Introduction
Percutaneous balloon aortic valvuloplasty (BAV) has been widely used in high-risk patients since it was first introduced by Cribier et al. [1]. The technique has been questioned in many trials due to the limited hemodynamic effect and a high rate of restenosis [2–4]. Nowadays, the role of BAV in the management of severe symptomatic aortic stenosis (AoS) has come under the spotlight following the development of the transcatheter aortic valve implantation (TAVI) technique. From palliative and bridge to aortic valve implantation therapy, the indications have been widened to include symptomatic improvement before the consideration of definitive TAVI intervention [5, 6]. The primary access for BAV is still the femoral artery access, but also subclavian, brachial, and radial artery (RA) access has been reported in the literature [7–14]. The aim of the study was to investigate the efficacy and safety of the BAV procedure performed from transradial access.
Aim
The aim of our study was to establish the safety and the technical success of transradial balloon aortic valvuloplasty (trBAV). The secondary objective was to determine the effectiveness and appropriate role of trBAV.
Material and methods
Methods
Clinical and angiographic data from 36 consecutive patients with symptomatic AoS were evaluated in a prospective pilot single-center study. Four patients were excluded from the study due to the small size or occlusion of the radial artery. Between 2017 and 2019, the patients were treated utilizing bilateral radial or radial and contralateral ulnar artery access with 6–10 F compatible balloons. The decision of the BAV was made by the Semmelweis University Heart and Vascular Center Valve Team and all the procedures were performed in a hybrid operating room. The impact of the learning curve was analyzed after 10 trBAV cases. In 21 cases trBAV was used as a bridge to TAVI or surgical aortic valve replacement (SAVR) and in 12 patients to palliate the symptoms as destination therapy. In 2 cases trBAV was performed before acute non-cardiac surgery. Our Hungarian State Ethical Review Committee approved the study (OGYÉI/50275/2017), and all patients provided written informed consent prior to study inclusion.
Inclusion criteria
We included high-risk patients with symptomatic significant AoS in the study after written informed consent.
Exclusion criteria
Preexisting aortic prosthesis, recent myocardial infarction, left ventricular or atrial thrombus, severe aortic, mitral or tricuspid regurgitation (grade III–IV), recent cerebrovascular event (within 3 months), carotid or vertebral artery stenosis (> 80%), active internal bleeding, thrombocytopenia (platelet count < 50,000/mm3), lack of written informed consent, severe mental disorder, drug/alcohol addiction, life expectancy < 1/2 year, participation in another drug or device study, pregnancy, and suboptimal vascular anatomy: patients with occluded, severely diseased or small (< 1.5 mm) radial or ulnar arteries on both sides were not included.
Endpoints
Our primary endpoints were technical success and major adverse events (MAE). The secondary endpoints were the access site complication rate, hemodynamic and clinical result of the intervention, procedure-related factors, and crossover rate to the femoral access site.
Duplex ultrasound (US) protocol
Duplex US was used in the operating room to investigate all forearm arteries. Radial artery diameter, ulnar artery (UA) diameter, and peak systolic velocity were measured at the wrist level. On the first postoperative day, all patients underwent follow-up Duplex US.
Access site selection
Two skilled operators trained in bilateral transradial access, BAV and peripheral vascular intervention performed all cases. The preferred access site for trBAV was the right RA (n = 21), the alternative access site was the left RA (n = 14) and in 1 case the right UA, which was used when Doppler ultrasound showed a small (less than 1.5 mm) and extremely calcified radial artery. Aortography was performed from the contralateral RA approach, except 1 case, when UA was used. Rapid pacing was performed via the femoral or jugular vein.
Antithrombotic regimen
All patients with known coronary artery disease were treated with daily 100 mg aspirin; the patients who underwent coronary stenting received dual antiplatelet therapy (aspirin 100 mg and clopidogrel 75 mg) for 6 months. In addition, 5000 IU heparin sodium, and 250 µg nitroglycerine were administered directly into the radial artery through the sheath. Additional Na-heparin was given until reaching 70 IU/kg. Routine ACT was not measured during the intervention.
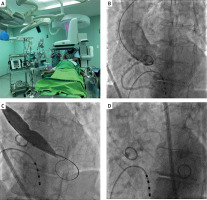
TrBAV technique (Figure 1)
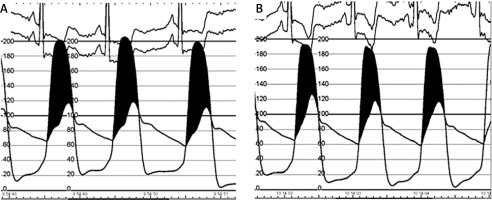
Puncture: After local anesthesia, the RA or UA was punctured under ultrasonography guidance with a dedicated transradial needle and sheath (Terumo Co., Japan, 5F). Aortography was performed using a pigtail catheter in a left lateral 30-degree view with 15 ml of contrast. RA puncture was carried out in the conventional or distal radial artery access (snuff box). The TrBAV procedure was started with a US based puncture of the right contralateral radial or ulnar artery. The aortic valve was passed with a Terumo Guidewire using Amplatz left 1 or 2 diagnostic catheters and then the Terumo GW was replaced with a 260 cm J tip 0.035’’ guidewire. The Amplatz catheter was replaced with a diagnostic pigtail catheter and the baseline pressure gradient was measured. After the baseline pressure recordings, the Amplatz Super stiff or Safari guidewire (Boston Sci, USA) was advanced into the left ventricle. At this point, the radial sheath was removed and a short hydrophilic femoral sheath (Terumo, Japan) was advanced in the RA through the 0.035 GW. The size of the introducer was selected as specified by the US measurement of the access site and the entry profile of the preferred balloon. According to the preoperative US parameters, there were no access site/introducer disproportion. The procedure was performed with 16–22 mm balloons under conventional rapid pacing. The balloon diameter was calculated from the preoperative thoracic CTA or echocardiography measurements (balloon to annulus diameter ratio 0.8). After balloon removal, the pressure measurements were made with the pigtail catheter again. Invasive hemodynamic measurement is presented in Figure 2. The second balloon dilatation was the operator’s decision.
Postoperative treatment
After the procedure, the sheath was immediately removed, and hemostasis was achieved by applying the Terumo Band dedicated compression device (Terumo, Japan) to the radial access site for 6 h. The femoral venous sheath was removed immediately and a compression bandage was applied for 6 h. During the postprocedural care, overnight intensive care unit observation was conducted.
Post-procedural follow-up
After the procedure, all patients underwent physical examination and echocardiography measurements. All patients were scheduled for a detailed clinical follow-up examination at 3, 6, and 12 months after the procedure.
Definitions
Major adverse events (MAE): MAE were assessed as the composite of death, stroke, myocardial infarction, and urgent major aortic valve replacement or implantation during the hospital stay or at the 1-month follow-up.
Definition of vascular complications: Major vascular complication was defined as diminished or lost arterial pulse or the presence of any pseudo-aneurysm or arteriovenous fistula during the clinical follow-up. Minor complications were defined as hematomas requiring no further treatment (EASY (Early Discharge After Transradial Stenting of Coronary Arteries Study) 1–2), measuring 2 cm in diameter over the radial or ulnar puncture area, or measuring 5 cm in diameter over the femoral puncture site. Major bleeding was defined as a drop in the hemoglobin level of > 3 g/dl, as well as any bleeding requiring blood transfusions. Major hematoma was defined as EASY 3–4 hematoma [7].
Technical success was defined as successful valvuloplasty balloon inflation at the aortic valve.
Hemodynamic success was defined as at least 25% drop in peak aortic gradient.
Clinical success: Primary clinical success was defined as an improvement of at least one clinical category in the NYHA classification.
Statistical analysis
Statistical analysis was performed using the commercially available Graph Pad Prism 8.0 software (USA). Continuous variables were expressed as the mean ± standard deviation or as the median with inter-quartile range. Categorical variables were tabulated as percentages. The different patient cohorts were compared using either the Mann-Whitney U test or the Kruskal-Wallis test. Probability values lower than 0.05 were considered to be significant.
Results
TrBAV was performed in 36 patients with significant AoS, using bi-radial RA (n = 34, 94.4%) or hybrid (radial and ulnar) (n = 2, 5.6%) access. The access site for pacemaker insertion was the femoral vein in 3 (8.3%) or jugular vein in 20 (55.5%) patients. In 10 cases the rapid pacing was done through the left ventricular guidewire. The indication for trBAV was the bridge to TAVI in 20 patients (55.5%), bridge to surgical valve replacement in 1 (2.7%) case, cardiogenic shock in 1 (2.7%) case, before acute non-cardiac surgery in 2 (5.5%) cases and destination therapy in 12 (33.3%) cases. Demographic and clinical data are summarized in Table I.
Table I
Demographic and clinical data
Procedural data are summarized in Table II. Technical success was achieved in all patients (100%). Clinical success was achieved in 36 (100%) patients. Hemodynamic success was achieved in 20 (55.6%) patients. In hemodynamic investigation, the peak-to-peak gradient decreased from 76.8 ±27.2 to 54.7 ±21.1 mm Hg (p = 0.001). Balloon angioplasty was performed in all cases, and the cross-over to urgent TAVI or surgical aortic valve implantation was 0%. Single balloon and dual trBAV were performed in 35 (97.2%) and 1 (2.7%) cases, respectively. Additional coronary angioplasty was done in the same session in 4 (11.1%) cases. The cross-over rate to the femoral access site was 0%. The fluoroscopy time, X-ray dose, procedure time, and contrast consumption were 10.10 ±7.3 min, 319.8 ±484.6 mGy, 50.3 ±28.1 min, and 64.9 ±28.1 ml, respectively. Ultrasonography parameters before and after the procedure are summarized in Table II.
Table II
Procedural results
Complications after the procedure and during follow-up are summarized in Table III.
Table III
Complications and clinical follow-up
Major procedural complications were not detected. RA or UA access site complications were encountered in 2 (5.5%) patients (1 asymptomatic RA occlusion and 1 forearm hematoma successfully treated with forearm bandage). Left distal radial access was used in 18 patients without any complication (0%). There was no significant difference in the complication rate between proximal radial or ulnar (n = 54) and distal radial access (n = 18) (complication rate 3.7% and 0%). The cumulative incidence of MAE at 3- and 12-month follow-up was 16.6% and 30.5%, respectively. The cumulative incidence of death at 3- and 12-month follow-up was 13.9% and 27.7%. The patients from the bridge to TAVI list underwent successful TAVI procedures in 11 (30.5 %) cases. One-year mortality in the successful bridge to TAVI or SAVR group vs. other patients was 25% vs. 33.3% (p = NS).
Discussion
At the beginning of the valvulotomy era, the acute hemodynamic and clinical results of BAV were optimistic [1–4], although the limitations of the procedure were identified soon after. At 6 months, the gradients are between baseline and post-BAV values, and the valvular restenosis rate is high, up to 50–70%, at 1 year, but symptomatic relief can extend up to 1.5–3 years [9]. Francesco Saia et al. analyzed 405 patients who underwent elective and emergency BAV and reported that cumulative 1-year and 2-year mortality rates were 33.2% and 57.4%, respectively, with the highest incidence in the cardiogenic shock group (70.7% and 80.4%) and the lowest in the bridge to AVR group (21.7% and 38.4%). Other events were stroke (0.5%), major vascular complications (2.2%), and life-threatening bleeding (1.5%) [5].
In the past decade with the rise of the TAVI procedures, the need for BAV emerged by reason of bridge to TAVI indications [12]. The fragility of the patients who undergo BAV before TAVI is high; in addition, most of the procedures are performed in acute settings. Improvement of the different tools and techniques used to perform BAV is remarkable, and the procedure-related complication rates were reduced during the past decades; nevertheless, most of the problems are related to the access site of the procedure.
Vascular complications at the puncture site are the most common issues associated with BAV [13]. Vascular complications in the literature are reported at the rate of 5–10%, even after the initiation of vascular closure devices. In order to avoid femoral artery access site complications during BAV and TAVI, the ultrasound or angiography-guided femoral artery puncture, the use of closure devices [13, 14], and the use of alternative access sites were introduced.
Alternative access sites for BAV are the brachial, axillary, and radial arteries [15, 16]. The brachial and axillary access sites can accommodate large access sites for BAV and TAVI and the puncture site can be closed with a Perclose device, but brachial access for endovascular procedures is associated with a high rate of thrombotic and bleeding complications in large series and these complications need surgical repair in most cases [16, 17].
The transradial approach for BAV has been presented in case reports and pilot studies [18–21]. The most common advantage of the technique is the lower rate of major vascular access complications and fast mobilization. The limitations of trBAV technique are mainly anatomical. The size and pathology of the radial artery must be investigated by vascular ultrasound. In many cases, the radial artery cannot accommodate big sheaths, due to calcification, size, or stenosis/occlusion. In these cases, the alternative access site can be the brachial or the femoral artery. The size of the balloon is very important to select the size of the sheath. The size of the balloon is determined by the perimeter of the annulus. The diameter of the delivery sheath can be decreased with the use of peripheral balloons or by decreasing the size of the balloon. Operators can select from semi-compliant (Tyshak II, Braun International Systems) or non-compliant aortic balloons (Z-Med, Braun International Systems; Maxi LD Balloon, Cordis Corporation; XXL balloon, Boston Scientific). Semicompliant balloons tend to have a lower profile and therefore require smaller vascular access sheaths which help with reduced vascular complications in this elderly cohort. The trade-off, however, is that they have less predictable inflation diameters than the non-compliant balloons and have lower rated burst pressures. Dedicated BAV balloons have a larger entry profile, but peripheral balloons can be used in smaller sheaths (Table IV). Smaller balloons can have a smaller hemodynamic effect than larger balloons. In our study, the 0.8 annulus to balloon ratio was safe and had a satisfactory clinical effect, but the hemodynamic effect was not always improved. In some cases, the use of 7F sheathless guiding catheters allows the use of balloons when the delivery of the balloons is difficult.
Table IV
Distribution of the applied balloon types, balloon diameters and final introducer sizes
In our patient population, we did not experience any major vascular event and the rate of minor events was 5.5%. We have used distal radial access in 18 patients without any complications (0%). Our data suggest that the vascular complication rate can be decreased by transradial access preference and it can improve the mortality and morbidity of the acute phase.
Study limitations: The primary limitation of the study is the small number of patients and the lack of randomization with the femoral approach. Another limitation of the study was the ultrasound-based balloon size selection.
Conclusions
The main findings of the study are the following: (1) trBAV is a safe and technically feasible procedure under the US control of the access artery size; (2) the vascular complication rate of the procedure is low; (3) 1-year mortality remains high in patients who are not candidates for TAVI and the procedure cannot significantly improve the mortality rate.
According to our study, bilateral radial artery access is a safe and effective option for balloon aortic valvuloplasty in patients with severe aortic valve stenosis and an optimal RA diameter. Long-term mortality is still high due to comorbidities and associated diseases.