Introduction
The treatment outcomes of patients with advanced/metastatic melanoma were poor before the use of new therapeutic options. The median overall survival period via a standard single agent or multidrug chemotherapy (CTH) was 6–11 months [1–5]. The development of new types of therapies such as immunotherapy (IT) and targeted therapy (TT) has significantly improved the outcomes of patients with advanced/disseminated melanoma. At present, the median aggregate survival period observed in clinical trials using IT or TT is over three years [6–12].
Immunotherapy uses immune checkpoint inhibitors (ICIs), which include anticytotoxic T-lymphocyte antigen 4 (anti-CTLA-4) antibodies – ipilimumab and antibodies against the programmed cell death-1 (anti-PD-1) inhibitors – nivolumab and pembrolizumab [13]. Targeted therapy includes BRAF proto-oncogene serine/threonine kinase inhibitors (BRAFi; vemurafenib, dabrafenib, and encorafenib) and MEK inhibitors (MEKi; cobimetinib, trametinib, and binimetinib) [14].
In Poland, IT or TT treatment of patients with melanoma was initiated and refunded by the National Health Fund (NFZ) in 2013 as part of the national drug program. Vemurafenib was the first of the new class of drugs to be refunded under the national drug program (from March 1, 2013). The next few drugs to be refunded were: ipilimumab, which was used as a second-line treatment in patients with advanced/metastatic melanoma (from March 1, 2014), dabrafenib (from July 1, 2015), and cobimetinib and trametinib (from 2017) as a supplement to vemurafenib and dabrafenib therapy, respectively. Nivolumab and pembrolizumab have been used and refunded from June 1, 2016. Initially, BRAFi/MEKi were only refunded prior to anti-PD-1 therapy. Since 2017, BRAFi/MEKi have been reimbursed in the first, second, and subsequent treatment lines and also after anti-PD-1 therapy. Therefore, from 2017, anti-PD-1 was used as first-line treatment for BRAF-mutated patients.
This study aims at analysing the treatment outcomes of patients with advanced/disseminated melanoma through new therapies under the national drug program.
Material and methods
A retrospective analysis was conducted for 287 patients with unresectable stage III and stage IV melanoma treated at the Maria Sklodowska-Curie National Research Institute of Oncology Krakow Branch, from 2013 to 2019. Moreover, this retrospective non-interventional study did not require any additional approval from an ethical authority. All the analysed data were gathered as part of routine diagnosis and treatment. Patients were diagnosed and treated in line with national guidelines and agreements. Treatments were covered according to the NFZ reimbursement regulations (Poland) outlined by the Agency for Health Technology Assessment and Tariff System (AOTMiT). All the patients signed an informed consent form for the treatment as a standard operating procedure in our hospital. The patients who participated in the clinical trials were excluded from the analysis.
All the enrolled patients were treated with IT (comprising pembrolizumab/nivolumab/ipilimumab) or TT (comprising vemurafenib ±cobimetinib or dabrafenib ±trametinib) in at least one treatment line. All the patients were included in the treatment according to the inclusion criteria of the drug programs. Data on age, sex, location of the primary lesion, presence or absence of BRAF mutation, stage of the disease, and type of therapy used in the first, second, third, and subsequent lines were recorded. Information on the stage of the disease, location of metastatic lesions, LDH level, and Eastern Cooperative Oncology Group (ECOG) performance status were collected at the beginning of first-line systemic therapy.
Statistical analysis
The primary aims of the study were progression-free survival (PFS), overall survival (OS), overall response rate (ORR), and disease control rate (DCR) defined by the response evaluation criteria in solid tumours (RECIST) 1.1. PFS and OS were measured from the first administration of medication until disease progression according to RECIST, death, or last documented/reported visit. Patients who were alive at the end of the study period were examined on the date of the last follow-up. The Kaplan-Meier method was used to calculate PFS and OS survival curves, and the log-rank test was used to compare these measures. The multivariate analyses, performed using Cox’s proportional hazards model, were used to evaluate connections between predictive factors (patients and treatment characteristics) as well as PFS and OS. The differences were considered statistically significant if the p-values were < 0.05. All statistical analyses were performed using Statistica 12 software.
Baseline characteristics
The median age in the study group was 63 years (range: 18–94 years), among whom 119 (42%) patients were above 65 years of age. Men represented 60% of the total population. The main location of the primary lesion was in the skin (85%), and less often in a mucous membrane or an unknown primary site (4% and 11%, respectively). BRAF mutation was detected in 152 (55%) patients. In general, the majority of patients (92%) were in very good or good condition (ECOG 0 or 1). Brain metastasis was detected in 64 (22%) patients. Table 1 summarises the baseline characteristics of patients.
Table 1
Patient characteristics according to therapy regimen.
Types and sequences of the treatments
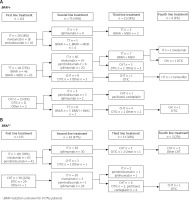
In the first line, IT was administered to 133 (46%) patients, TT to 111 (39%) patients, and CTH to 43 (15%) patients. In the entire group, 138 (48%) patients qualified for the second line of therapy: 114 (82%) patients received IT, 13 (10%) received TT, while the remaining 11 (8%) received CTH. Only 34 (13%) of the enrolled patients received the third line of therapy: IT was prescribed to two (6%), TT to 10 (30%), and CTH to 22 (64%) of the patients. The fourth-line treatment was limited to seven (2%) of the patients who received CTH (n = 5, 72%), IT (n = 1, 14%), and hormone therapy (HT; n = 1, 14%). The fifth-line treatment (dacarbazine chemotherapy) was provided to only one patient. Figure 1 displays the treatment sequences for the first, second, third, and fourth lines for BRAF-mutated (Fig. 1A) and BRAF-wild type (Fig. 1B) patients.
Treatment outcomes corresponding to the line of treatment
Median OS and PFS in the study group from the beginning of the first-line treatment were 14.9 and 6.7 months, respectively (Fig. 2A and 2B). In the second line, OS and PFS were 7.2 and 3.1 months, respectively, and in the third line they were 5.5 and 3.9 months, respectively (Fig. 2C). Based on the first-line treatment, the median OS was 19.2, 12.6, and 15.9 months for IT, TT, and CTH, respectively. The median follow-up was 12.6 months (range 0.2–85.9). However, it was longer for the CTH subgroup at 15.0 (range 1.2–85.9) months compared to IT and 11.7 (range 0.2–36.5) months and TT at 11.9 (range 0.2–62.5) months. Table 2 represents the survival outcomes and response rates based on the types and sequences of the therapy.
Fig. 2
Kaplan-Meier curves for overall survival in first-line treatment (A), progression-free survival in first-line treatment (B), overall survival stratified by type of first-line treatment (C), overall survival (D), and progression-free survival (E) for immunotherapy in first-line treatment for BRAF-mutated vs. BRAF-wild type population (IT – immunotherapy, TT – targeted therapy, CHT – chemotherapy)

Table 2
Results of treatment depending on the type of therapy and treatment line
Treatment results in the BRAF-mutated and BRAF-wild type population
The median OS in the BRAF-mutated and BRAF-wild type populations were 14.2 and 14.9 months, respectively, and there was no statistically significant difference (p = 0.7972). There was also no statistically significant difference in PFS in these groups (p = 0.3663).
The median OS for IT in first-line treatment in the BRAF-mutated population was not reached, and in the BRAF-wild type population it was 14.8 months. There was a statistically significant difference between the groups (p = 0.0243) (Fig. 2D). However, no significance was demonstrated for PFS (p = 0.5134) (Fig. 2E). The median OS for CHT in first-line treatment in the BRAF-mutated and in the BRAF-wild type population was 15.1 and 14.3 months, respectively, and there was no statistically significant difference (p = 0.9167). However, PFS was longer in the BRAF-mutated population, and there was statistically significant difference (p = 0.0237).
Univariate and multivariate analyses of the experimental group
A statistically significant positive effect on OS and PFS at the start of the first line of treatment were as follows: the absence of metastasis in the brain (p < 0.0001 and p = 0.0004, respectively), normal LDH levels (p < 0.0001 and p = 0.0063, respectively), ECOG – 0 (for both p < 0.0001), lower stage (p < 0.0001 and p = 0.0027, respectively), less than three locations of metastatic lesions (p = 0.0001 and p = 0.0031, respectively), objective response (complete response + partial response) to the treatment (for both p < 0.0001), more than one treatment line (p = 0.0072 and p = 0.0432, respectively), and termination of the treatment due to toxicity (p < 0.0001 and p = 0.0001, respectively). Chemotherapy as a treatment regimen in the first line had the worst effect on PFS (p = 0.0035), but without affecting OS (p = 0.1336).
Other variables, including gender, age > 65 years, location of the primary lesion, and the presence of BRAF mutation had no statistically significant effect on OS and PFS.
Multivariate analyses confirmed that normal LDH levels, the absence of brain metastasis, ECOG 0, and objective response to treatment were strong predictors of longer OS. For PFS, the absence of brain metastasis, very good general condition, and response to treatment were found to be predictive factors. Table 3 presents the results of the multivariate analyses.
Table 3
Results of the multivariate Cox proportional hazards models in metastatic melanoma patients before first-line therapy
[i] PFS – progression-free survival, OS – overall survival, IT – immunotherapy, TT – targeted therapy, CHT – chemotherapy, LDH – lactate dehydrogenase, ECOG/PS – Eastern Cooperative Oncology Group/Performance status, CR – complete response, PR – partial response, SD – stable disease, PD – progression of disease
Results and discussion
In the present study, we have indicated current trends and treatment effects of patients with advanced/disseminated melanoma through real-world results. The presented results confirm the effectiveness of these therapies.
The survival outcomes in a similar population of patients treated between 1995 and 2005 at the National Research Institute of Oncology in Warsaw and Krakow, before the discovery and approval of new therapies, were worse [5]. The median OS and PFS were 7.1 and 3.5 months, respectively, with one-year and two-year survival rates of 32% and 12.5%, respectively [5]. In our study, patients had dramatically better treatment results: median OS and PFS were 14.9 and 6.7 months, respectively, and the one-year and two-year survival rates were 52% and 22%, respectively. It has been found that the number of responses to treatment is higher when new therapies are used. In our study, 43% ORR and 77% DCR were noted, and 19% ORR and 65% DCR were recorded in the 1995–2005 study. All the results obtained in our study are comparable with the treatment outcomes via new therapies in other centres in Poland [15–18] and other studies with real-world data [19–22].
The most unexpected finding in our study is the correlation between the type of first-line treatment and OS. Across the experimental group, patients received IT, TT, and CTH as a first-line treatment, and the median OS was 19.2, 12.6, and 15.9 months, respectively. The median OS was longer in the CTH group than in the TT group and shorter than in the IT group. This could be because the group of patients receiving CTH had better prognostic factors than patients receiving IT or TT. In the CTH group, only 9% of patients had brain metastases and 26% exhibited elevated LDH than in the group receiving IT (15% and 39%, respectively) or TT (36% and 62%, respectively). However, the most interesting finding is that each patient who received CTH in the first-line treatment qualified for the second-line treatment – either IT (81% of patients) or TT (19% of patients). Therefore, the CTH-IT or CTH-TT treatment group is a very special group. It should also be noted that patients in a good or very good condition more often qualified for the second-line treatment (this is due to the inclusion criteria in the drug program with IT or TT). Conclusions from real-world data in the United Kingdom (UK) for ipilimumab were also similar [19]. The UK study also revealed that patients in a better condition and with favourable prognostic factors had a better prognosis [19]. Hence, new therapies (especially IT) are active and effective in subsequent treatment lines.
Another important finding is that patients with more than one treatment line had a better prognosis. It should be noted that the median PFS in the second and third lines were similar, at 3.1 and 3.9 months, respectively. Also, in a real-world study, Cimminiello et al. revealed the efficacy of subsequent treatments [20]. Hence, the third or fourth lines of treatment in patients with advanced melanoma could be beneficial and should be considered as a treatment option. However, this hypothesis should be tested in prospective clinical trials.
Another observation is the difference in OS, PFS, and treatment responses between IT and TT in the first and second lines of treatment. A comparison of IT and TT treatment outcomes in the first and second lines indicated that patients receiving IT in the second line had a worse prognosis than patients receiving IT treatment in the first line. Existing studies indicate that immunotherapy administered before the targeted therapy may provide better results even in patients with BRAF mutation [23–27]. In our analysis, better prognosis in the first-line treatment with IT could be due to a lower number of patients receiving TT in the second line. However, 50% of our patients continued treatment with IT in the second line (received ipilimumab, anti-CTLA-4), which also affected the obtained results. However, this sequence of the treatment requires further research and observation.
Another observation is the difference in OS between BRAF-mutated and BRAF-wild type populations in first-line IT treatment. This may be the result of selecting BRAF-mutated patients for IT because BRAF-mutated patients who had better prognostic factors were more often qualified for IT treatment in the first line.
Our analysis has certain limitations because it is a retrospective study. Nevertheless, the experimental group is representative and comparable with other groups with respect to prognostic factors. The presented analysis (2013–2019) has indicated that normal LDH levels, absence of brain metastases, and good performance status have a positive impact on prognosis. Other studies with real-world data have also revealed similar prognostic factors [19–22]. Similar results were also found in research conducted before the era of new therapies [28–33] and in the analysis from 1995–2005 [5], which prove the similarity between these two populations. As expected, our data indicated that the presence of brain metastases predicts worse OS and PFS, which supports recently published studies [34, 35]. However, older analyses did not reveal such a strong impact of brain involvement on OS and PFS [3–5, 36–38], which is probably due to the absence of neuroimaging techniques in the detection of brain metastases. In our analysis, the prognostic factors associated with OS are similar to those obtained in other analyses and Melanoma Staging: American Joint Committee on Cancer (AJCC) 8th Edition [35, 39]. It should also be noted that despite the use of new treatment options and improved survival rates, prognostic factors remain the same, and the same group of patients still has a poor prognosis (poor general condition, brain metastases, and elevated LDH).