Introduction
In metastatic colorectal cancer (mCRC) with unresectable metastases, the combination of 5-fluorouracil/leucovorin (5-FU/LV) with oxaliplatin (FOLFOX regimen) or irinotecan (FOLFIRI regimen) is part of the first-line standard treatment that improved patient survival when compared to 5-FU/LV administered alone [1–4]. The use of monoclonal antibodies against VEGF and EGFR has also proved beneficial in this situation [5].
These treatments are intended to be administered at a certain dose and regimen. However, due to the onset of toxicities, it is common in clinical practice to make dose reductions or delay the administration of these molecules, which has the consequence of decreasing the relative dose intensity (RDI) [6, 7]. In many cancers, low chemotherapy RDI is associated with decreased disease control and survival [8–11]. With regard to mCRC, certain retrospective analyses of patients treated with FOLFOX or FOLFIRI have highlighted a link between survival and RDI of oxaliplatin and irinotecan [12, 13]. For 5-FU, one study has shown a possible link between RDI and response rate [14]; however, these results were not confirmed in a second trial when 5-FU was combined with LV [15]. In these studies, data resulted from clinical trials. As far as is known, in real-life, no study has shown a negative impact on survival of a low RDI of oxaliplatin, irinotecan or 5-FU in patients treated with FOLFOX or FOLFIRI [16, 17].
The objective of this analysis is to determine, in real-life, the impact on progression-free survival (PFS) and overall survival (OS) of RDI of 5-FU, oxaliplatin and irinotecan in patients treated for a mCRC, as part of FOLFOX and FOLFIRI regimens.
Material and methods
Patients
Data were collected from patients treated between September 2008 and January 2019 with FOLFOX or FOLFIRI as a first-line treatment in our center for a histologically proven mCRC with unresectable metastases. Some patients were treated with bevacizumab, cetuximab, panitumumab or a colorectal resection. Patients treated with capecitabine or who received less than 2 cycles of chemotherapy were not included. Co-morbidities were assessed by the Charlson index [18]. This work was done in accordance with the Declaration of Helsinki and the General European Data Protection regulation (number 2016/679). The methodology was validated by the Data Protection Officer of our center.
Treatment and dose intensity
Oxaliplatin was administered at a planned dose of 85 mg/m2, irinotecan at 180 mg/m2 and leucovorin at 400 mg/m2. 5-FU contained a bolus of 400 mg/m2 followed by an infusion of 2400 mg/m2 over 46 hours. Bevacizumab was administered at 5 mg/kg, cetuximab at 400 mg/m2 in the first cycle, 500 mg/m2 in following cycles and panitumumab at 6 mg/kg every 2 weeks.
The planned duration of treatment (weeks) corresponded to the number of cycles multiplied by the duration of a cycle, i.e. two weeks. Actual duration of treatment (weeks) was calculated as follows: [(last cycle date) – (first cycle date) + 14] / 7. For a given molecule, planned dose intensity (DI) was obtained by dividing the planned cumulative dose (CD, mg) by the planned duration of treatment (weeks). Actual DI was calculated by dividing the actual CD by the actual duration of treatment. RDI, expressed as a percentage, corresponded to the ratio between the actual and the planned DI.
Study groups
For each patient, the RDI of 5-FU was calculated and, depending on the case, the RDI of oxaliplatin or irinotecan. Because of the small number of patients receiving oxaliplatin in our cohort, RDIs of oxaliplatin and irinotecan were pooled and studied together (noted OXA-IRI) to increase the statistical power of the analysis. The median RDI of 5-FU was 78.3% (interquartile range [IQR]: 67.4–86.2). For OXA-IRI, the median RDI was also 78.3% (IQR: 68.6–86.1). To determine the impact of the RDI of 5-FU and OXA-IRI on survival, the patients were separated according to the median RDI: patients with a high RDI (> 78.3%) and patients with a low RDI (≤ 78.3%).
Outcomes
PFS corresponded to the time (months) from the start of treatment to progression or death, regardless of cause. Overall survival was defined as the time (months) from the date of treatment initiation to the date of death from any cause.
Statistical analysis
Categorical variables were described using number and percentage, while continuous variables were expressed as median and interquartile range. Patient characteristics were compared using the χ2 test or Fisher’s exact test for categorical variables, and the Kruskal-Wallis test for continuous variables. Kendall’s tau correlation coefficient was used to assess the correlation between the RDI of 5-FU and the RDI of OXA-IRI. OS and PFS were estimated by the Kaplan-Meier method and presented with a 95% confidence interval for the overall population and by analysis group. Events that did not occur were right-censored. A univariate and multivariate analysis of PFS and OS was performed using the Cox proportional hazards model. The significance level used was 0.05.
Results
Patients
75 patients meeting the inclusion criteria were identified over the study period. The characteristics of the patients at the start of treatment are presented in Table 1. Median age was 67.1 years (IQR: 61.7–75.3) and the majority of patients (77%) had undergone colorectal resection surgery before treatment with chemotherapy. There were no statistically significant differences between the groups studied.
Table 1
Patient characteristics in the overall population and according to the relative dose intensity (RDI) of 5-fluorouracil (5-FU) and oxaliplatin or irinotecan (OXA-IRI)
Treatment and relative dose intensity
The characteristics of the treatment are presented in Table 2. Kendall’s tau correlation was 0.78 (p < 0.01), indicating a significant and strong correlation between RDIs of 5-FU and RDIs of OXA-IRI. In other words, the majority of patients who had a low RDI for 5-FU also had a low RDI for OXA-IRI. Patients with a high RDI had a significantly shorter actual duration of treatment compared to patients with a low RDI, for both 5-FU and OXA-IRI.
Table 2
Treatment and dose intensity in the overall population and according to the relative dose intensity (RDI) of 5-fluorouracil (5-FU) and oxaliplatin or irinotecan (OXA-IRI)
Survival
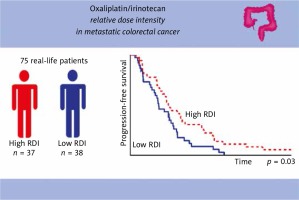
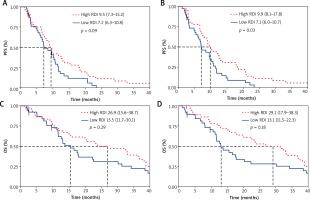
The median follow-up was 17.5 months. In the overall population, the median PFS was 8.9 months (95% confidence interval [CI], 7.07–10.65) and the median OS was 17.9 months (95% CI, 13.1–31.3). We found a statistically significant difference in PFS in favor of patients with a high RDI of OXA-IRI (hazard ratio [HR], 0.58; 95% CI, 0.36–0.95; p = 0.03) (Fig. 1). For 5-FU, there was a trend in favor of patients with a high RDI (HR, 0.66; 95% CI, 0.41–1.07; p = 0.09). There was no statistically significant difference in OS for either patients with a high RDI of 5-FU (HR, 0.77; 95% CI, 0.48–1.25; p = 0.29) or patients with a high RDI of OXA-IRI (HR, 0.72; 95% CI, 0.45–1.17; p = 0.18).
Univariate and multivariate analysis of progression-free survival and overall survival
Univariate and multivariate analysis of progression-free survival and overall survival are presented in Table 3 and Table 4. The presence of the BRAF V600E mutation was associated with an increased risk of progression or death, both in the univariate and the multivariate analysis, while colorectal surgery decreased the risk of progression or death. There was a higher risk of death in patients with poorly differentiated tumors, both in the univariate and the multivariate analysis. Body mass index, chemotherapy regimen and biologic therapy were predictive of overall survival but only in the univariate analysis. Initial performance status and Charlson comorbidity index had no impact on progression-free survival or overall survival.
Table 3
Univariate and multivariate analysis of progression-free survival
Table 4
Univariate and multivariate analysis of overall survival
Toxicity
The toxicity profile of the patients is presented in Table 5. Nausea and vomiting were the most frequent toxicity effects (37% of the patients) followed by diarrhea (32%) and neuropathy (19%). The majority of toxicities (91%) were grade I or II, grades III or IV accounting only for 8% of toxicities. No statistically significant difference was observed according to the RDI.
Table 5
Toxicity in the overall population and according to the relative dose intensity (RDI) of 5-fluorouracil (5-FU) and oxaliplatin or irinotecan (OXA-IRI)
Discussion
For oxaliplatin and irinotecan, our study on real-life patients showed a significant link between RDI and PFS. These results are consistent with the work of Nakayama et al. and Maindrault-Goebel et al., whose analysis focused on patients from clinical trials [12, 13]. Several models have been proposed to explain the relationship between dose intensity/density of chemotherapy and tumor growth [19]. In Norton’s model, the higher the density, the more limited is the tumor re-growth between cycles [20]. However, our analysis did not find an impact on OS of low RDI of 5-FU or OXA-IRI.
In our cohort, the median RDI was 78.3% for both 5-FU and OXA-IRI. In clinical trials with FOLFOX and FOLFIRI, the RDIs are often higher, for example up to 92% for 5-FU, 87.8% for oxaliplatin and 93% for irinotecan [1, 3, 4]. This difference is likely because patients in clinical trials are typically younger, healthier, and better able to withstand higher does intensities than patients from real-life.
In addition, there is a significantly longer treatment duration for patients with low RDI, associated with a higher number of cycles. This could be explained by a better tolerance to treatment in patients with a low RDI, i.e. patients whose dose has been reduced or cycles delayed, allowing more cycles to be administered.
Our analysis identified the BRAF V600E mutation and colorectal surgery as independent factors for progression-free survival and overall survival. A recent meta-analysis of nearly 160,000 patients showed a significant effect on survival of primary tumor resection in patients with localized incurable or mCRC [21].
There are several limitations of our study. First of all, due to the limited number of patients in our center, we decided to study the impact of RDI of oxaliplatin and irinotecan on survival in an undifferentiated way, in an OXA-IRI group. The effect of RDI on survival for each molecule could therefore not be determined. In the analysis of Nakayama et al., only high RDI of irinotecan had a significant impact on PFS [12]. The improvement in PFS in the OXA-IRI group in our study could be explained by a higher proportion of patients who received irinotecan (71%) instead of oxaliplatin. A second limitation is represented by a probable confounding factor which consists of reducing the doses of chemotherapy or in spacing out the cycles in patients who become clinically impaired. The poorer survival could be explained by clinical deterioration and not by the reduction in RDI. Finally, hematological toxicity is likely underestimated in our cohort due to missing data.
Conclusions
Our analysis suggests that a low RDI of oxaliplatin or irinotecan has a negative impact on PFS in patients treated in real-life for a mCRC with FOLFOX or FOLFIRI. However, RDI had no significant effect on OS in our cohort. Based on this last observation, it seems unreasonable to try to maintain a high RDI, with a greater risk of toxicity, in palliative patients whose quality of life should be maintained as long as possible.