Introduction
Metastasis to the lung – which can develop in up to 30% of patients with different primary solid tumors [1] – has often been considered by clinicians as a portentous sign with a dismal prognosis and short survival. Most of these patients were treated with systemic therapy on a palliative intent basis.
However, it has been shown that in carefully selected patients with isolated pulmonary metastasis, surgical resection can render them free of disease, offering a chance of cure and prolonged survival [2, 3]. Classically, pulmonary metastasectomy (PM) is offered in the setting of oligometastases, which is generally limited to five or fewer metastases or better defined as ‘amenable to a curative therapeutic strategy’ [4, 5]. Strict criteria should be met for PM: limited number of technically resectable nodules, controlled/controllable primary tumor; no other extra-thoracic lesions and good functional reserve to tolerate surgery [2, 3, 6].
Open thoracotomy (OT) has been considered as the standard approach for management of pulmonary metastasis due to enhanced ability of bimanual palpation of the entire lung parenchyma and higher chance of detection – and resection – of more lung nodules that were undetectable in the pre-operative imaging studies [7]. However, the advances in radiological imaging (1 mm thin slice single-breath CT scan), peri-operative localization techniques and in minimally invasive surgery have made video-assisted thoracoscopic surgery (VATS) an increasingly popular approach for management of lung secondaries.
Although more lung nodules are detected by OT, there is a little evidence supporting improved survival from thoracotomy in this disease entity. Survival rates were consistently shown to be not inferior in VATS as compared to OT in management of pulmonary metastasis [8–10]. This should be interpreted cautiously as most of these data are derived from retrospective studies.
Aim
There are sparse prospective studies investigating the role of VATS in management of pulmonary metastasis. Therefore, we aim to prospectively investigate short-term surgical and pathological outcomes for PM patients operated on by either VATS or OT for management of lung secondaries.
Material and methods
This is a prospective study investigating short-term outcomes in patients undergoing PM with either OT or VATS. All patients who were eligible for PM at the Surgical Oncology Department, South Egypt Cancer Institute and Cardiothoracic Surgery Department, Faculty of Medicine, Assiut Univerity, Egypt between October 2017 and December 2020 were recruited. Exclusion criteria were patients with evidence of recurrence at the primary site, progressive disease (increased number of metastatic lesions in imaging studies over the last 6 months), uncontrolled extra-thoracic metastasis, histologically proven primary tumors in the lung, lesions necessitating pneumonectomy and poor pulmonary reserve (FEV1< 30% and/or DLCO < 30%). The study protocol was approved by our Institutional Review Board. Informed written consent was obtained from each patient before inclusion. All procedures are in accordance with the Helsinki Declaration.
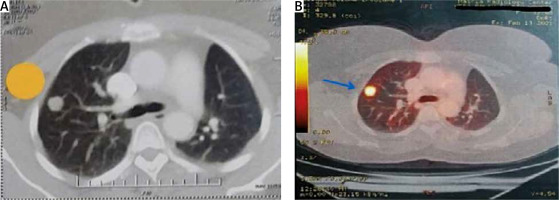
All eligible patients were discussed in multidisciplinary team (MDT) meetings including a surgeon, medical oncologist, radio-oncologist, radiologist and pathologist to estimate the potential benefit of surgical resection. For all patients, full medical history (including primary site type and previous chemo/radiation therapy received) was sought. Thorough clinical examination, routine laboratory investigations and lung function tests were performed. Single breath thin-slice IV contrast chest CT was performed within 4 weeks of the surgery to plan the type of procedure (Figure 1 A). Staging for extra-thoracic disease was performed depending on the primary tumor. Fluorodeoxyglucose positron emission tomography (FDG-PET) was proposed in most cases to exclude extra-thoracic metastases, determine the metabolic activity of lung nodules and assess the nodal disease (Figure 1 B). In selected cases, a brain magnetic resonance imaging (MRI) study was also done to exclude central nervous system (CNS) metastasis.
Figure 1
Preoperative imaging studies: showing right upper lobe solitary lung nodule in chest CT (A) (yellow tag) and PET-CT (B) (blue arrow) for 48-year-old female patient with history of urinary bladder urothelial carcinoma operated by VATS wedge resection
After MDT discussion, patients were allocated to receive either open thoracotomy (OT) (group 1) or VATS (group 2) based on the number, size and location of pulmonary metastasis as well as lung function. A limited number (≤ 2) of peripherally located nodules with good underlying lung function (to tolerate single-lung ventilation) were more likely to be allocated to the VATS group.
Surgical technique (VATS)
Under general anesthesia, with selective single-lung ventilation using a double-lumen endobronchial tube, the patient is positioned in full lateral decubitus position, and the operating table is flexed to widen the rib spaces on the operation side.
A small 10 mm long incision is done at the 8th/9th intercostal space at the anterior axillary line for camera entry. A 10-mm video thoracoscope with 30º lens is used. Exploration of the thoracic cavity is performed. A larger 30–40 mm working incision is placed between mid and anterior axillary lines at the 4th/5th intercostal space depending on the location of nodule(s) on upper/middle or lower lobes. A silicone (Alexis) wound retractor is used to keep the working incision open. If uniportal VATS is planned, the first 10 mm incision is omitted and the camera is placed at the most posterior part of the working incision.
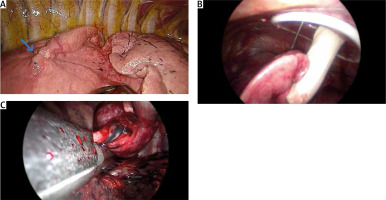
Any adhesions present were freed. Complete ipsilateral atelectasis improves visibility and instrument manipulation, and often allows the subpleural lesion to become visible once the lung is completely collapsed (Figure 2 A). If the lesion is not readily apparent, then palpation of the lung parenchyma with the aid of a blunt-tipped endoscopic instrument is done. Moreover, a Duval forceps grasping lung parenchyma towards the port with introduction of a palpating finger is also used to help identify lesions by digital palpation of the lung substance (Figure 2 B). Failure to readily localize the image-detected nodule/s will promote conversion to standard thoracotomy to achieve complete resection of the nodule/s.
Figure 2
Operative views in VATS surgery: A – visual identification of lung nodule (blue arrow) after atelectasis of lung by doublelumen ventilation, B – digital confirmation of the lung nodules and search for undetected lesions, C – wedge resection of peripheral lung nodule by linear stapler
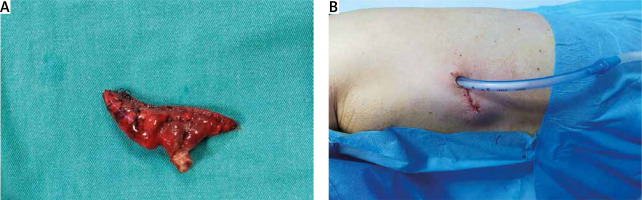
Peripherally located lesions were resected (wedge resection) with an Endo GIA Reload Covidien endoscopic stapler (Medtronic, United States) (Figure 2 C). All lesions were excised with a rim of ≥ 1cm grossly normal lung parenchyma (Figure 3 A).
Figure 3
Post-operative views of uniportal VATS wedge resection: A – wedge specimen with rim of normal lung parenchyma between nodule and staple line, B – uniportal VATS incision at 5th intercostal space with chest tube placed at same incision
More centrally located lesions not amenable to wedge resection or in the setting of multiple nodules in the same segment/lobe are resected by anatomical resection (segmentectomy or lobectomy, respectively) with hilar and mediastinal lymph node dissection. In wedge resection cases, lymph node sampling/dissection was done if suspicious nodes were detectable in preoperative imaging or intraoperatively.
Checking for an air leak was done in all cases then an intercostal 28 Fr chest tube was placed in the thoracic cavity before wound closure (Figure 3 B).
Post-operative follow-up
All patients had a chest X-ray done within 6 hours after surgery and before chest tube removal/patient discharge. Chest X-ray was repeated at the end of the 2nd post-operative week. Patients were reviewed 6 weeks after surgery, then after 3 months, and at 6-month intervals thereafter, or whenever they became symptomatic. Chest CT with IV contrast was done at the 3-month visit and then at 6-month intervals. After recovery from surgery and review of pathology, patients were referred to the tumor board group meetings to be assigned to adjuvant therapy – if indicated.
Statistical analysis
Prospectively collected patients’ data were expressed as numbers and percentages for qualitative variables (which were compared by the c2 test or Fisher’s exact test as appropriate) or as mean ± standard deviation for quantitative variables (which were tested by Mann-Whitney U test). For all statistical tests done, the threshold of significance was fixed at the 5% level (2-tailed unless otherwise specified). All analyses were performed using IBM SPSS Statistics ver. 24.0 (NY, USA: IBM Corp.).
Results
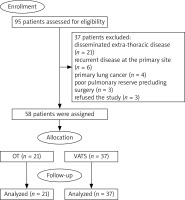
Between October 2017 and December 2020, a total of 95 patients were eligible for the study with mean age of 53.02 ±5.89 years. Thirty-seven patients were excluded from the study (21 patients had disseminated extra-thoracic disease, 6 patients had evidence of recurrent disease at the primary site, 4 patients proved to have primary lung cancer, 3 patients had poor pulmonary reserve precluding surgery and 3 patients refused the study). Fifty-eight patients were assigned to OT (n = 21) or VATS (n = 37). The consort flow diagram is presented in Figure 4.
Patients’ demographic data
The most common primary tumor was colorectal cancer, which represented 40% of our cases followed by breast cancer and soft tissue sarcoma. Fifty-nine percent of the cases were female with no significant differences between the two groups as regards mean age, gender distribution, BMI, or smoking status. Nodules were similarly distributed between right and left lungs as shown in Table I.
Table I
Clinicodemographic data of patients
More cases with a solitary nodule were in the VATS as compared to the OT group (78.38% vs. 28.57%, p < 0.001). Most of these (62.07%) were peripherally located with a significantly higher percentage in the thoracoscopic group (86.49% for VATS vs. 19.05% for OT, p < 0.001). More people with cN0 as evident in chest CT and PET-CT were located in the VATS group (p = 0.01). Cases in the VATS group had a significantly longer mean disease-free interval (DFI) between resection of the primary tumor and evidence of lung metastasis as compared to the OT group (p = 0.000) (Table I).
Operative data among the study population
Due to being more solitary and peripherally located, most of the VATS cases (81.08%) were resected by wedge resection using stapling devices. On the other hand, more than three-quarters of OT cases were resected by anatomical techniques (lobectomy and segmentectomy) (p < 0.001). The operative time was longer and the blood loss was larger in the OT group than the VATS group. Most cases in the OT group (85.71%) had lymph node sampling/dissection while only 30% of the VATS group had nodal assessment (p < 0.001). The mean number of retrieved lymph nodes was significantly higher in the OT group. More new nodules were likely to be detected during surgery in OT than VATS cases (p = 0.027). Twelve nodules that were not evident in the pre-operative imaging studies were detected during surgery for the OT group patients while only three more nodules were discovered in the VATS group patients. All nodules evident in the pre-operative imaging were successfully located during surgery in both OT and VATS procedures (Table II).
Table II
Operative data of patients
Post-operative short-term outcomes
Patients in the VATS group experienced fewer complications (prolonged air leak, atelectasis and surgical site infection) than the OT group despite not reaching statistical significance. Two cases of the OT group were taken back to the operative theatre (1 patient had post-lobectomy massive hemorrhage and the other patient had prolonged air leak not responding to conservative measures). Both the duration and amount of output drainage in the chest tube were in favor of VATS surgery (p < 0.0001). Length of hospital stay was shorter in the VATS group (p < 0.0001). There was no reported mortality in the post-operative period in our study (Table III).
Table III
Post-operative short-term outcomes
Pathologic outcomes
More than 90% of cases had R0 resection of all detectable nodules with no difference between the two groups while the mean width of the surgical margin (between the tumor border and the staple line) was significantly longer in the OT group than the VATS group (48.62 ±7.12 mm vs. 11.32 ±3.94 mm, p < 0.001).
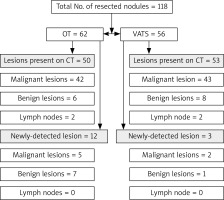
Seven out of the fifteen newly detected nodules (46.67%) were found to be metastatic malignant nodules while 7 (out of 12) in the OT group and 1 (out of 3) in the VATS group were found to be non-malignant (p = 0.44) (Table IV). These data are summarized in Figure 5.
Table IV
Pathologic outcomes
Figure 5
Pathologic results of resected lung nodules by OT and VATS: this diagram shows the number and nature (benign, subpleural lymph node or malignant) of all resected nodules, either those planned resections based on pre-operative imaging or newly detected nodules during surgery in OT and VATS groups
Discussion
Pulmonary metastasectomy (PM) has been established as a therapeutic option for pulmonary metastases of extra-thoracic solid organ malignancies in order to improve survival [2, 8, 9]. However, the appropriate approach for PM, whether OT or VATS, is still a debatable issue [7, 10].
Colorectal lung metastases represent the majority of cases of PM in the literature [11]. This finding was also observed in our study, in which metastatic colorectal cancer accounted for 39.7% of all our patients, followed by breast cancer (24.1%) then sarcoma (13.8%).
PM is used for patients with solitary or oligo-metastasis defined as five or fewer lesions [5] or metastasis amenable to curative therapy [4]. Solitary metastasis represented 60.3% of our patients. This reflects selection of patients by the MDT group aiming to take the maximum benefit from PM. Also, the majority of patients with solitary metastasis were treated via the VATS approach. This difference was statistically significant (p < 0.001). These cases were preferentially selected to be assigned to VATS rather than OT procedures. Most metastases in the VATS group were peripherally located and were treated by wedge resection (81.8%) using stapling devices. This is also explained by preoperative selection by the authors to facilitate the resection and decrease the conversion rate.
It has been believed that lung palpation through an open thoracotomy is the gold standard for detection of metastasis in comparison to VATS, which has a higher potential for incomplete resection due to loss of tactile feedback. This is enhanced by the low sensitivity of imaging in picking up pulmonary metastases [12, 13]. The previous concept has been challenged by five factors: i) advances in imaging modalities with thin-slice cuts which can detect nodules smaller than 2–5 mm [14], ii) advances in pre- and intra-operative localization techniques in hybrid operating rooms facilitating identification of all image-detectable lesions [15], iii) not all detectable lesions are found to be malignant [7, 16], iv) enhanced recovery offered by minimally invasive VATS procedures [17, 18] which enables faster initiation of adjuvant therapy (integral part of disease management), and v) the lack of survival benefit of OT over VATS has been consistently shown in many reports [19–21].
Increased detection of nodules by digital palpation when compared to imaging was reported by Macherey et al. [16]. However, nearly half (48.5%) of these lesions were benign. Moreover, the majority of imaging studies included scans with large slice thickness (5–10 mm) [16]. Similarly, Eckardt et al. found only 6 (20.7%) out of 29 newly detected nodules to be metastases, while 19 (65.5%) lesions were benign, 3 (10.3%) lesions were subpleural lymph nodes and one (3.4%) was a primary lung cancer (7). In our series, 15 more nodules were detected during surgery for planned resection of nodules (103 nodules). Twelve of these nodules were detected by OT as compared to only three nodules detected in VATS. However, most of these nodules (53.33%) were not malignant.
Patients with lung metastasis can be rendered free of disease only by means of complete R0 resection of all detectable nodules, which is the most important prognostic factor for long-term survival in PM [22, 23]. R0 resection can be obtained with either wedge or anatomical resection (lobectomy or segmentectomy). For most thoracic surgeons, a stapled segmental or parenchymal conserving resection is often the first choice to minimize risks of postoperative morbidity, while more extensive anatomic resections are reserved for larger, more centrally located or multiple tumors [24]. In this study, there was no detectable difference in the resection status of the pathological reports in either group. In patients who underwent VATS as well as in those who underwent thoracotomy, the majority of metastases were resected via R0 resection (VATS 89.2%, thoracotomy 95.2%).
It is worth noting that up to 50% of patients will experience disease recurrence during follow-up [2]. In more than 80% of cases, recurrence is located in the non-operated, contralateral lung or at a distant extra-thoracic site [25, 26]. Moreover, repeated thoracoscopic resection is more tolerated than repeated thoracotomy with fewer adhesions [27]. Therefore, PM should be a trade-off between benefits to improve survival and risks of pulmonary functional reserve compromise.
The therapeutic benefit of lymph node dissection in PM remains an unsettled issue. The involvement of mediastinal lymph nodes or unknown nodal status is associated with significantly decreased survival [28]. However, reports have been unable to establish differential survival based upon nodal sampling versus radical lymphadenectomy. Surgeons who favor systematic lymph node dissection argue that it can provide important prognostic data for better staging [29]. On the other hand, for many surgeons, nodal sampling or systematic lymphadenectomy is often only undertaken in the setting of suspicious nodes or biological markers of more aggressive disease. In a survey among members of the European Society of Thoracic Surgeons (ESTS), 68% of surgeons reported performing mediastinal lymph node sampling (55%) or complete lymphadenectomy (13%) for assigning to adjuvant therapy. Thirty-two percent of surgeons reported neither sampling nor systematic dissection [30]. It is notable that the vast majority of respondents considered the presence of mediastinal node metastasis as an absolute (64%) or relative (32%) contraindication to PM surgery [30].
In our study, the selection to perform lymph node sampling, systematic lymph node dissection or to omit lymphadenectomy is based on imaging criteria of both lung nodules and lymph nodes. Systematic lymph node dissection was performed significantly more often in patients who underwent OT in our study (p < 0.001). This may be because patients with multiple metastases or suspicious lymph nodes on imaging were preferably treated with an open procedure. In contrast, the rate of patients with cN0 lymph node status was higher in the VATS group, which certainly contributed to the decision to perform VATS.
In our study, operative time was significantly longer in OT than VATS (129.48 ±8.86 vs. 64.24 ±14.32 minutes). On the other hand, mean blood loss was significantly less in VATS than OT (56.19 ±8.23 vs. 277.62 ±17.52 ml). VATS has a lower risk of post-operative complications, such as pneumonia, atelectasis, prolonged air leaks and surgical site infection, with a lower ICU/HDU admission rate and shorter hospital stay compared to open procedures. These results are highly supported in the majority of reports [9, 17, 31, 32]. VATS is also associated with minimal trauma to tissues with less immune response, which can affect the disease progression [33]. These advantages make VATS an attractive alternative strategy for PM.
Our study has some limitations. First, patients included in the analysis were highly selected and might not be representative of all patients with pulmonary metastases. Second, the number of patients was small with a relatively short follow-up. Third, the decision to perform OT or VATS is author/MDT dependent, considering that the authors are in the learning curve in performing VATS anatomic lung resections. In general, in patients with large and/or centrally located tumors, an open approach was used. Small, peripheral located nodules were removed by VATS.
Conclusions
In a highly selected cohort of pulmonary metastasis patients with favorable criteria (peripherally located, small, solitary/oligo-metastasis and cN0), VATS may provide acceptable onco-pathologic outcomes as compared to the standard open thoracotomy. Further randomized controlled trials of large numbers of patients and longer follow-up are needed.