Primary thymic carcinomas are very rare tumors. Their incidence is estimated around 1 to 3 cases per 10 million inhabitants. In this group, lymphoepithelioma-like carcinoma occurs (LELC) in 5–32% and the prognosis of advanced cases is poor.
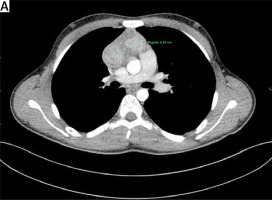
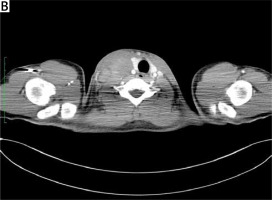
A 17-year-old man was admitted to the hospital because of an enlarged right cervical lymph node. A computed tomography (CT) scan of the chest revealed an 8 cm tumor in the anterior mediastinum, with suspected left brachiocephalic vein infiltration (Figure 1). Besides the tumor in the mediastinum and metastasis in the right cervical lymph node (Figure 2), no other lesions with increased 18F-FDG uptake on positron emission tomography (PET)-CT were demonstrated. All other tests (laryngological examination, abdominal CT, blood tests including α-FP and β-HCG, bone marrow biopsy) were negative for cancer involvement. The right cervical lymph node was excised and the patient was diagnosed with thymic LELC.
In correlation with radiological findings, the patient was diagnosed with primary thymic cancer at stage IV according to the 8th edition of the WHO classification (T3N2M0) and IVB according to the Masaoka classification. Because of local progression and lymph node involvement, the patient was recommended for multimodal treatment. As first-line treatment, three cycles of neoadjuvant chemotherapy comprising an etoposide, doxorubicin, cisplatin regimen were applied. Subsequent chest CT revealed partial remission of the target lesion and substantial decrease of 18F-FDG uptake in PET-CT. Because of a good response to the chemotherapy, the patient was referred for surgery. The surgery was performed by longitudinal sternotomy. A tumor sized 6 × 8 cm was found, infiltrating the parietal pleura of the apex of the right lung, surrounding the superior caval vein and venous angle. The tumor was dissected from the pericardium and vena cava, then resected en-bloc with part of the infiltrated mediastinal pleura. The mediastinal lymph nodes were removed. The final pathological diagnosis confirmed LELC, R0, ypT1N0. The postoperative period was uncomplicated and the patient was discharged on postoperative day 10. Subsequently, the patient was treated with two cycles of adjuvant chemotherapy using the etoposide, doxorubicin, cisplatin regimen. After the chemotherapy, a follow-up CT scan revealed enlarged right supraclavicular and bilateral submandibular lymph nodes. The total bilateral cervical lymphadenectomy, with submandibular lymphadenectomy and submandibular gland resection, was performed. Pathological examination did not reveal any metastases. In order to improve local control and obtain better long-term results, the patient was treated with 54 Gy external beam radiotherapy for the mediastinum and neck.
Since the treatment completion, the patient has been under routine follow-up. There have been no symptoms of the recurrence of the disease for 10 years.
Primary thymic carcinomas occur in less than 5% of anterior mediastinal tumors. In this group, the incidence of LELC is 5–32% [1]. Previously they were classified as type C thymomas and treated as such. In 2004, they were reclassified as a separate group according to the WHO classification secondary to the very different clinical prognosis and histological features, compared to thymomas [2]. Besides clinical stage, in the group of thymic carcinomas, histologic differentiation of tumor is an important prognostic factor of the clinical course.
In the Ogawa et al. study, high-grade tumors, including LELC and high stage of the disease, according to the Masaoka classification, were independent, significant negative prognostic factors [3]. Similar observations were made by Suster and Rosai in a group of 60 patients with thymic carcinomas; they found 85% mortality of patients with high-grade histology [4].
In general, LELC is characterized by aggressive clinical behavior. It grows rapidly, frequently invades adjacent structures and metastasizes. LELC, similarly to the other primary thymic carcinomas, usually presents with symptoms arising from local progression of disease or distant metastases, whereas paraneoplastic syndromes occur occasionally [5]. The diagnostic process is mostly aimed at obtaining a definite histologic diagnosis, which allows one to differentiate it from other more frequently occurring tumors of the anterior mediastinum and establish the clinical stage of the disease. Among imaging tests, the PET-CT with measurement of 18F-FDG glucose uptake is useful; in addition to CT, it allows one to assess better stage of the disease.
An interesting feature of LELC is relatively frequent occurrence of Epstein-Barr virus (EBV) in cancer cells, which can suggest the role of this virus in the etiopathogenesis of cancer [6]. In this case, the immunohistochemical staining did not reveal antigens typical for EBV (LMP-1), whereas DNA EBV was found in blood.
The primary treatment of LELC is multimodal treatment, which can include surgery, radiotherapy and chemotherapy; however, because of the small volume of cases and their clinical variability, there are no unified treatment guidelines.
Most authors agree that in cases in the early stage, limited to the mediastinum, which are potentially resectable, the surgical treatment is the mainstay of multimodal treatment. In Ogawa et al.’s retrospective analysis of forty patients treated for thymic cancer, R0 resection was the most important prognostic factor of survival [3]. In the case of locally advanced tumors, it is worth considering even an extensive procedure including a partial resection of the vena cava superior or brachiocephalic vein. Besides an anterior mediastinal lymphadenectomy, the cervical lymphadenectomy should be considered if there is a suspicion of metastases. In most cases of LELC treatment, chemotherapy is used; however, timing (before, after surgery, or both) and a chemotherapeutic agent remain a matter of debate. According to NCCN guidelines, as the first-line chemotherapy regimen, carboplatin and paclitaxel are recommended [1]. In the present case etoposide, doxorubicin and cisplatin were used with evident local remission. Since thymic cancers are highly invasive tumors and local recurrences are common, postoperative radiotherapy should be considered in all cases after surgery, including R0 resections.
In the reported case, multimodal treatment including neoadjuvant chemotherapy, R0 resection and adjuvant chemo- and radiotherapy allowed the patient’s complete remission. This outcome corresponds with the results of Lucchi et al. In the analyzed group of patients, neoadjuvant chemotherapy with cisplatin, epidoxorubicin and etoposide followed by surgery and adjuvant radiotherapy enabled 5-year survival to be achieved in 5 of 7 patients. The authors concluded that neoadjuvant chemotherapy may improve the resectability rate [7]. Aggressive multimodal treatment including surgery, chemotherapy and radiotherapy, in selected cases can allow one to obtain a good long-term result of a regionally advanced primary thymic LELC.