Introduction
Age dominates risk factors for cardiovascular disease (CVD) [1, 2]. Indeed, the advent of contemporary treatments for acute coronary syndromes and stroke have helped to extend life expectancy [3]. Although an enormous success from an individual perspective, the resultant demographic shift presents one of the greatest challenges for the social and health care systems worldwide [4]. The population over 65 years of age will double from 12% in 2010 to 22% in 2040 [2]. Indeed, by 2020, the number of people 60 years of age and older will surpass the number of children below 5 years of age. Although aging presents an array of disorders [5], CVD carries the greatest burden for older persons, their care givers, and health systems [6]. Coronary heart disease (CHD) associates strongly with age, and it is the leading cause of death in Europe and the United States [7–9]. The prevalence of CVD increases in people > 65 years of age, especially in those > 80 years of age, and will increase by 10% over the next 20 years [2]. From 2010 to 2030, an additional 27 million people will have hypertension, 8 million will have CHD, 4 million will have stroke, and 3 million will have heart failure due to the rapid accumulation of the elderly [3]. The cardiovascular system shows several modifications with advancing age [10] in all its sections. These modifications are related to the inflammatory reactions activated by several endogenous and exogenous stimuli. Stem cells emerged as new therapy to reduce the ageing related damage in the cardiovascular system. Additionally, other intervention measures have been discovered. Based on these observations, we provide an overview on emerging literature data about the role of stem cells and new intervention measures as emerging drugs for inflammation and vascular ageing in cardiac surgery.
Stem cells as therapeutic option in cardiovascular disease
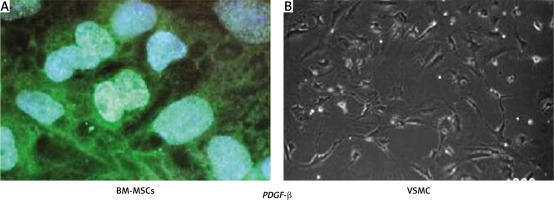
Tissue loss follows heart damage in consequence of the activation of a remodeling process, in which the original cardiac tissue is replaced by a scar [11] (Figure 1). Cardiac nonmyocyte cells, including cardiac mesenchymal stem cells (CMSCs), currently represent candidate targets for novel therapeutic strategies to counteract heart diseases [12]. Once activated, CMSCs contribute to the re-establishment of all the cardiac cell subpopulations, including cardiomyocytes [13]. Indeed, because of their plasticity, reduced immunogenicity, and the relative simplicity of isolation and culture ex vivo, CMSCs are a suitable adult stem cell population to consider for therapies based on cardiac regeneration. Recently, a significant effort has been made to enhance CMSC differentiation toward the cardiac lineage. As an example, the generation of induced pluripotent stem cells (iPS) starting from fibroblasts opened a new era for cell manipulation [14]. In 2010, Ieda et al. obtained for the first time induced cardiomyocytes (iCMs) from murine adult cardiac and skin fibroblasts by co-transduction of well-characterized cardiomyogenic transcription factors Gata4, Mef2c, and Tbx5 (GMT). By using this approach, the authors detected the rapid formation of cardiac troponin T positive cells (cTnT+) and the formation of contractile cells at a later time point. Once injected into the heart, GMT-transduced fibroblasts underwent in vivo reprogramming leading to the generation of new cardiomyocytes within 2 weeks [15]. Qian et al. demonstrated that nonmyocyte cells resident in the murine heart can be successfully reprogrammed in vivo into cardiomyocyte-like cells by the local delivery of GMT after coronary ligation [16]. Song et al. improved the effect of GMT transduction by adding Hand2, a transcription factor crucial for reprogramming of adult mouse cardiac fibroblasts into beating cardiac-like myocytes in vitro, through the so-called GHMT cocktail [17]. Nkx2.5, Srf, Smartcd3, Mesp1, and Myocd have also been described as other factors that can further increase fibroblast reprogramming when used in combination with GMT factors [18, 19]. In addition to the expression of coding genes, microRNAs have also been described as a powerful tool for non-myocyte cell reprogramming. Indeed, Jayawardena and coauthors found a combination of microRNAs suitable for cardiac fibroblast reprogramming both in vitro and in vivo. This combination included miR-1, miR-133, miR-208, and miR499. This set of microRNAs induced the expression of cardiac markers and also efficiently converted fibroblasts into functional cells with cardiomyocyte-like features such as the expression of ion channels, the presence of spontaneous calcium oscillations, and in vivo improved contractility and cardiac performance after injury [20, 21]. Of note, in 2013, Nam et al. combined microRNAs with transcription factors to further enhance cell reprogramming [22]. Recently, small molecules have also been tested in reprogramming protocols to obtain iCMs or cardiovascular precursors from CMSCs. In 2012, Vecellio et al. described a novel active epigenetic cocktail (EpiC) composed of all-trans retinoic acid (ATRA); phenyl butyrate (PB), a known HDAC inhibitor; and a nitric oxide donor (diethylenetriamine/nitric oxide – DETA/NO) as a HDAC modulator [23–26], which when combined with low serum (5% FBS) could reprogram human CMSCs into functionally competent cardiovascular precursors [27]. HDAC inhibitors such as VPA have been further used by Fu et al. in combination with three compounds, CHIR9921 (GSK3 inhibitor), RepSox (TGFβR1 inhibitor), and forskolin (adenylyl cyclase activator), to generate iCMs from fibroblasts without any transcription factor [28]. Notably, CHIR9921 is one of the three inhibitors also responsible for the “ground state” in mouse embryonic stem cells [29], and it has been recently used in combination with transcription factors and other small molecules to obtain iCMs [30]. The possibility to design an optimal culture medium to generate in vitro iCMs by a combination of small molecules [31] represents a tool of unlimited potential and a big challenge for the future. In light of this finding, Cao et al. have recently reported the reprogramming of somatic fibroblasts into cardiomyocyte-like cells by a combination of nine chemicals. The chemical cocktail opens chromatin of the cardiac developmental genes, permitting their transcription and thus the acquisition of a phenotype similar to that of embryonal cardiomyocytes [32]. A number of preclinical studies in large animal models of MI indicated significant positive effects of mesenchymal stem cell on cardiac function and on the reduction of scar size [33–37]. In these studies, MSCs were administered by intravenous or intracoronary injection or through direct injection into the cardiac tissue. It is noteworthy that in some of these studies, MSCs were injected in combination with other precursor cells [38]. In fact, the stochastic lifetime accumulation of alterations consequential to DNA and mitochondrial damage, metabolic changes, oxidative stress, and inflammation, coupled with other environmental risk factors, may directly influence the homeostasis of mesenchymal niches. In 2007, APOLLO (ClinicalTrials.gov Identifier: NCT00442806_Phase 1) established the safety and feasibility of adipose-derived MSCs infused intra-coronary in patients with MI and characterized by ST elevation. The efficacy of the treatment was demonstrated by a significant improvement of the cardiac perfusion and a 50% reduction in scar formation after a 6-month follow-up [39]. Similar results were obtained by the ADVANCE (NCT01216995_Phase 2) study. However, the MyStromalCell trial (NCT01449032_Phase 2) found an increase in exercise duration, a reduction of angina, and a general improvement of the quality of life after a 6-month follow-up after injection of autologous MSCs in patients affected by ischemic cardiomyopathy. Other such trials reported positively about the overall safety and tolerance of the treatment after 13-month follow-up and a decreased number of arrhythmic events or reduced chest pain, paralleled by an improved ventricular function, in patients with acute myocardial infarction treated with bone marrow mesenchymal stem cell [40]. In the REGENT Trial (Myocardial Regeneration by Intracoronary Infusion of Selected Population of Stem Cells in Acute Myocardial Infarction), the authors compared intracoronary infusion of bone marrow (BM)-derived unselected mononuclear cells (UNSEL) and selected CD34+CXCR4+ cells (SEL) in patients with acute myocardial infarction (AMI) and reduced < 40% left ventricular ejection fraction (LVEF). Two hundred patients were randomized to intracoronary infusion of UNSEL (n = 80) or SEL (n = 80) BM cells or to the control (CTRL) group without BM cell treatment. Primary endpoint: change of LVEF and volumes measured by magnetic resonance imaging before and 6 months after the procedure. After 6 months, LVEF increased by 3% (p = 0.01) in patients treated with UNSEL, 3% in patients receiving SEL (p = 0.04) and remained unchanged in CTRL group (p = 0.73). There were no significant differences in absolute changes of LVEF between the groups. Absolute changes of left ventricular end-systolic volume and left ventricular end-diastolic volume were not significantly different in all groups. A significant increase of LVEF was observed only in patients treated with BM cells who had baseline LVEF < median (37%). Baseline LVEF < median and time from the onset of symptoms to primary percutaneous coronary intervention > or = median were predictors of LVEF improvement in patients receiving BM cells. There were no differences in major cardiovascular events (death, re-infarction, stroke, target vessel revascularization) between groups [41]. In another randomized trial, Grajek et al. assessed change in left ventricle ejection fraction (LVEF) and myocardial perfusion in patients with acute myocardial infarction (AMI) of the anterior wall treated with bone marrow stem cells (BMSCs), compared with the control group – from baseline in the acute phase up to 12 months of follow-up. Forty-five patients were randomized 2 : 1 to the BMSC group (n = 31) or to the control group (n = 14). Bone marrow stem cells were administered into the infarct-related artery (IRA) at 4–6 days after primary PCI. Groups were followed up with Tc-99m-MIBI SPECT, radionuclide ventriculography (EF-RNV), echocardiography (ECHO), and the spiroergometric stress test. Coronary angiography was repeated after 6 months. EF-RNV did not differ significantly in both groups, but a trend towards increase in EF at 6 months and its maintenance after 12 months was noted in the BMSC group. In the rest study, perfusion index (PI) of the region supplied with blood by IRA distal to its previous occlusion (PI-IRA) improved significantly in the BMSC group at 6 months: PI-IRA at 4–6 days vs. PI-IRA at 6 months (3.00 ±0.97 vs. 2.65 ±0.64; p = 0.017). At 12 months, PI-IRA at rest was 2.66 ±0.55; p = 0.07. A difference between BMSC and control groups in the rest study in PI-IRA was not observed. In the dipyridamole study (PI-dip), perfusion in the BMSC group was better compared with controls at 6 months (2.26 ±0.44 vs. 2.47 ±0.40; p = 0.033) and at 12 months (2.34 ±0.55 vs. 2.52 ±0.42; p = 0.014), including for the region supplied with blood by IRA (PI-IRA-dip; at 6 months 2.63 ±0.77 vs. 3.06 ±0.46; p = 0.021 and at 12 months 2.71 ±0.63 vs. 3.15 ±0.51; p = 0.001). Results of LVEF, LVEDV, LVESV in ECHO and results of the spiroergometric stress test did not differ significantly between groups. Major adverse cardiac events occurred more often in the control group (p = 0.027). In conclusion, BMSC intracoronary transplantation in patients with anterior AMI did not result in increase in EF. Slight improvement of myocardial perfusion was noted in the BMSC group. This finding may indicate better microcirculation enhanced by BMSCs, but the small number of patients allows for a hypothesis rather than a final statement [42].
Stem cell and tissue engineering: an advanced strategy to treat cardiovascular pathology in cardiac surgery
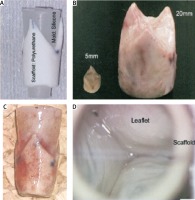
At present, although the previously established treatments for cardiac and vascular disorders, such as transplantation, surgical reconstruction, use of mechanical and synthetic devices, or administration of metabolic products, are effective, they still have several constraints and complications. Hence, the development of in vitro and in vivo biomimetic constructs for specific target organs or tissues are more suitable for regeneration of damaged vessels (Figure 2). Throughout recent years many advances have been made toward using adult stem cells clinically, and this includes the development of tissue engineered vascular grafts [43] and tissue-engineered heart valves [44]. By incorporating various progenitor cells such as bone marrow-derived mononuclear cells, mesenchymal stem cells, or endothelial precursor cells into biodegradable materials, vascular grafts and heart valves can be created that address limitations currently seen with other treatment approaches. Some of these cells have also shown the ability to initiate regenerative processes within the graft to develop tissue mimicking native arteries and valve. Tissue engineering strategies have three basic components: firstly, the cells or source which must express the appropriate genes and maintain the appropriate phenotype in order to preserve the specific function of the tissue [45]; secondly, the bioreactive agents or signals that induce cells to function; and thirdly, the scaffolds that house the cells and act as a substitute for the damaged tissue [46]. The source may be either embryonic stem cells (ESC) or adult stem cells (ASC), the scaffolds may be categorized as synthetic, biological, or composite, and the signals may include growth factors/cytokines, adhesion factors, and bioreactors [47]. Currently, downstream and upstream approaches in tissue engineering have been continuously investigated by many groups as the most promising tissue engineering approaches. The downstream approach usually employs implantation of precultured cells and synthetic scaffold complexes into the defect area. The cells or source, generally isolated from host target tissues, are expanded in vitro and pre-seeded into the scaffold to provide a porous three-dimensional structure that accommodates the seeded cells and forms the extracellular matrix (ECM) [48, 49]. Subsequently, multiple methods such as cell aggregation, microfabrication, cell sheeting, and cell printing are used to generate modular tissues. These are then assembled through random assembly, stacking of cell sheets, or directed assembly into engineered tissues with specific micro-architectural features. Thereafter, the engineered tissue is transplanted into the defective area. This approach allows scientists to finely transform the nanostructure of materials by balancing polymer degradation rates with ECM production and cellular infiltration which results in increased cell binding sequences, enzymatic cleavage sites, and tethering of chemoattractant molecules [49, 50]. Conversely, in the upstream approach, there are two ways to manufacture the engineered tissue: (1) cells and biomaterial scaffolds are combined and cultured until the cells fill the support structure to create an engineered tissue [51]; or (2) acellular scaffolds with incorporated biomolecules are delivered immediately after injury. The biomolecules are released from the scaffolds in a controlled manner, and they may recruit progenitor cells in the injured area and promote their proliferation and differentiation, eventually repairing the injured tissues [52, 53]. Tissue engineering is a promising approach that may lead to novel constructs that will satisfy this unmet need and overcome the limitations of current valve prosthetics. The tissue-engineered heart valve (TEHV) will be constructed using a combination of a porous scaffold, a cell population, and signaling factors and has the potential to provide (1) excellent hemodynamics, eliminating the need for anticoagulation therapy; (2) active tissue remodeling, preventing degeneration; and (3) growth characteristics, preventing the need for reoperation. The two primary types of valve scaffolds for the TEHV are natural scaffolds, such as decellularized tissue or biological materials, and synthetic constructs fabricated from degradable polymers [54]. Each type has inherent benefits and challenges, but decellularized heart valves are of significant interest. Decellularized heart valves are composed of biological materials that can positively impact cell differentiation and serve as building blocks during the remodeling process [55, 56]. Additionally, decellularized valves do not necessitate complete biodegradation and often maintain the mechanical anisotropy of the native valves from which they are derived [56–59]. Decellularized heart valves have been more clinically relevant than polymeric valves thus far, as they have been implanted as stand-alone valve substitutes and as TEHVs in animals and humans, albeit with mixed results [60–68]. However, decellularized heart valves are not without their limitations. For manufacture, decellularized valves require human or animal tissue, which is limited in supply, and necessitates cryopreservation for storage. Freeze-drying of biologic heart valves has been explored to facilitate long-term storage; however, freeze-drying leads to collapse of the extracellular matrix (ECM) structure and disruption of biomolecules. Research with lycoprotectants may overcome this limitation in the future [69]. Additionally, the success of decellularized heart valves is highly reliant upon the decellularization process and the potential immune response following implantation. On the other hand, man-made scaffolds, fabricated from synthetic or biological materials, do not require donor tissue but have struggled to recreate the macro- and micro valve anatomy and mechanical anisotropy of the native valve. Fabricated scaffolds must also undergo complete biodegradation in synchrony with the production of ECM to remain functional. Fabricated or synthetic scaffolds have been used as a TEHV in animals, but have seen far less use clinically than decellularized valves. Therefore, decellularized valve scaffolds have the greatest potential for expeditious development of a TEHV due to the regulatory history, long clinical experience with homografts, as well as a deep research focus by many groups. To date, synthetic scaffolds have not been used for clinical application and still are in the research and development stage. Currently, the mechanical and biological valves are used for clinical purposes. However, the mechanical prostheses have the adverse aspect of lifelong anticoagulation medication, and biological ones suffers from progressive degeneration. Biocompatible and biodegradable materials have been widely used in surgical application with neither immunogenic nor adverse side effects of degradation. Application of biocompatible scaffolds will result in a completely autologous living structure with the potential to grow and remodel. Therefore, the risk of an immunogenic response and infection is low due to the use of biodegradable materials. Shinoka et al. [70] in May 1999 developed synthetic heart valve scaffolds from a copolymer of PLA and PGA, seeded with either bone marrow cells or cells expanded from the saphenous vein harvest and conduit in 40 children with different forms of complex congenial heart valve disease. Serial post-operation investigation such as magnetic resonance imaging (MRI) and angiography represent no dilatation or even rupture while the histological evaluation image revealed calcification in these patients [70]. The application of bone marrow stem cells has been proposed recently and is still ongoing for further investigation. The harvesting is less invasive, and they grow faster than interstitial cells. However, the mechanism of differentiation is still unknown and leaves space for further survey. Neuenschwander and Hoerstrup [71] initially reported the application and implantation of a PGA scaffold seeded by autologous cells from sheep in the animal but the high stiffness of PGA and lack of mechanical properties made it unsuccessful. Later, they accepted polyhydroxyoctanoate (PHO) seeded with the autologous cells of ovine arterial. The result after 20 weeks of the implantation confirms no stenosis or thromboembolism. The experiments on natural porcine heart valve leaflets suggested that the elastic modulus and tensile strength of the valve cups are higher in the circumferential direction (collagen architecture organized direction) compared to the radial direction. Thus, the uni-axial tensile tests do not fully resemble the mechanical properties of the valve leaflets. The biaxial test is usually used to measure the circumferential and radial direction simultaneously. Stella et al. [72] investigated the biaxial time-dependent tensile behavior of a porcine heart valve including stress relaxation and strain rate under 0–60 N/m. They found that the stretch and tensile relation was insensitive to the strain rate. Their study indicates that there is no relation between the creep mechanism and the relaxation in the heart valves.