Introduction
Interventional cardiology has undergone a paradigm shift over the last decade, transitioning from transfemoral access (TFA) to transradial access (TRA) as the preferred route for coronary angiography and percutaneous coronary intervention (PCI). Historically, the femoral artery was preferred due to its large caliber and ease of catheter manipulation. However, the femoral route carries inherent risk of vascular complications, including retroperitoneal bleeding, hematoma formation, and the need for prolonged immobilization. These risks are particularly pronounced in high-risk patients requiring aggressive dual antiplatelet therapy or anticoagulation [1, 2]. The adoption of TRA has successfully addressed many of these limitations. The superficial course of the radial artery (RA) and its compressibility against the radius make it uniquely suited for effective hemostasis, resulting in significantly lower rates of major bleeding and vascular complications. These procedural advantages translate into robust clinical benefits, including reductions in mortality and major adverse cardiovascular events (MACE), especially in patients with acute coronary syndromes (ACS) [1–3]. A large meta-analysis by Ferrante et al. [3] demonstrated that TRA consistently reduces all-cause mortality, major bleeding, and vascular complications compared with TFA, with benefits evident across the full/entire spectrum of coronary artery disease (CAD). Current evidence supports a “radial-first” strategy in ACS due to improved outcomes, lower costs, shorter hospitalization, and enhanced patient experience through early ambulation and greater post-procedural comfort. However, analyses from MATRIX by Valgimigli et al. [4], RIVAL by Jolly et al. [5], and contemporary registries indicate that operator experience significantly influences these advantages. Recent data suggest a complex “proficiency paradox”: while low individual radial volume is associated with increased risks such as periprocedural stroke [6], exclusive reliance on radial access may degrade femoral skills, potentially leading to increased complications when TFA is required for complex anatomical scenarios [7]. These findings underscore the need for proficiency in both radial and femoral techniques to ensure procedural safety. Although TRA adoption has expanded considerably, it requires specific technical skills, including management of radial spasm and navigation of anatomical variants; occasional crossover to femoral access remains necessary. Maintaining competence in TFA is therefore essential for complex interventions and cases unsuitable for radial access. Increased procedural volumes, standardized training, and technological innovation have minimized these limitations and facilitated the routine use of TRA in elective and emergency settings. As interventional cardiology advances towards minimally invasive, patient-centered care, TRA has become a foundational component of contemporary practice, supported by robust evidence and international guidelines. Access-site selection has thus evolved from a matter of operator preference to a key determinant of clinical outcomes.
Historical background
Although transfemoral access dominated in early PCI, TRA has a long but underrecognized history. Radner first described radial artery catheterization in 1948 using a surgical cutdown; however, limitations in catheter technology led to its abandonment in favor of larger vessels [8]. Interest resurfaced in 1989 when Kiemeneij et al. demonstrated the feasibility of TRA for diagnostic angiography in 100 patients, laying the groundwork for its therapeutic application [9]. Through the 1990s and early 2000s, TRA remained confined to a small group of operators owing to limited experience and a lack of dedicated equipment. Nevertheless, accumulating observational and randomized evidence consistently demonstrated marked reductions in bleeding and vascular complications, prompting broader adoption [8]. Over the following decades, TRA achieved widespread validation and was progressively incorporated into major guidelines: the ESC issued Class I recommendations for ST-segment elevation myocardial infarction (STEMI) in 2017 and for non-ST segment elevation myocardial infarction (NSTEMI) in 2020, followed by similar ACC/AHA guidance in 2021 [10]. The 2023 ESC ACS guidelines further endorsed TRA as the default strategy [11]. Today, TRA is the standard approach for coronary angiography and PCI in many centers worldwide. Its evolution – from Radner’s early cutdown technique to a guideline-recommended first-line access route – illustrates the steady, evidence-driven advancement of interventional cardiology over more than 7 decades [9].
Anatomy of the radial artery
The RA is one of the two terminal branches of the brachial artery, typically originating within the cubital fossa at the level of the radial neck, just distal to the elbow joint [10, 12]. It courses laterally along the forearm, initially deep to the brachioradialis muscle, before emerging superficially in its distal third between the tendons of the brachioradialis and flexor carpi radialis. The RA then passes anterior to the radius and pronator quadratus, courses dorsally around the wrist through the anatomic snuffbox, and ultimately contributes to the deep palmar arch of the hand [10, 13].
Anatomical variations
The RA exhibits several important anatomical variations that can influence vascular interventions (Figure 1). A high-origin RA, also termed the brachioradial artery (BRA) can arise proximally from the brachial or axillary artery and was observed in approximately 8% of cases in cadaveric studies [10]. The BRA is the preferred and unifying term for what has previously been described by various names such as ‘high origin of the radial artery’, ‘radial artery originating from the axillary artery’, ‘high bifurcation of the brachial artery’, ‘continuance of the superficial brachial artery as the RA’, and ‘double brachial artery’ [13]. Specifically, the BRA denotes RA arising proximally to the elbow, either from the brachial artery or, less commonly, the axillary artery.
Figure 1
Normal anatomy of the radial artery and anatomical variations of the brachioradial artery with cubital crossover patterns
BA – brachial artery, RRA – recurrent radial artery, AIA – anterior interosseous artery, RA – radial artery, UA – ulnar artery, BRA – brachioradial artery, HBA – hypoplastic brachioradial artery.
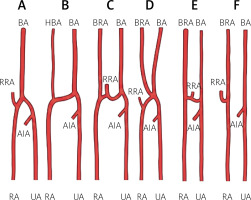
Frequently, an anastomosis exists between the BRA and the conventional brachial artery in the cubital fossa, known as the ‘cubital crossover’ or ‘cubital connection’. This anastomosis can present in several variations [13]:
Conventional anatomy with no BRA artery present (Figure 1 A).
Dominant type of cubital crossover, characterized by a hypoplastic pre-anastomotic segment of the BRA (Figure 1 B).
Balanced cubital crossover, in which the anastomosis has a diameter comparable to that of the BRA (Figure 1 C).
Arterial island, a rare variant in which the BRA and brachial artery form a complex resembling an island at the radial neck before dividing into the radial and ulnar arteries; this form is extremely uncommon and has been reported only in isolated cases (Figure 1 D).
Minimal cubital crossover, in which the anastomosis is narrower than the BRA (Figure 1 E).
Absent cubital crossover, indicating no anastomosis between the BRA and the conventional brachial artery within the cubital fossa (Figure 1 F).
The presence of the BRA is considered a risk factor for vascular complications during TRA procedures [13]. Accordingly, interventionists must recognize these anatomical variations to perform catheterization and interventions safely and effectively. Given that the BRA is present in nearly 10% of individuals, careful imaging – including subtraction angiography and roadmap guidance – should be employed routinely to prevent inadvertent wire perforation during retrograde catheterization of the brachial artery [13].
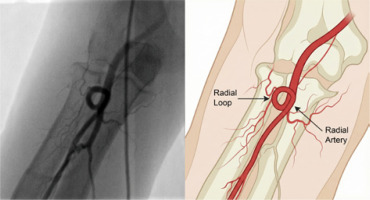
These variants encompass not only differences in origin but also altered courses and relationships to surrounding neurovascular structures. Another notable variant is the RA loop (Figure 2), which can complicate wire advancement and increase the risk of vessel trauma [13].
Figure 2
Radial artery loop during transradial catheterization: angiographic appearance (left) and anatomical illustration (right)
Branching patterns also vary; the radial recurrent artery, superficial palmar branch, and palmar carpal branch typically arise along the forearm, although their presence and origin sites can differ [10]. The RA most commonly terminates by anastomosing with the deep branch of the ulnar artery to form the deep palmar arch, although bifurcated and trifurcated endings have also been reported [10].
An exceptionally rare anatomical variation is the unilateral or bilateral absence of the RA. Zheng et al. [14] reported a case involving bilateral RA absence in which the distal upper limb was supplied primarily by an enlarged anterior interosseous artery. Their report also included a review of previously documented instances of unilateral RA absence, highlighting the variability and rarity of this vascular anomaly. These findings further underscore the importance of preprocedural imaging before any intervention involving the RA [14].
Morphometry
Radial artery morphometry provides essential information about vessel size, structure, and variability, thereby supporting safer and more effective use of the RA in interventional cardiology and vascular surgery (Table I).
Left and right radial access comparison
As TRA continues to gain favor in interventional cardiology owing to its lower risk of access-site complications, the question of whether right radial access (RRA) or left radial access (LRA) is superior has become increasingly relevant (Table I for morphometric comparison).
Procedural time and fluoroscopy time
Pelliccia et al. [15], in a cohort of 509 patients (304 RRA, 205 LRA), demonstrated that LRA significantly reduced both procedural and fluoroscopy times compared with RRA. These benefits were consistent across centers and independent of operator experience. A large meta-analysis by De Rosa et al. [16] further supported these findings, showing that LRA was associated with shorter fluoroscopy and procedural times, particularly in non-randomized real-world studies – likely reflecting anatomical advantages of the left subclavian trajectory. In contrast, Biondi-Zoccai et al. [17], pooling data from five randomized trials comprising 3,210 patients, reported no statistically significant difference between RRA and LRA, although results slightly favored the left side. Similarly, Shah et al. [18] found comparable total procedure times but a modest reduction in fluoroscopy time with LRA [15–18]. These observations might be explained by the more direct anatomical alignment of the left subclavian artery with the ascending aorta, which facilitates easier catheter advancement and reduces the need for prolonged fluoroscopic guidance, particularly among less experienced operators.
Contrast volume
In the study by Pelliccia et al. [15] contrast use was similar between the two access routes, a finding corroborated by a recent pooled analysis [17]. However, a small but statistically significant reduction in contrast volume with LRA has been reported in a limited number of studies [15–18], likely attributable to the anatomical advantages described above.
Access site failure and crossover
Recent studies reported comparable crossover rates between the two access sites [15–18]. However, Biondi-Zoccai et al. [17] observed a higher rate of crossover to TFA with RRA compared with LRA in their pooled analysis, identifying this as a procedural drawback of right-sided access, particularly for operators early in their learning curve. The higher crossover rates observed with RRA in some studies might be attributable to increased vessel tortuosity or challenging angles at the origin of the right subclavian and brachiocephalic arteries, which can complicate catheter navigation to the ascending aorta. Conversely, as operator experience increases, these anatomical limitations may be overcome, leading to equivalent success rates between the two approaches.
Radiation exposure
Pelliccia et al. [15] demonstrated that RRA was associated with greater radiation exposure to the patient, despite most operators being more familiar with the right side. This finding is consistent with the shorter fluoroscopy times observed with LRA, as confirmed by large meta-analyses [16, 17]. Furthermore, large-scale studies such as TALENT by Sciahbasi et al. [19] and OPERA by Dominici et al. [20], also reported lower operator radiation exposure with LRA [15–18]. Additionally, if the operator was left-handed, his right side might cross the midline of the patient thus exposing the operator to more radiation. Furthermore, LRA is usually used in complex PCI or procedures in patients with coronary artery bypass grafting, thus both radiation and contrast volume might be associated with bias related to longer procedure times.
Operator ergonomics and clinical implications
Operator experience is crucial for the success and safety of TRA. Tokarek et al. found that operators performing more than 75% of procedures via TRA had significantly better outcomes, including reduced periprocedural mortality [21]. Although no universal threshold exists for TRA PCI volume, the ESC requires a minimum of 75 PCIs per year and the AHA 50 PCIs per year for operators to maintain proficiency, regardless of access route. These data highlight the importance of structured training and procedural volume benchmarks in TRA adoption. Despite these procedural efficiencies associated with LRA, RRA remains the preferred approach in many centers because of operator convenience, especially for right-handed cardiologists [16]. Both De Rosa et al. [16] and Biondi-Zoccai et al. [17] recommend considering LRA during the early learning curve, as it may reduce crossover and procedural difficulties. Pelliccia et al. [15] found that LRA benefits persisted even among experienced high-volume operators, reinforcing its broader value in clinical practice. Shah et al. [18] further emphasized that while LRA may be ergonomically challenging for some operators – particularly in obese patients – it offers less subclavian tortuosity and may reduce the risk of cerebral microembolization owing to fewer catheter manipulations. Nonetheless, rates of stroke, transient ischemic attack, and vascular complications were similar between approaches [15–18].
Based on the current evidence, LRA offers modest but consistent procedural advantages over RRA (Table II), including shorter fluoroscopy and procedural times, reduced radiation exposure, and, in some datasets, lower crossover rates. Clinical safety endpoints remain equivalent. While RRA may still be favored for ergonomic reasons, LRA should be strongly considered, especially in settings aiming to improve procedural efficiency, minimize radiation exposure, or support training for less experienced operators [15–18].
Table II
Comparison of procedural parameters between left radial access (LRA) and right radial access (RRA)
| Parameter | LRA | RRA | Key findings |
|---|---|---|---|
| Procedural time | Shorter (33 ±24 min [15]; ~18.1 min [18]) | Longer (46 ±29 min [15]; ~18.8 min [18]) | Significant reduction with LRA [15, 16]; no significant difference [17, 18] |
| Fluoroscopy time | Lower (533 ±502 s [15]; 5.3 ±4.2 min [18]) | Higher (765 ±787 s [15]; 5.8 ±4.4 min [18]) | Significant reduction with LRA [15, 16, 18]; trend only [17] |
| Contrast volume | Slightly lower (176 ±69 ml [15]; 82 ±34 ml [18]) | Slightly higher (182 ±65 ml [15]; 84 ±35 ml [18]) | Small but significant reduction with LRA [16, 18]; no difference [15, 17] |
| Access site crossover | 3.1% [17]; 4.1% [18]; 8% [15] | 5% [15]; 4.2% [18]; 5.2% [17] | Higher with RRA [17]; no significant difference [15, 18] |
| Radiation exposure | Lower DAP (45.6 ±37.2 Gy·cm2 [15]); lower operator dose [18] | Higher DAP (59.6 ±35.5 Gy·cm2 [15]); higher operator dose [18] | Reduced with LRA [15, 18] |
| Anatomical alignment | More direct aortic path; easier catheter control [15, 18] | Greater subclavian tortuosity; more complex manipulation [15, 18] | Anatomical benefit with LRA [15, 18] |
| Operator ergonomics | Less ergonomic for right-handed operators; challenging in obese patients [18] | More ergonomic for right-handed operators [18] | LRA may cause discomfort during access in obese patients [18] |
| Stroke/TIA risk | Very low (0.1%); no increased risk observed [18] | Slightly higher theoretical risk (0.2%); not statistically significant [18] | No clinical difference [18] |
| Clinical safety | Equivalent to RRA [13–18] | Equivalent to LRA [15–18] | Comparable vascular and neurological complication rates across all studies |
| Learning curve | Favored during training; lower crossover and shorter fluoroscopy [16, 18] | Greater difficulty for less experienced operators [17, 18] | LRA recommended during the early learning phase [16, 18] |
Comparison with femoral access
Clinical outcomes
The meta-analysis by Chiarito et al. [22] (summarized in Table III) highlights the clinical advantages of RA over FA for both angiography and PCI. Randomized controlled trials (RCTs) such as RIVAL by Jolly et al. [5], RIFLE-STEACS by Romagnoli et al. [23], and AKI-MATRIX by Andò et al. [24] consistently demonstrated that RA reduces vascular complications and major bleeding compared with FA, confirming its role as an effective bleeding-avoidance strategy. These trials also suggested a potential mortality benefit, particularly in ACS patients, where bleeding risk and hemodynamic instability are higher. While ischemic outcomes such as myocardial infarction and stroke were similar between access sites, the consistent safety advantages of RA translated into improved composite outcomes (Table III), including MACE and net adverse clinical events (NACE). Collectively, RCT data support RA access as the default access strategy in contemporary interventional practice, especially in high-risk or bleeding-prone populations [22].
Table III
Clinical outcomes of radial versus femoral access: meta-analysis of 31 studies by Chiarito et al. [22]
Limitations of radial access
Despite its clinical benefits, RA access has several limitations (summarized in Table IV). According to Chiarito et al. [22], RA access is associated with technical challenges, particularly in patients with small RAs, severe vessel tortuosity, or a propensity for arterial spasm. These anatomical factors can prolong procedures and increase the risk of access failure or crossover to FA. Operator experience plays a critical role, as the bleeding reduction benefits of RA access were more pronounced in centers with high radial volume. Concerns have also been raised regarding a potential increase in subclinical cerebral embolism due to catheter manipulation in the aortic arch during RRA. Although no significant difference in clinical stroke rates was observed in the meta-analysis, most included trials did not conduct systematic neurological evaluations. Additionally, radial artery occlusion (RAO) – while often asymptomatic – may preclude future access or use as a graft conduit in coronary bypass surgery. Finally, large-bore or complex interventions may not be feasible through the radial approach, necessitating femoral access. These limitations underscore the importance of individualized access site selection based on patient anatomy, procedural requirements, and operator proficiency [22].
Table IV
Limitations of radial access for coronary interventions, adapted from Chiarito et al. [22]
Patient satisfaction and recovery
TRA has been consistently associated with greater patient satisfaction and improved recovery profiles compared with TFA. Multiple randomized trials, including RIVAL by Jolly et al. [5], SAFE-PCI by Rao et al. [25], and CARAFE by Louvard et al. [26] have demonstrated a clear patient preference for TRA due to increased comfort, reduced procedural pain, and enhanced postprocedural mobility. In the RIVAL trial by Jolly et al. more than 70% of surveyed patients favored TRA for future procedures [5], a preference echoed across several other studies. By contrast, patients undergoing TFA reported more back pain and ambulation difficulties. TRA also facilitates significantly earlier ambulation and shorter hospital length of stay (LOS). The CARAFE trial by Louvard et al. [26] showed that bed rest time was halved in the TRA group (4.9 h vs. 9.9 h), with a concurrent reduction in LOS (31 h vs. 42 h). These findings are reinforced by other trials, including RADIAMI by Chodór et al. [27] and RIFLE-STEACS by Romagnoli et al. [23], which demonstrated statistically significant reductions in both intensive care unit and total LOS with TRA. Collectively, the evidence supports TRA not only as clinically safer but also as conducive to improved patient experience and faster recovery, aligning with the goals of patient-centered care and value-based healthcare delivery [28].
Complications and risk management
Access site complications
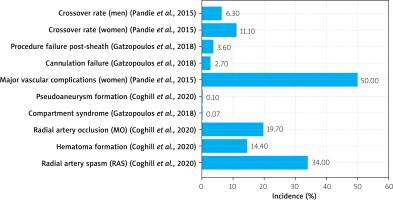
TRA is widely regarded as safer than TFA for coronary procedures; however, it is not without complications (Figure 3). The most frequent is radial artery spasm (RAS), which may occur in up to 34% of patients, particularly during cannulation or catheter manipulation. RAS is triggered by the artery’s small diameter, high smooth muscle content, and sympathetic innervation, which together render it highly responsive to mechanical and chemical stimuli [29]. Spasm can cause significant patient discomfort and procedural failure, and may exacerbate endothelial injury, thereby increasing the risk of thrombosis or dissection. Other access site complications include hematoma formation (up to 14.4%), RAO (up to 19.7%), and rare but serious events such as compartment syndrome and pseudoaneurysm formation. Although less frequent than with TFA, these complications necessitate meticulous technique and preventive strategies to optimize patient safety [29].
Risk factors and prevention strategies
Several risk factors predispose patients to RA access complications, including female sex, smaller artery diameter, diabetes, peripheral vascular disease, and anatomical anomalies [29]. The RIVAL trial subgroup analysis by Pandie et al. [30] demonstrated that women are particularly susceptible to bleeding and vascular complications, and that TRA significantly reduces these risks compared to TFA (hazard ratio for major vascular complications: 0.50; 95% confidence interval: 0.32–0.78; p = 0.002;) [29, 30]. Multivariable analysis further revealed that female sex, older age, and prior coronary artery bypass grafting were independent predictors of crossover and vascular complications.
To mitigate these risks, a multifaceted prevention strategy is recommended. This includes adequate patient sedation, use of smaller hydrophilic sheaths, and administration of antispasmodic cocktails combining vasodilators like glyceryl trinitrate and calcium channel blockers such as verapamil. Patent hemostasis and techniques like ULTRA (ulnar compression to preserve radial patency) are also employed to prevent RAO [31]. Local delivery of nitric oxide has shown promise as a novel method for achieving targeted vasodilation without systemic hypotension, potentially offering a safer and more effective means of spasm prevention [29].
Furthermore, although manual compression can be used to stop the bleeding after sheath removal, most contemporary practice relies on compression bands specifically designed for radial access hemostasis. These bands apply controlled, gradual pressure over the puncture site, which helps maintain arterial patency while allowing enough external force to stop bleeding. Proper technique involves initially inflating or tightening the bands to achieve immediate hemostasis, followed by incremental stepwise deflation over several hours to avoid excessive compression that could compromise flow. Excessive or prolonged pressure increases the risk of RAO, whereas insufficient pressure predisposes to hematoma formation [29]. The TR Band® (Terumo) remains the most widely used device globally, offering dual-balloon inflation for precise modulation of compression. Alternative systems such as the RadAR™ (Advanced Vascular Dynamics) and D-Stat® Radial provide ergonomic designs and simplified deployment, whereas the Vasc Band™ (Medtronic) and Safeguard Radial® (Merit Medical) offer transparent platforms enabling direct visualization of the puncture site. Comparative studies demonstrate that proper application technique – rather than device selection alone – is the key determinant of RAO prevention [32, 33]. Overall, modern radial compression devices play a crucial role in enhancing safety, maintaining arterial patency, and supporting the growing dominance of transradial access in interventional cardiology.
Management of radial access failure
Radial access failure, though uncommon, remains a technical challenge during coronary interventions. Gatzopoulos et al. [34] reported that in a high-volume radial center, initial RA cannulation failure occurred in approximately 2.7% of cases, with an additional 3.6% experienced procedural failure after successful sheath insertion. Rather than defaulting to TFA, their structured algorithm prioritized alternative forearm routes – including contralateral radial and ulnar arteries – to preserve the advantages of upper limb access. This approach reduced the overall TFA use to just 1.4% of cases. Management of failed RA accesses therefore depends on early recognition of anatomical or mechanical obstacles, thoughtful escalation to other forearm vessels, and operator expertise in less commonly used approaches like translunar catheterization. Maintaining a forearm-first strategy is associated with fewer access site complications and shorter recovery times, making it a preferred approach even following initial radial failure [34]. Pandie et al. [30] also reported significantly higher crossover rates in women (11.1%) compared to men (6.3%), with RAS and anatomical loops as leading causes, reinforcing the need for proactive planning in high-risk subgroups.
Special populations
Elderly patients
Elderly patients, who often present with frailty and a heightened risk of bleeding and vascular complications, stand to benefit significantly from TRA. Basu et al. [35] conducted a meta-analysis of patients aged 75 years or older and found that TRA led to an 85% reduction in access site complications and a 67.9% decrease in major bleeding events compared to TFA. Additionally, TRA shortened ambulation time by more than 14 h – a particularly important factor in reducing delirium, deconditioning, and hospital LOS in this population. Although there was a modest increase in crossover rates and procedural time (2.64 min longer), the observed mortality benefit (risk ratio: 0.66) further supports TRA as the preferred route in older adults [35]. Andò et al. [36] corroborated these findings in their ACS-focused meta-analysis, demonstrating consistent benefits across age subgroups.
Women and patients with smallcaliber arteries
Women and individuals with small-caliber arteries face unique challenges in PCI, particularly a higher risk of access site complications and bleeding. The SAFE-PCI for Women trial by Rao et al. [25], the first RCT specifically evaluating women, showed a 60% reduction in bleeding and vascular complications with TRA in the PCI cohort, although this did not reach statistical significance due to early trial termination. In the total randomized population (diagnostic angiography plus PCI), TRA was associated with a statistically significant 70% reduction in the primary endpoint (occurrence of Bleeding Academic Research Consortium type 2, 3, or 5 bleeding or vascular complications requiring intervention). However, the trial also revealed higher crossover rates in the TRA group (6.7% vs. 1.9%), primarily owing to RAS, which disproportionately affects smaller and more reactive vessels. Chester et al. [37] emphasized that pre-procedure ultrasound and sheath-to-artery ratio optimization could mitigate these issues and improve outcomes for women and those with smaller vessels.
Chronic kidney disease and dialysis patients
TRA for cardiac catheterization in patients with chronic kidney disease (CKD) and end-stage renal disease was traditionally avoided due to concerns that injury or occlusion of the RA could compromise its future use for arteriovenous fistula creation, which is essential for hemodialysis. Both Vora et al. [38] and Sutton et al. [39] note that this caution stemmed from the risk of RAO (reported up to 8%) and the fear of jeopardizing existing upper-extremity dialysis access. Additionally, CKD patients have a higher baseline risk of bleeding and vascular complications following PCI, leading many clinicians to favor TFA despite its higher bleeding risk. These concerns limited the inclusion of CKD and dialysis patients in major clinical trials, leaving limited direct evidence to guide access-site selection in this high-risk population [38, 39].
However, both studies provide strong evidence that TRA is not only safe but potentially advantageous for CKD and dialysis patients. Vora et al. [38] using data from the VA CART program (229,108 patients; 35,979 patients underwent TRA and 193,129 TFA), found that TRA was associated with significantly lower rates of postprocedural transfusion – particularly in severe CKD – and reduced progression to dialysis at 1 year compared to TFA. Sutton et al. [39] (analyzing data from the Blue Cross Blue Shield of Michigan registry (6,359 dialysis patients), reported that TRA adoption increased from 2.4% in 2010 to 22.3% in 2018, with lower rates of bleeding, transfusion, and in-hospital mortality (2.3% vs. 3.9%) compared with TFA. Both studies conclude that the benefits of reduced bleeding and mortality with TRA outweigh the theoretical risks of compromising dialysis access, suggesting that RA PCI should be more widely considered for CKD and dialysis patients when anatomically and technically feasible [38, 39].
Acute coronary syndromes
In patients presenting with ACS, TRA offers not only procedural safety but also significant mortality benefits. The large-scale meta-analysis by Nardin et al. [40], which included more than 19,000 ACS patients, demonstrated a 28% relative reduction in mortality (1.8% vs. 2.5%) and a 43% reduction in major bleeding with TRA compared to TFA. These advantages were consistent across both STEMI and NSTEMI. Andò et al. [36] corroborated these findings, showing reductions in MACE, major bleeding, and all-cause mortality with TRA. Cantor et al. [41] additionally noted that the benefits were observed even in pretreatment settings for STEMI, reinforcing TRA as the first-line strategy in ACS management where operator experience permits.
Distal radial artery access
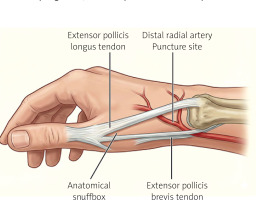
Anatomical considerations
Distal radial artery (DRA) access, is increasingly adopted for angiography and PCI, offers several anatomical and procedural advantages that distinguish it from traditional forearm radial access. The DRA is located in the anatomical snuffbox (Figure 4), a superficial triangular depression on the dorsolateral aspect of the wrist bordered by the tendons of the extensor pollicis longus and brevis, underlain by the scaphoid and trapezium bones [42]. This superficial location, combined with the underlying bony structures, facilitates effective manual compression and enables quicker, more secure hemostasis after sheath removal [42, 43]. Importantly, the DRA lies distal to the origin of the superficial palmar branch of the RA, which contributes to the superficial palmar arch, allowing hand perfusion even in the event of vessel occlusion. As a result, occlusion of the distal segment typically does not compromise hand viability and may carry a lower initial risk of occlusion compared with proximal RAO [42, 43].
Reduced complication risk
The superficial anatomy of the snuffbox reduces the risk of certain complications. Because the DRA is constrained by surrounding fascial and tendon structures, it remains more stable during puncture, decreasing the likelihood of vessel movement and failed access attempts [25]. This positioning also limits hematoma propagation and avoids deep forearm structures, thereby reducing the risks of compartment syndrome and vascular trauma compared to traditional TRA [43, 44]. The bony confines of the anatomical snuffbox facilitate uniform compression, promoting quicker hemostasis and minimizing the need for prolonged pressure devices [42].
Ergonomic and procedural benefits
DRA access offers significant ergonomic benefits. When using left-hand DRA access, the patient’s arm can rest comfortably across the abdomen or groin, enabling the operator to remain on the right side – standard positioning for most right-handed interventions – thereby avoiding the need to lean across the patient [42, 44]. This improves operator comfort and efficiency while reducing radiation exposure [42, 44].
Technical considerations and learning curve
Early DRA access may be more technically challenging due to the smaller vessel diameter – often cited as approximately 2.0 to 2.4 mm compared to 2.3 to 2.7 mm in the proximal RA – although operators have noted improvements with experience [42, 43]. In the DARFORA trial by Daralammouri et al. reported longer initial puncture times (56.6 ±61.1 s vs. 20.0 ±18.4 s, p < 0.001) and more attempts (1.9 ±1.3 vs. 1.2 ±0.6, p < 0.001) for DRA compared to forearm radial access. However, with increased operator experience, both puncture duration and the number of attempts decreased significantly, demonstrating a tangible learning curve. Notably, despite longer access times, overall procedural time, fluoroscopy time, and radiation exposure were lower in the DRA group, likely reflecting more favorable vascular geometry and catheter control [44].
Hemostasis and radial artery preservation
Another advantage of DRA is reduced hemostasis time. Daralammouri et al. [44] found that hemostasis duration was significantly shorter in the DRA group (114 ±44.4 min) compared to the forearm radial access group (134 ±50.1 min, p = 0.002). This can be attributed to the vessel’s superficial location and supportive surrounding anatomy, facilitating faster and more complete compression. Shorter compression time has been linked to reduced patient discomfort, lower risk of access site bleeding, and decreased incidence of RAO [43]. Preservation of RA patency is one of the most compelling benefits of DRA. Cao et al. [43], in a meta-analysis of six RCTs, reported a significantly lower incidence of RAO in DRA compared to TRA (risk ratio = 0.203, p < 0.001), highlighting its protective effect on this critical vascular conduit [43, 44].
Preserving the proximal RA is particularly important in patients who may require future access for repeated coronary procedures, hemodialysis fistula creation, or as a conduit in coronary artery bypass grafting [42, 43]. The DRA spares this segment by using a more distal site, which is rarely involved in vascular harvests or fistula formation. Furthermore, in cases where proximal RA is lost owing to previous occlusion or trauma, the DRA can serve as a site for retrograde recanalization [42].
Summary
The anatomical and procedural benefits of DRA – namely improved vessel preservation, safer access, ergonomic advantages, reduced complication rates, and shorter hemostasis times – position it as an increasingly favorable alternative to traditional TRA for angiography and PCI. Although the learning curve may be steeper due to anatomical constraints, evidence suggests that these challenges are mitigated with experience, leading to procedural efficiency and excellent clinical outcomes. As operator familiarity increases, the DRA is likely to play an increasingly central role in interventional cardiology [42–44].
Cost and hospitalization time analysis
The economic and operational benefits of TRA for PCI have been consistently demonstrated across multiple healthcare settings. In a large single-center study using data from the Fuwai Hospital in China, Jin et al. [45] analyzed 5,306 PCI patients and found that those treated via TRA had significantly lower total hospital costs compared to those undergoing TFA intervention. The unadjusted mean total hospital cost was ¥57,900 ($9,190) for TRA versus ¥67,418 ($10,701) for TFA intervention. After adjusting for confounders using inverse probability weighting, TRA was associated with a mean cost reduction of ¥8,081 (~$1,283). More than 80% of the savings were due to lower PCI-related costs (¥5,162/~$819) and hospitalization costs (¥1,399/~$222). These cost differences were primarily attributed to the use of expensive vascular closure devices in the femoral group (used in 67.0% of TFA cases and 0% of TRA) and overall shorter hospital stay in the TRA group. Clinically, patients in the TRA group also experienced significantly fewer post-PCI bleeding events and MACE [45].
In a national analysis from England, Mamas et al. [46] evaluated 323,656 PCI procedures performed between 2010 and 2014 using data from the British Cardiovascular Intervention Society database. On average, TRA reduced the cost per procedure by £250.59, representing a 22% reduction compared to TFA. The majority of the cost savings (£190.43; 76%) was attributable to reduced hospital LOS. The real-world economic impact of this transition was substantial: TRA adoption during the study period saved the National Health Service an estimated £13.31 million. Furthermore, if all regions had adopted TRA at the same rate as the highest-uptake region (North East England), the NHS could have saved up to £33.40 million. The greatest per-case savings were observed in high-acuity patients, particularly those presenting with STEMI, where the TRA approach yielded a cost reduction of £348.26 per procedure [46].
In the United States, Mitchell et al. [47] conducted a systematic review and cost–benefit analysis of 14 RCTs comparing TRA and TFA for coronary angiography and PCI. Their simulation model estimated that TRA yielded a per-patient cost saving of $275 (95% confidence interval: –$374 to –$183) from the hospital perspective. Although TRA was associated with a slight increase in procedure time (mean difference of 1.4 min), it significantly reduced hemostasis time by approximately 13 min (mean difference: –13.1 min; 95% confidence interval: 23.9 to –2.3) and was substantially lower rates of major bleeding, vascular complications, and hematomas. These reductions translated into lower costs for managing complications, which more than offset the minor increases in procedural time and crossover rates. Under all modeled scenarios, TRA remained the cost-favorable approach [47].
Collectively, these studies provide strong and consistent evidence that TRA offers meaningful cost savings and reductions in hospital stay while simultaneously improving clinical outcomes. The cost advantage stems primarily from fewer access-related complications, reduced use of closure devices, and significantly shorter postprocedural recovery and hospitalization times. These benefits support the broader adoption of TRA in routine PCI practice, particularly in high-risk patients where the economic and clinical advantages are most pronounced.
The future of radial artery access in interventional cardiology
Robotic percutaneous coronary intervention (R-PCI) represents an emerging technology with the potential to enhance procedural precision and operator safety. As demonstrated by Mahmud et al. [48], R-PCI achieved high clinical success rates (more than 97%) with low MACE and no device-related complications. The second-generation CorPath GRX system, in particular, demonstrated higher technical success in treating complex lesions, including bifurcations, severe calcifications, and chronic total occlusions. By enabling operators to perform interventions remotely from a radiation-shielded cockpit, R-PCI minimizes radiation exposure and orthopedic strain, addressing occupational hazards without compromising efficacy. These data confirm that robotic technology can safely expand the scope of PCI, improving accuracy, consistency, and overall procedural outcomes in routine clinical practice [48]. The integration of robotic technology with TRA may prove transformative.
Conclusion
TRA has emerged as a cornerstone in modern interventional cardiology, progressively replacing TFA as the default approach for PCI. Its adoption is supported by a substantial body of evidence demonstrating consistent advantages across clinical outcomes, patient experience, and healthcare resource utilization. Compared with TFA, TRA is associated with lower rates of bleeding, vascular complications, and mortality, especially in high-risk groups such as patients with ACS [49].
TRA offers important procedural benefits, including easier hemostasis, quicker patient mobilization, and reduced hospital stays – all of which contribute to enhanced patient comfort and lower overall costs. Advances such as LRA and DRA have further improved procedural ergonomics and vessel preservation, supporting broader adoption across diverse patient populations and case complexities. Despite these benefits, challenges remain. Operator preference, difficult vascular anatomy, and limited familiarity with advanced radial techniques may necessitate TFA. These barriers, however, are increasingly mitigated through dedicated training, ultrasound guidance, and adjunctive technologies such as sheathless systems and alternative forearm access routes [50]. As radial-first adoption expands, operator proficiency continues to improve. In summary, TRA enhances clinical outcomes, patient experience, and resource utilization. With growing evidence and rising operator expertise, TRA is poised to remain the preferred route for coronary interventions, provided that continued efforts focus on skill development, technical refinement, and institutional support [21, 47, 49].