-treatment clinical, laboratory, ultrasonographic, and endoscopic parameters. Mortality was reported as percent and per person-year. Survival analysis was performed to compare the 3 treatment groups regarding the time free of bleeding. Because bleeding was found in a sufficient number of patients, univariate and multivariate analysis was performed to identify independent predictors of bleeding.
Results
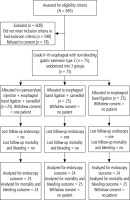
Between November, 2018 and December, 2019, 683 patients with liver cirrhosis were assessed for eligibility criteria. Among them, 590 patients were excluded because they did not meet the criteria, and 18 patients refused to be enrolled in the study. Figure 1 shows the CONSORT flow diagram of the clinical trial. During the study, one patient withdrew consent after allocation and two patients refused the second endoscopy. Therefore, baseline data were available for only 74 patients and follow-up endoscopic data were available for 72 patients. Table 1 shows a comparison among the groups regarding baseline variables. There were no significant differences in baseline demographic, clinical, laboratory, abdominal ultrasound or endoscopic characteristics among the 3 groups.
Table 1
Comparison of baseline variables among the 3 groups
Out of the 24 patients in group A, 15 (62.5%) patients achieved complete obturation of GVs in one session. Eight (33.6%) patients needed a second session to achieve obturation. Only one patient needed a third session. The median number of cyanoacrylate ampoules needed to achieve obturation was 2 (range, 1-5). The median number of sessions of EBL needed to achieve EV eradication was 3 (range, 2-5).
The mean follow-up duration was 36.7 ±17.9 weeks (1-60 weeks). The total follow-up duration was 2718 weeks (52.3 person years). Table 2 shows a comparison of the study outcomes of GV changes, bleeding, death, and PHG changes among the 3 groups.
Table 2
Comparison of study outcomes among the 3 groups
Gastric varices changes
There was a significant difference in the number of patients showing an increase in GV sizes and risky signs of bleeding among the groups. Group A had no patients (0%), group B had 5/24 (20.8%), and group C had 11/24 (45.8%) showing an increase in GV sizes or presence of risky signs of bleeding (p = 0.005).
Bleeding
During follow-up, 14 (18.9%) out of 74 patients developed upper GI bleeding (26.8/100 person-years). The overall bleeding rate was 2/24 (8.3%) in group A, 6/25 (24%) in group B, and 6/25 (24%) in group C (p = 0.2). The relative risk (RR) of bleeding due to any cause in group A was 0.35, and the RR reduction was 0.67 (67%). The cause of bleeding was GV bleeding in 5 patients (6.8%), post-banding ulcer in 4 (5.4%), PHG in 2 (2.7%) and undetermined source in 3 (4.1%). The distribution of GV bleeding was 1 patient in group A (4.2%), 2 in group B (8%) and 2 in group C (8%). The RR of GV bleeding was 0.5. Prophylactic injection of GV extension decreased the risk of GV bleeding by 47.5%. The two patients who bled due to PHG were in group C and did not receive carvedilol.
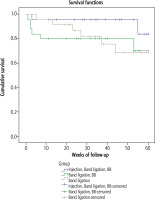
Comparison between the patients who had upper GI bleeding and those without bleeding was performed to identify factors associated with bleeding in all patients (Table 3). In multivariate analysis, the only predictor of bleeding was large gastric extension size (p = 0.03) (Table 4). A survival analysis to test comparison of time free of bleeding among the 3 groups showed that group A had the best result, although the difference was not statistically significant (Fig. 2).
Table 3
Comparison of baseline parameters between bleeders and non-bleeders
Portal hypertensive gastropathy
A decrease in PHG was found in 6/24 (25%) in group A, 6/24 (25%) in group B, and 2/24 (8.3%) in group C (p = 0.5). The groups that received carvedilol had a greater decrease in PHG, although the difference was not statistically significant. PHG was the cause of bleeding in 2 patients, with both in group C.
Discussion
Although the incidence of bleeding was less from GVs than from EVs, the GVs were associated with more severe bleeding and higher mortality. The incidence of bleeding from GVs was greater for isolated GVs, followed by GOV2 [8]. Prophylactic band ligation is the standard of care for management of high-risk EVs; however, management of GOV2 is not well standardized. Although it is a common practice for some endoscopists to inject the gastric extension with cyanoacrylate before EBL to decrease the chance of bleeding, there is no evidence supporting the effectiveness of this practice. In this randomized controlled trial, we evaluated injection of cyanoacrylate into gastric extensions followed by EBL and carvedilol in group A, EBL and administration of carvedilol in group B, and only EBL in group C.
In this study, the 3 treatment groups were comparable in baseline parameters, including size and risk level of EVs, size of gastric extension, and presence and grade of PHG (Table 1). During follow-up, we found that increased gastric extension size or development of risky signs of bleeding was significantly lower in group A than in groups B and C (p = 0.005). The overall bleeding rate and GV bleeding were also lower in group A than in groups B and C (Table 2). We also found that the time free of bleeding with injected gastric extension was longer in group A than in the other 2 groups, but the difference was not statistically significant (p = 0.2) (Fig. 1). Few studies have evaluated prophylactic cyanoacrylate injection for GVs. One of these studies was a single-center study in India published by Mishra et al. [21]. They evaluated the role of prophylactic cyanoacrylate injection for GOV2 (30 patients) or isolated GVs compared with β-blockers (29 patients) and placebo (30 patients). They found that the risk of bleeding from GVs was significantly decreased for prophylactic cyanoacrylate injection relative to the risk for β-blockers and placebo. However, in our study, the rate of bleeding from gastric extension during follow-up was less than that reported by Mishra et al. [21]. This discrepancy may be due to the differences in the studied patients. Most of the patients included in the study by Mishra et al. had high-risk impending rupture GOV2 or isolated GVs. Another study published by Kang et al. evaluated the long-term efficacy and safety of cyanoacrylate injection of GVs. Although they included 127 patients, only 27 were injected prophylactically, and the other 100 were injected after bleeding. They reported that no treatment complications were found in those 27 patients during the follow-up period [22]. In our study, we found that the RR of bleeding in group A was 0.35, and the RR reduction was 0.67 (67%). This finding means that prophylactic cyanoacrylate injection of GV extensions decreased the risk of bleeding by 67% relative to the risks in the other 2 groups. The RR of GV bleeding was 0.5, and prophylactic injection into GV extensions decreased the risk of GV bleeding by 47.5%. We also found that the number needed to treat is 6.25, which means that treating 6 patients with injection of gastric extension with cyanoacrylate will prevent 1 patient from bleeding.
Regarding the prediction of bleeding in patients with GOV2, we found that a large GV extension of > 2 cm was an independent predictor of bleeding (Table 4). Mishra et al. also reported that a large GV extension of > 2 cm, a MELD score > 16, and PHG were independent predictors of bleeding [21]. Our data support the use of prophylactic cyanoacrylate injection into gastric extensions in combination with EBL to reduce the risk of variceal bleeding, especially in patients with GV extensions of > 2 cm.
Non-selective BBs have been recommended for primary prophylaxis of bleeding from medium or large EVs. Carvedilol is known to cause a greater decrease in the hepatic venous pressure gradient than a traditional non-selective β-blocker (propranolol); however, there has been no head-to-head comparison in a clinical trial [12]. The role of β-blockers in prophylaxis of GOV2 is not well studied. In this study, we used carvedilol. Although the primary outcome, including overall bleeding and mortality, was not improved by carvedilol, there was a significant improvement in the endoscopic changes. Patients who received carvedilol plus EBL had decreased GV size, risk signs for bleeding, and severity of PHG relative to those in patients who had only EBL. All patients who received carvedilol complied with taking the drug and showed no side effects.
Few clinical trials have tested the role of primary prophylaxis of GVs by injection of cyanoacrylate. However, to our knowledge, no study has tested the role of primary prophylaxis of cyanoacrylate injection for GOV2 specifically. This area has not had clear guidelines previously, so our data should help fill this gap. The main shortcoming of this study was the small sample size. Although the sample size was able to answer the primary question of the study regarding bleeding, it was insufficient to answer the question regarding mortality differences. A second shortcoming was the absence of a measurement method for evaluation of carvedilol efficacy as hepatic venous pressure gradient changes after treatment. Although this technique is very helpful in research, it is not available in general practice in many centers.
In conclusion, we found that prophylactic injection of GOV2 with cyanoacrylate before EBL significantly decreased the size of gastric extensions and risk signs of impending bleeding, especially in patients with extensions > 2 cm. Addition of carvedilol decreased the severity of PHG and may be recommended in these patients. Although there was a difference in rate of bleeding, it was not statistically significant. We recommend an independent study with a larger sample size to confirm the difference in rate of bleeding and mortality.
Approval number of the Bioethics Committee: PACTR202008491799224.