Introduction
Pulmonary tumour embolism (PTE) is a rare condition characterised by obstruction made up of tumour cells and thrombi in the pulmonary arterioles and capillaries, diagnosis of which antemortem is notoriously difficult. Clinically it is manifested by nonspecific symptoms such as dyspnoea, cough and pulmonary hypertension [1]. The majority of routine diagnostic procedures fail to accurately identify PTE. Moreover, PTE can occur as the first manifestation of cancer and, as such, mistaken for a cardiopulmonary pathology, it may cause a delay in diagnosis of the underlying malignancy.
Here we report a case of a 46-year-old Caucasian male with multiple pulmonary tumour embolism derived from hitherto undiagnosed malignant disease. The presented case demonstrates the ambiguities surrounding the diagnosis of PTE.
Case report
In 2018, a 46-year-old male was admitted to the Department of Internal Medicine with a one-month history of progressing dyspnoea at rest. The patient reported cough with mucus and severe exercise intolerance. In the last year he had weight loss of 15 kilograms. The patient had no past history of either pulmonary or heart problems. He was a heavy smoker for 17 years and a stonemason by profession. Because of an occupational hazard, pneumoconiosis was initially suspected.
On examination the patient was tachycardiac with normal blood pressure (120/78 mm Hg), tachypnoeic and hypoxic (SpO2 93.7% in room air). Mild exercise induced oxygen desaturation. Laboratory tests indicated acute inflammation. Spirometry demonstrated mild lung restriction. Bronchoscopy did not reveal any relevant anomalies. Microbiological analysis of the aspirate provided negative results. No significant abnormalities were detected by abdominal ultrasound, electrocardiography or echocardiography. Pulmonary angiography disclosed no evidence of emboli. X-ray imaging showed several pathologic features such as fluid in pleural space, loss of pulmonary lobes volume and reticular opacification (Fig. 1). High-resolution computed tomography (HRCT) (Fig. 2) displayed left lung base atelectasis, interlobular septal thickening and pleural effusion (left space – 65 ml, right space – 20 ml). The HRCT also detected an elevation of the left hemidiaphragm and mediastinal nodes enlargement. Subsequently, thoracentesis was performed to aspirate exudative fluid from the pleural cavity (2650 ml). The patient received antibiotics and oxygen therapy, but no improvement was observed. Based on the clinical presentation as well as on the CT scan, the differential diagnosis included sarcoidosis, silicosis and lymphangitic carcinomatosis. In search for a specific causal factor the patient was further subjected to video-assisted thoracoscopic surgery (VATS), during which pleurodesis was performed and biopsies of both parietal pleura and lung parenchyma were taken.
Fig. 1
X-ray imaging showing fluid in pleural space, loss of pulmonary lobes volume and reticular opacification
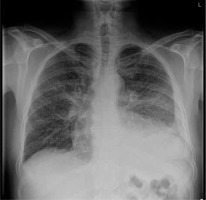
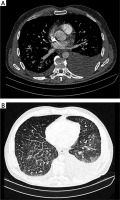
Fig. 2
High-resolution computed tomography (HRCT): fluid in pleural cavity (A), interlobular septal thickening, mediastinal nodes enlargement (B)
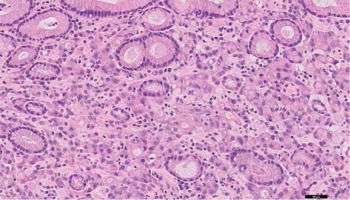
Results of histopathological examination excluded all initially suggested underlying conditions. On high magnification, multiple emboli of atypical epithelial cells were found in pulmonary blood vessels (Fig. 3A). The cells had a characteristic signet – ring shape with a central, optically clear droplet of cytoplasmic mucin and eccentrically displaced nucleus (Figs. 3B and 3C). Immunohistochemistry (IHC) revealed i) positivity for cytokeratin 7, cytokeratin 20, cytokeratin 19, Ki67, high molecular weight cytokeratin and cytokeratin AE1/AE3 (Fig. 4) and ii) negativity for CD68, thyroid transcription factor 1 (TTF1), cytokeratin 5/6, p63, synaptophysin, CD10, napsin A, PAX-8, prostate specific membrane antigen (PSMA), adult renal cell carcinoma (RCC), CD56 and chromogranin A. Results for CDX2 were heterogenous (Table 1). In addition, the biopsy was found positive for mucin.
Fig. 3
The haematoxylin and eosin staining shows multiple emboli of atypical epithelial cells (A, B). The cells have a characteristic signet – ring shape with a central, optically clear droplet of cytoplasmic mucin and eccentrically displaced nucleus (C). Magnification of 0.5× (A), 20× (B), 40× (C)
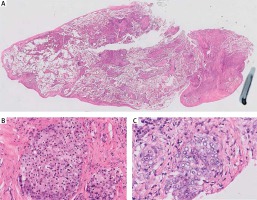
Fig. 4
Immunophenotyping of tumour cells: positivity for cytokeratin 7 (A), cytokeratin 20 (B), negativity for TTF1 (C). The biopsy was positive for mucin (D). Magnification of 10×
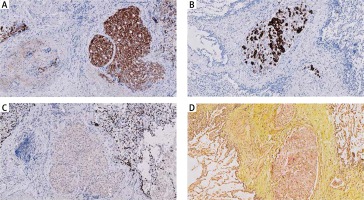
Table 1
List of primary antibodies used in the immunohistochemistry panel
Since the IHC data strongly suggested the gastrointestinal (GI) tract as the origin of cells, GI endoscopy was conducted. Inflammatory changes within the gastric mucosa were observed. Two ulcers covered with fibrin measuring approximately 6 mm and 10 mm in diameter were detected in the stomach. Biopsy was taken and histopathology confirmed the diagnosis of signet ring cell gastric adenocarcinoma (Fig. 5) with haematogenous dissemination involving pulmonary vessels. The patient was referred for palliative EOX (epirubicin, oxaliplatin, capecitabine) chemotherapy. After completing two treatment cycles he reached partial remission and stabilisation of the disease. One month later his condition deteriorated and because of exacerbation of dyspnoea, he required an oxygen concentrator. He received four cycles of docetaxel, but pleural effusion was constantly increasing. The patient underwent several thoracenteses. There was no improvement and he died within two months from the first chemotherapy.
Discussion
We described a case of PTE presented with dyspnoea that was associated with previously unrecognized signet ring cell gastric adenocarcinoma, diagnosis of which created considerable difficulties.
PTE is characterized by a presence of isolated cells or clusters of tumour cells within the pulmonary arterioles causing microthrombi and occlusion, which leads to the elevation of pulmonary vascular resistance and development of hypertension (PH) [2]. The condition was documented for the first time by Schmidt in 1897 [3]. In the 2004 World Health Organization (WHO) Classification, PTE belongs to the “group 4” of the Clinical Category of Pulmonary Hypertension [4]. The most recent WHO Classification’s update (2016) assigned PTE to the ‘Other malignant tumours” group (4.2.2) [5, 6]. PTE can evolve into concomitant pulmonary tumour thrombotic microangiopathy (PTTP), characterised by fibrocellular proliferation of arterial intima [7].
PTE is rarely reported in cancer patients, which is mainly due to the considerable difficulty of antemortem diagnosis. It is assumed that microscopic PTEs are found posthumously in approximately 3% to 26% of solid malignant neoplasms that do not have metastases to the lung parenchyma [2, 8]. Since the condition is exceptionally infrequent, the patient might be misdiagnosed and, as a consequence, treated for more common diseases. In fact, making a diagnosis of PTE, like in our case, while the patient was still alive, is uncommon. The most of PTE cases originate from adenocarcinomas of the breast, stomach, lung, colon and prostate [9]. There are few reports of PTE associated with choriocarcinoma [10, 11], pelvic chondrosarcoma [12] myxofibrosarcoma [13] and renal sarcoma [14]. Diagnosis of PTE is usually made coincidentally at postmortem examination [15–17]. It is assumed that PTE may be the cause of death in 3.6% of cancer patients [18].
Clinical manifestation of PTE is uncharacteristic. The condition can present in a wide range of nonspecific symptoms, sometimes very similar to those of other more common cardiopulmonary diseases. Progressive dyspnoea is the main clinical manifestation of PTE (present in 57% up to 100% cases) [2]. Shortness of breath results in arterial hypoxia and respiratory alkalosis. Cough and chest pain commonly accompany PTE (47% of cases) and 15-20% of PTE patients demonstrate classic signs of pulmonary hypertension [19] likely to be associated with right heart failure (pulmonary heart disease) [17, 20, 21]. Except for progressive dyspnoea, the above symptoms did not contribute to the clinical presentation of the reported case. Instead of pulmonary hypertension and right heart failure, the patient suffered from tachypnoea and tachycardia. A few symptoms characteristic of PTE such as cough, dyspnoea and mild lung restriction the patient complained of were misinterpreted as silicosis, a condition more common and, because of the patient’s occupational hazard (a stonemason) and cigarette smoking, deemed more likely to be true. Results of the initial examination could also suggest sarcoidosis or lymphangitic carcinomatosis, as the patient suffered from massive pleural effusion, a symptom reported in some silicosis [22], lymphangitic carcinomatosis [23], and sarcoidosis [24] but never in PTE cases.
PTE has typically a poor prognosis. In the majority of cases patients die shortly after admission due to cardiopulmonary arrest. Unfortunately, there are no treatment guidelines available – only a few treatment options have been recommended. Anticoagulants were widely proved not to increase the survival rate. As reported in some cases, early chemotherapy or surgical embolectomy of pulmonary embolism followed by chemotherapy might be successful and lead to prolonged survival [10, 12, 25–27].
Early recognition of PTE would be essential, but it is difficult before death since the range of diagnostic procedures described over the years is very limited. Surgical lung biopsy with histopathological evaluation is the gold standard and indeed, in the presented case, it played a key role in establishing the diagnosis. Microscopically, the pulmonary vessels are occluded by emboli consisting of atypical malignant cells. Typically, there is no evidence of adjacent primary or metastatic tumours. A wide panel of immunohistochemical staining is highly recommended for verification of possible cell origin. In our case, the cells of gastric adenocarcinoma were found within pulmonary vessels and the extrapulmonary origin of cells was confirmed by performing specific immunohistochemistry. Concerning hemodynamically unstable patient, transbronchial biopsy is considered to be an alternative diagnostic test, but it must be avoided in patients with severe PH due to the high risk of bleeding [26]. Radiographic examination usually shows a normal chest X-ray with associated hypoxia and is not diagnostic in PTE [27]. Parenchymal abnormalities (atelectasis, reticular pattern) in chest radiography, as seen in the presented case, are rare [17]. Moreover, they are characteristic of other more common conditions, and did not assist proper assessment and diagnosis. Pulmonary angiography typically fails to identify PTE [17, 21], although in a few cases it was reported to guide the diagnosis [10]. HRCT is often normal, therefore as a diagnostic tool it also remains of limited value [26]. There are some reports about the utility of a ventilation-perfusion scintigraphy, which usually reveals numerous, symmetrical, peripherally located, subsegmental perfusion defects (“mottled” or “beaded” appearance) with correct ventilation [26, 29, 30]. Pulmonary microvascular cytology (PMC) has been proposed as an additional test, however its sensitivity and specificity are doubtful [27, 28]. Therefore, in the majority of cases the actual diagnosis of PTE is made at autopsy.