Introduction
Anatomical lung resection (lobectomy and segmentectomy) is the mainstay treatment for early-stage non-small cell lung cancer (NSCLC) [1]. Owing to several advantages, such as less surgical trauma, a lower complication rate and improved postoperative rehabilitation, a majority of the aforementioned operations are currently performed through the video-assisted thoracic surgery (VATS) approach [2, 3]. Pneumonectomy, as a significantly more extensive procedure, is applied in advanced cases of NSCLC, including centrally located tumors invading large vessels or the main bronchus. Due to this reason, operations are usually carried out through the conventional thoracotomy approach. Of note, in carefully selected cases, the minimally invasive approach for pneumonectomy may be an intriguing option [4]. However, contrary to VATS lobectomy or segmentectomy, minimally invasive pneumonectomy is not a routine procedure; thus clinical studies dealing with particularly long-term outcomes should be widespread among the thoracic surgical community.
Aim
The aim of this study was to analyze the feasibility of VATS pneumonectomy, including the qualification criteria and surgical technique, as well as to evaluate the short- and long-term outcomes of VATS pneumonectomies performed in a single thoracic surgical center.
Material and methods
The Local Bioethical Committee waived the need for ethics approval and the need to obtain written consent for the collection, analysis and publication of the retrospectively collected and anonymized data for this non-interventional study.
Demographics, preoperative evaluation, and qualification for surgery
The medical records of all patients who underwent lung anatomical resection from September 1, 2010, to January 31, 2020 were reviewed. The exclusion criteria were: planned open approach (thoracotomy) and extent of resection lesser than pneumonectomy (lobectomy, sublobar resection). All patients who underwent VATS pneumonectomy were included.
During preoperative assessment, all patients underwent a detailed interview, physical examination, chest computed tomography (CT) scan, abdominal ultrasound, electrocardiography, pulmonary function tests (PFTs), and fiberoptic bronchoscopy. If indicated, echocardiography, exercise tests, diffusion lung capacity for carbon monoxide (DLCO), and positron emission tomography-computed tomography (PET-CT) were performed. Perioperative risk was assessed according to the American Society of Anesthesiologists (ASA) scale, the Thoracic Revised Cardiac Risk Index (ThRCRI) and European Risk Models for Morbidity (EuroLung1) and Mortality (EuroLung2). The predicted postoperative pulmonary function was calculated on the basis of PFTs (FEV1 and FVC) after considering the extent of lung parenchyma involvement by the tumor as well as the bronchial obturation observed in fiberoptic bronchoscopy.
Patients qualified for VATS pneumonectomy only when less extensive (sleeve) resection was not possible and it was certain that VATS could be performed safely, allowing for complete (R0) resection. The detailed qualification criteria were as follows:
Invasion of the distal part of the main bronchus or pulmonary artery when sleeve resection was not possible.
Invasion of two (left-sided) or three (right-sided) lobes.
Tumor with metastasis to the other lobe of the same lung after exclusion of distant metastases in PET-CT, when less extensive resection (lobectomy with wedge resection or Nd:YAG laser resection) was not possible.
Patients did not qualify for VATS pneumonectomy in the following situations:
Anesthesia, surgical approach, and technique
Surgery was performed under general anesthesia with double-lumen intubation and single lung ventilation. Patients were placed in the lateral decubitus position with flexion of the surgical table at the level of the scapula tip. Two types of VATS approach were applied: 3-portal VATS (3P-VATS) and uniportal VATS (U-VATS). In the case of 3P-VATS, a 5 cm long utility incision in the 4th or 5th intercostal space anterior to the latissimus dorsi muscle was performed, and two ports were placed in the 7th intercostal space of the anterior axillary line and in the 8th intercostal space of the posterior axillary line. In the case of U-VATS, only a 5 cm long utility incision was made in the 4th or 5th intercostal space. No rib spreader was used. However, in U-VATS, an Alexis wound protector/retractor (Applied Medical, Rancho Santa Margarita, CA) was placed for utility incision. A 30° camera was introduced through the anterior port in the case of 3P-VATS or through the posterior portion of the utility incision in U-VATS. An endoscopic dissector, endoscopic hook, and LigaSure (Medtronic, Dublin, Ireland) were used for soft tissue dissection and lymphadenectomy. The pulmonary vessels and bronchus were sutured and divided with EndoGia endostaplers (Medtronic, Dublin, Ireland): the veins and arteries with vascular (white or gold) and the bronchus with violet reloads. The typical steps involved in the surgery were as follows:
On the right side, dissection of the pulmonary ligament and posterior mediastinal pleura; stations 9, 8, and 7 lymph nodes; upper pulmonary vein; right pulmonary artery; lower pulmonary vein; stations 10, 4, and 2 lymph nodes; and the main bronchus was performed.
On the left side, the dissection of the upper pulmonary vein; stations 5 and 6 lymph nodes; left pulmonary artery; lower pulmonary vein; stations 7, 8, and 4 lymph nodes; and the main bronchus was performed.
The lung was removed from the pleural cavity in an endobag. The bronchial stump was submerged under water and checked for any air leak. The bronchial stump was not covered with tissue flaps. If an epidural anesthesia catheter was not placed in the patient before surgery, paravertebral block with bupivacaine was performed at this time point. One 28-F chest tube was inserted through the anterior port (3P-VATS) or the posterior part of the utility incision (U-VATS).
After surgery, all patients were initially admitted to the Intensive Care Unit and, subsequently, transferred to the Thoracic Surgery Department. The patients were released from the hospital if there were no complications and the follow-up fiberoptic bronchoscopy was normal. Short- and long-term results, including probability of survival according to the Kaplan-Meier method, were assessed.
Data presentation
Normally distributed continuous variables are presented as mean with standard deviation (mean ± SD) whereas the others are presented as median with the range (minimum–maximum). Categorical data are expressed as numbers (n) with percentages (%). Probability of survival was assessed according to the Kaplan-Meier method.
Results
From a total of 324 patients who underwent pneumonectomy between September 1, 2010, and January 31, 2020, 19 (5.9%) patients were qualified for VATS pneumonectomy. The study group comprised 15 (78.9%) men and 4 (21.1%) women, mean age of 62.6 ±5.5 years (range: 50–73 years). Data on the demographics are presented in Table I.
Table I
Patient demographics (n = 19)
| Characteristic | Value |
|---|---|
| Age [years]a | 62.6 ±5.5 (50–73) |
| Sex, n (%): | |
| Male | 15 (78.9) |
| Female | 4 (21.1) |
| Comorbidities, n (%): | |
| Hypertension | 9 (47.4) |
| Chronic obstructive pulmonary disease | 9 (47.4) |
| Diabetes mellitus | 4 (21.0) |
| Coronary heart disease | 2 (10.5) |
| Pulmonary function testsa: | |
| FEV1% | 77.9 ±22.6% |
| FVC% | 86.2 ±24.9% |
| ASA scale, n (%): | |
| 2 | 9 (47.4) |
| 3 | 10 (63.6) |
| ThRCRI, n (%): | |
| A | 17 (89.5) |
| B | 2 (10.5) |
Seventeen (89.5%) patients were operated on by 3-portal and 2 (10.5%) patients by uniportal VATS. Conversion to anterolateral thoracotomy was necessary in 2 (10.5%) patients.
Patients with VATS pneumonectomy converted to thoracotomy
In the first case, the patient with class 3 obesity (BMI > 40 kg/m2) and a large (7.1 cm diameter) right lung tumor was qualified for 3-portal VATS pneumonectomy. Because of the high position of the diaphragm, tumor dimension, and the partially expanded lung, the space in the pleural cavity was extremely limited. During dissection of the soft tissues in the area of A1/A3 arteries and the azygos vein, dark blood bleeding occurred. Because of difficult anatomical conditions, determination of the bleeding source was not possible. The bleeding area was compressed, thoracotomy was performed, and the azygos vein injury was located and sutured. The remaining part of the surgery was uneventful. The surgery time was 105 min, and the estimated blood loss was 800 ml. Blood transfusion was not deemed necessary. The postoperative course was uneventful, the chest tube was removed on POD 3, and the patient was discharged on POD 8.
In the second case, the patient qualified for left 3-portal VATS pneumonectomy because of squamous-cell carcinoma infiltrating the upper and lower lobar bronchi. A major part of the surgery was uneventful. However, after division of the main bronchus with the endostapler, the suction catheter tip was found to be embedded in the bronchial suture line. Thoracotomy was performed, the bronchial stump was shortened, and the catheter was removed, followed by closing the bronchus with a running suture. The surgery time was 100 min, and the estimated blood loss was 100 ml. The postoperative course was uneventful, the chest tube was removed on POD 5, and the patient was discharged on POD 10.
Patients with pneumonectomies completed by VATS
Among the group of 17 patients in whom surgery was completed by VATS, the mean surgery time was 121 min (range: 85–200 min) and the median estimated blood loss was 147 ml (50–300 ml). The median chest drainage time was 3 days (range: 2–4 days), and the median hospital stay was 8 days (range: 5–90 days). Postoperative complications occurred in 6 (35.3%) of the 17 patients in this group:
Atrial fibrillation in 2 patients.
Respiratory failure, sepsis and multi-organ dysfunction syndrome in 1 patient.
Esophageal perforation which required re-thoracotomy with suturing of the perforation on POD 6, and subsequent omentopexy, gastrostomy, and open thoracostomy on POD 15 because of recurrence.
Atrial fibrillation and subsequent superior mesenteric artery embolism on POD 4 in 1 patient, which required small intestine resection and total parenteral nutrition.
Bronchopleural fistula diagnosed on POD 5 in 1 patient. Despite chest tube placement, the patient’s condition deteriorated rapidly, with sepsis, multi-organ dysfunction and death on POD 5. In this patient, during the surgery, a small air leak was noted from the bronchial stump. The stump was then additionally sutured with a running suture and confirmed to be airtight.
One patient required readmission because of empyema of the postpneumonectomy space without bronchopleural fistula. Treatment with chest drainage and antibiotics was successful (Table II).
Table II
Indications for VATS pneumonectomy
| Indicationa | Value |
|---|---|
| Infiltration of distal part of main bronchus | 12 (63.1) |
| Peripheral infiltration of two or three lobes of the lung | 6 (31.6) |
| Tumor with metastasis to other lobe not amenable for less extensive resection | 1 (5.3) |
Results of histopathological examination
Squamous cell carcinoma was the most common type of NSCLC and was diagnosed in 15 (79.0%) patients. Most patients were in clinical stage II or III according to the 8th TNM classification (Table III).
Table III
Results of histopathological examination
| Characteristic | Value |
|---|---|
| Histology, n (%): | |
| Squamous cell carcinoma | 15 (79.0) |
| Large-cell carcinoma | 2 (10.5) |
| Adenocarcinoma | 2 (10.5) |
| Maximal tumor diameter [mm]a | 42 ±12 (29–71) |
| Clinical stage, n (%): | |
| I | 2 (11.1) |
| II | 9 (47.5) |
| III | 8 (44.4) |
| Lymph node metastases, n (%): | |
| N0 | 6 (33.3) |
| N1 | 9 (47.5) |
| N2 | 4 (22.2) |
| Bronchial margin width in NSCLC [mm]a: | |
| All patients | 11.4 ±4.2 (4–20) |
| Non-converted VATS (n = 17) | 11.9 ±4.1 (4–20) |
| Conversion to thoracotomy (n = 2) | 7.5 ±2.5 (5–10) |
| Complete resection (R0) in NSCLC, n (%) | 19 (100) |
Resection was complete (R0) in all patients with NSCLC, both completed by VATS and converted to thoracotomy. The mean distance from the tumor to the bronchial suture line was 11.4 ±4.2 mm, range: 4–20 mm. In 1 patient with a 4 mm margin, dysplastic epithelial cells were found in the suture line; however, at 33 months no local recurrence was observed.
Adjuvant treatment and follow-up
A total of twelve of 19 (63.2%) patients received adjuvant treatment, of whom ten received chemotherapy and two received chemo- and radiotherapy.
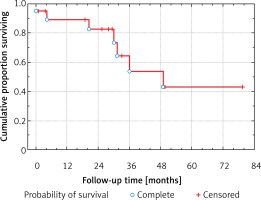
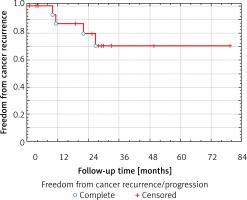
The median follow-up time was 882 days (range: 105–2614 days). Progression of the disease was noted in 4 patients, with distant metastases in 1, chest wall metastases in another 1, and mediastinal recurrence in 2 patients. The mean time to progression was 529 days (range: 300–806 days). A total of 7 patients died during the follow-up: 1 because of surgical complications, 4 as a result of cancer progression, and 2 for reasons not related to cancer and its treatment. Median survival was 74 months (interquartile range (IQR) = 49–106 months). One-, two- and 5-year probability of survival estimated by means of the Kaplan-Meier method was 0.88 ±0.07, 0.82 ±0.09 and 0.43 ±0.15, respectively (Figure 1). Estimated 1-, 2- and 5-year freedom from lung cancer recurrence after VATS pneumonectomy was 0.87 ±0.09, 0.79 ±0.11 and 0.71 ±0.13, respectively (Figure 2).
Discussion
Pneumonectomy is one of the most extensive operations performed by thoracic surgeons. It is associated with higher incidence of complications compared with lesser resections and can significantly impair a patient’s fitness and quality of life, both in short- and long-term perspectives. Pneumonectomy is generally performed through an open approach, usually by antero-lateral or postero-lateral thoracotomy. However, with the development of minimally invasive thoracic surgery, such operations are now even conducted through the minimally invasive approach. The VATS approach was introduced to thoracic surgery in the 1990s. Initially, it was used for less extensive procedures, but it was gradually adopted for anatomical lung resections [5, 6]. VATS lobectomy was proved to be associated with less surgical trauma, decreased postoperative pain, a lower complication rate, especially in terms of pulmonary complications (e.g., atelectasis and pneumonia), improved postoperative rehabilitation, shorter hospital stay, and better quality of life compared with the thoracotomy approach [7, 8]. Despite the fact that in many countries, the cost of VATS anatomical lung resection far exceeds the cost of thoracotomy, the former became the preferred approach in major thoracic surgery centers worldwide because of its evident clinical benefits [9, 10].
The rapid development of minimally invasive thoracic surgery did not, however, involve VATS pneumonectomy. Despite the fact that the first few VATS pneumonectomies were performed in the early 1990s, the number of procedures performed worldwide remained low [11]. Our experience was also similar. Although a minimally invasive surgery program was started at our center in 1999 and the total number of VATS lobectomies performed for oncological and other indications exceeded 1000, the numbers of attempted VATS pneumonectomies is low. The most important reason for this is probably the fact that the majority of the patients are qualified for pneumonectomy because of more locally advanced tumors and usually larger, invading anatomical structures of the pulmonary hilum (pulmonary artery and vein and main bronchus) or the chest wall [12]. Situations in which the patient can be qualified for the VATS approach (e.g., smaller tumors invading the distal part of the main bronchus, peripheral infiltration of two or three lobes of the lung or peripheral tumor with hilar lymph node metastasis, and tumor with metastasis to other lobes not amenable for less extensive resection) are much less common. Therefore, only a very selected group of patients is eligible to undergo VATS.
It must be emphasized that during the process of qualification for surgery, the possibility of performing sleeve resection should always be excluded. It has been proven that sleeve lobectomy is superior to pneumonectomy in terms of postoperative complications and the quality of life and provides comparable long-term results [13, 14]. Although cases of VATS sleeve lobectomies have been described, these operations are extremely demanding from the surgical perspective and rarely performed [15, 16]. Hence, if it is not possible to unequivocally exclude sleeve lobectomy based on the results of preoperative studies, thoracotomy should be considered.
The preferred surgical approach in the case of VATS pneumonectomy is similar to that in the case of VATS lobectomy and so is the mode of division of the pulmonary vessels. Because of the potentially catastrophic consequences of eventual pulmonary artery injury, particular care must be taken during its dissection and division. In the case of centrally located tumors, the mobility of the hilum is limited and the length of the vessel for stapler application is short; therefore, the traction must be performed very carefully. Bleeding from the vessel staple line may sometimes occur, but it is rare and usually self-limiting. If there are any doubts regarding the quality of staplers, a vascular clamp can be applied proximally around the artery before stapler placement [17]. Another useful technique, described in the case of right U-VATS with a large artery, is the separate division of the Boyden trunk first and the intermediate artery afterwards [18]. On the left side, prior dissection of the lymph nodes of the aorto-pulmonary window facilitates left main pulmonary dissection and division.
Division of the main bronchus is the last step during VATS pneumonectomy. It is of utmost importance to obtain sufficient distance between the tumor and bronchial suture line in case of main bronchus invasion so as to achieve complete resection (R0). Our results indicated that particular attention should be paid regarding this aspect in patients operated on the right side because of the shorter length of the right main bronchus, as well as in patients with more proximal tumor invasion. Because the intraoperative manual examination is virtually impossible during VATS, preoperative fiberoptic bronchoscopy by the operating surgeon and intraoperative frozen section of the resected bronchial line are mandatory. Another important aspect is to obtain the shortest possible bronchial stump in order to diminish the risk of bronchopleural fistula. In this aspect, the right side does not usually pose any particular problem. On the left side, however, it may be more difficult because of the location of the left main bronchus under the aortic arch. Applying more traction to the bronchus can help obtain a short bronchial stump [17, 19].
The issue of conversion to thoracotomy in the case of VATS pneumonectomy has been relatively poorly researched. We adopted the division of causes of conversion into three groups: “oncologic”, “technical”, and “complications” [20]. In this study, the conversion was necessary in 2 patients owing to complications (bleeding) and technical reasons (suction catheter tip in the bronchial suture line). Despite increased intraoperative blood loss in the first patient, the postoperative period for both patients was uncomplicated and the fact of conversion did not lead to an increased postoperative complication rate. The conversion rate of VATS pneumonectomy reported in the literature is varied and falls between 2% and 25% [21]. It must be stressed that except for emergent indications, the conversion has not been proven to be related to the postoperative complication rate and thus should be not considered as an indicator of the surgery quality [22].
The complication rate after pneumonectomy seems to be similar after VATS and thoracotomy [23]. Although it has been observed that the postoperative pain was lower after VATS pneumonectomy as compared with that after thoracotomy, it did not translate into lower incidence of pulmonary complications [24]. In our case series, most of the complications were not directly related to the minimally invasive approach. In only 2 patients, the cause of complication was strictly surgical. Esophageal perforation, due to its delayed occurrence, could have been caused by thermal injury resulting from the use of monopolar electrocautery during dissection of the subcarinal and paraoesophageal lymph nodes. The use of high-energy devices, which were unavailable at that time, could have presumably prevented esophageal injury. Bronchopleural fistula occurred in a patient operated on the left side, in whom the bronchial stump air leak was detected after division of the bronchus during surgery. After applying an additional running suture, the stump was airtight. Because the stump was short and located deeply under the aortic arch as well as surrounded by the neighboring tissues, covering it with a pedicled tissue flap was not considered necessary. It is uncertain, but not likely, that this maneuver would have prevented bronchopleural fistula.
Most of the studies published so far have focused on early VATS pneumonectomy results, while only a few have analyzed long-term outcomes. In our study, despite the limited number of patients, long-term outcomes seem to be good. Only 4 patients out of 19 had recurrence of cancer during follow-up, which presumably resulted from the complete resection (R0) obtained in all cases and at the same time confirmed the oncological effectiveness of VATS pneumonectomy. Median survival was 74 months, and 43% of patients survived 5 years after surgery. Comparable results were obtained by Battoo et al. – median survival in stage I–II was 80 months and in stage III–IV was 42 months [25]. In a multi-center study conducted by Yang et al. which included the largest group of patients so far (359 patients after pneumonectomy, including 124 operated on by the thoracoscopic approach), 5-year survival after VATS pneumonectomy was 47%. It is worth mentioning that the authors found no significant differences in overall survival between the open and VATS group [26]. The results of all those studies suggest that VATS pneumonectomy allows one to achieve good outcomes of treatment.
The most important limitation of the study was the relatively low number of patients included. This was due mainly to two reasons: first, the number of pneumonectomies performed in NSCLC is low and is still decreasing; secondly, the criteria for qualifying for VATS pneumonectomy are very narrow. Another limitation was the lack of comparison of VATS to a thoracotomy group. Both problems could probably be solved by conducting a retrospective, multicenter study with propensity score matching of VATS and thoracotomy patients. A prospective study with randomization of patients is unlikely to be feasible due to the long time that would be necessary to gather the appropriate group of patients.