Introduction
The prevalence of metabolic dysfunction-associated steatotic liver disease (MASLD), a hepatic manifestation of metabolic syndrome, is increasing worldwide [1]. One out of four people has MASLD, and 1% of MASLD patients has cirrhosis in Japan [2]. Approximately half of MASLD patients have dyslipidemia, and the prevalence of MASLD is proportionate to the level of triglycerides and body mass index [2]. MASLD is also associated with cardiovascular disease and chronic kidney disease [3, 4]. The fibrosis stage of MASLD is strongly associated with liver-related events and hepatocellular carcinoma [5]. The long-term control of hepatic fibrosis in patients with MASLD is important to improve the prognosis, but no specific treatment of MASLD has been approved, apart from resmetirom, which has been conditionally approved by the United States Food and Drug Administration.
Pemafibrate, a selective peroxisome proliferatoractivated receptor-α modulator (SPPARMα), has been available in Japan since 2018. In a large prospective study, pemafibrate therapy significantly reduced the incidence of MASLD [6]. In a phase II trial, 72-week pemafibrate therapy significantly reduced magnetic resonance elastography (MRE)-based liver stiffness [7]. Our group previously reported its therapeutic effectiveness during a period of three months and one year [8, 9]. However, data regarding long-term effectiveness for more than two years are scarce. The development of hepatic fibrosis requires several decades in MASLD; therefore, long-term data showing the effectiveness of a therapeutic agent would be valuable. Although one-year pemafibrate therapy clearly improves hepatic inflammation, long-term maintenance of that sustained improvement has not been confirmed. Furthermore, it is still unclear whether sustained reduction of hepatobiliary enzymes caused by pemafibrate could also result in improvement of hepatic fibrosis. The aim of this study was to assess three-year outcomes under pemafibrate therapy in patients with MASLD.
Material and methods
Study population
This was a dual-center retrospective observational study including patients with MASLD treated with pemafibrate for more than three years at the Shinozaki Medical Clinic and the Saiseikai Utsunomiya Hospital. Patients were started on pemafibrate therapy between June 2019 and April 2021. We retrospectively reviewed their medical records. The inclusion criteria were as follows: 1) steatotic liver diagnosed by abdominal ultrasound, 2) presence of dyslipidemia (patients under treatment for hypercholesteremia or hypertriglycemia with elevated values observed at least once within six months), 3) sustained elevation of alanine aminotransferase (ALT) > 30 U/l for more than three months before starting pemafibrate, 4) negative hepatitis B surface antigen and hepatitis C virus antibody test, 5) normal serum immunoglobulin-G level, 6) alcohol consumption < 30 g/day in males and < 20 g/day in females [8]. The exclusion criteria were as follows: 1) severe chronic kidney disease (serum creatinine > 2.5 mg/dl), 2) history of previous pemafibrate use, or 3) patients with diabetes mellitus. The adherence was evaluated by frequent outpatient clinic appointments. The treating physician instructed every patient regarding diet, exercise, and life-style modifications using unified colored pamphlets on a monthly basis. The Institutional Review Board approved this retrospective review (ID#31-R002).
Evaluation of hepatic inflammation and fibrosis
Hepatic inflammation was evaluated according to serum ALT levels. The American Association for the Study of Liver Diseases practice guidance stated that ALT normalization can predict metabolic dysfunction-associated steatohepatitis resolution and ALT > 30 U/l should be considered abnormal [10]. Hepatic fibrosis was estimated by the FIB-4 index and the Mac-2 binding protein glycosylation isomer (M2BPGi), and a chemiluminescence enzyme immunoassay kit (HISCL-M2BPGi; Sysmex, Kobe, Japan) was used [11].
Statistical analysis
Changes in parameters from baseline to three years were assessed using the Wilcoxon signed-rank sum test. Comparisons of multiple timing in parameters were evaluated using Friedman’s test. In addition, subgroup comparisons of ALT levels between patients treated with pemafibrate alone and those treated with pemafibrate plus statins were performed using the Mann-Whitney U test. To evaluate the association between parameters, Spearman’s rank correlation coefficient was used. All statistical analysis was performed using the BellCurve for Excel (Social Survey Research Information Co., Ltd., Tokyo, Japan). Differences were considered significant at p < 0.05.
Results
Baseline characteristics
Forty-six patients fulfilled initial inclusion criteria, but six patients with diabetes were then excluded. Consequently, 40 patients were included in the final analysis. All patients received pemafibrate 0.2 mg daily for three years without dose escalations. Regarding the treatment of dyslipidemia, a statin was administered in 65% of patients for at least six months (4.7 years on average), and pemafibrate was added to the statin (Table 1). There was no combination use of sodium-glucose transport protein-2 (SGLT2) inhibitor, pioglitazone or tocopherol. There were no adverse events during the study period. In addition, all patients had > 95% adherence to treatment. At the baseline M2BPGi levels, the number of patients with hepatic fibrosis stages 0, 1, 2, 3 and 4 was 17, 12, 2, 8 and 1, respectively, and therefore 29 of 40 patients (73%) were categorized in hepatic fibrosis stage 0 or 1.
Table 1
Baseline characteristics of patients
Outcome of patients at three years
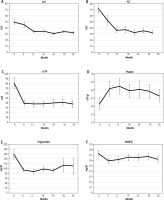
During the three-year pemafibrate therapy, patients’ weight remained stable (Table 2). With respect to hepatobiliary inflammation, there were significant reductions in aspartate aminotransferase (AST), ALT, and γ-glutamyl transpeptidase (γ-GTP) levels. ALT levels continuously decreased within year one and subsequently remained almost normal (Fig. 1). The platelet count showed a significant increase compared to the baseline at three years; however, although it increased within one year from the initiation of pemafibrate, the rate of increase diminished over the second and third years. Regarding lipid profiles, low-density lipoprotein (LDL) cholesterol and triglycerides decreased significantly at three years. The triglyceride levels decreased at six months and then plateaued. As for hepatic fibrosis, M2BPGi significantly improved at six months and then remained low, although slight fluctuations were noted. The change in FIB-4 index was not statistically significant.
Table 2
Changes in clinical parameters after a three-year course of pemafibrate therapy
Fig. 1
Changes in parameters for three-year pemafibrate therapy. A) Aspartate aminotransferase (AST) (p < 0.001, Friedman’s test), B) alanine aminotransferase (ALT) (p < 0.001), C) γ-glutamyl transpeptidase (γ-GTP) (p < 0.001), D) platelet count (p = 0.001), E) triglycerides (p < 0.001), and F) Mac-2 binding protein glycosylation isomer (M2BPGi) (p < 0.001)
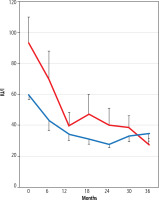
To evaluate the potential impact of statin use on treatment outcomes, we performed a subgroup analysis comparing changes in ALT levels between patients treated with pemafibrate alone and those treated with pemafibrate plus statins. As shown in Figure 2, baseline ALT levels were significantly lower in the statin group (p = 0.01), which may reflect prior long-term statin use. However, after three years of treatment, there was no significant difference in ALT levels between the two groups (p = 0.46). In both groups, ALT levels significantly improved over the three-year follow-up period (p < 0.01).
Fig. 2
Comparison of alanine aminotransferase (ALT) levels between patients treated with pemafibrate alone (red line) and those treated with pemafibrate plus statins (blue line). At baseline, ALT levels were significantly lower in the statin group compared to the non-statin group (p = 0.01, Mann-Whitney U test). However, after three years of treatment, no significant difference in ALT levels was observed between the two groups (p = 0.46). In both groups, ALT levels significantly improved over the three-year follow-up period (p < 0.01, Friedman’s test)
Correlation analysis with improvement of hepatic fibrosis
Correlation analysis between three-year changes in M2BPGi and other parameters were performed (Table 3). Changes in weight, AST, ALT and γ-GTP showed a significant positive correlation with changes in M2BPGi. Long-term control of hepatic inflammation markers was associated with the improvement of hepatic fibrosis markers.
Table 3
Association of changes in M2BPGi with other parameters
Discussion
Three-year treatment of pemafibrate appears to have favorable effects on MASLD, including sustained improvement in markers of hepatic inflammation and fibrosis as well as lipid profiles. Thus, pemafibrate could emerge as a treatment choice to prevent progression in patients with MASLD and dyslipidemia.
We previously reported the effectiveness of 12-month pemafibrate treatment on MASLD [8, 9, 12, 13]. In this study, we evaluated the beneficial effects of pemafibrate treatment for a three-year period. Any long-term treatment could encounter challenges that include adverse events, adherence to treatment, and the rebound phenomenon. In the present study, no adverse events were documented. As a result, the patients showed high adherence to the treatment. These data are in accordance with a phase II trial of pemafibrate treatment [7]. Long-term medical treatments may exhibit the rebound phenomenon. For instance, patients with type 2 diabetes treated with SGLT2 inhibitor often show a rebound in their weight during treatment [14]. In the present study, there was no evidence of the rebound phenomenon.
Hepatic inflammation markers such as serum transaminase levels remained within almost normal ranges during the three-year period. Nevertheless, a beneficial effect on high-density lipoprotein (HDL) cholesterol was noted only at the earlier study points, mainly the 6-month and 12-month marks. We previously reported the elevation of HDL cholesterol in half- and one-year studies [8, 13], and another two mid-term studies (follow-up period: one year or less) also reported significant elevation in HDL cholesterol from the baseline [15, 16]. This study did not document significant changes in HDL cholesterol at three years. A recent Japanese study also reported no significant change in HDL cholesterol level at 96 weeks [17]. However, in a phase II trial, HDL cholesterol levels were lower in the treatment group than the placebo group during a 72-week period [7]. In that regard, further studies are required in order to evaluate the effects of pemafibrate treatment with respect to HDL cholesterol level in patients with MASLD.
In our study, the platelet count showed a statistically significant increase over the three-year treatment period. However, we acknowledge that this change may have limited clinical relevance, as both baseline and follow-up values remained within the normal range. Platelet count is often considered a surrogate marker of portal hypertension and hepatic fibrosis in hepatology. Nevertheless, the majority of our patients had early-stage fibrosis, with 73% categorized as fibrosis stage 0 or 1, and only one patient had cirrhosis at baseline. This distribution of fibrosis stages likely explains the absence of any clinically meaningful thrombocytopenia and limits the interpretation of platelet changes in our cohort. In this study, we also evaluated the FIB-4 index as a simple and widely accepted non-invasive marker of liver fibrosis. Although a slight decrease in FIB-4 was observed over the three-year period, the change was not statistically significant. The fact that FIB-4 index is highly dependent on age, and the three-year follow-up period inherently led to aging of the cohort, could partially explain this difference. Furthermore, FIB-4 incorporates platelet count in its calculation, and the marked increase in platelet levels caused by pemafibrate may confound its interpretation. Lastly, as FIB-4 has relatively low sensitivity for detecting early-stage fibrosis, its utility may have been limited in our study population, the majority of whom had mild fibrosis at baseline.
The underlying mechanisms responsible for the beneficial effects of pemafibrate treatment in MASLD patients are largely unknown. Hepatic fat deposition, a hallmark of MASLD, was not attenuated by pemafibrate in an animal study [18], nor in a phase II trial where magnetic resonance imaging-derived proton density fat fraction (MRI-PDFF) was evaluated [7]. In PPAR-α knockout mice, the hepatic steatosis was aggravated [19]. One of the potential mechanisms might involve the size of the lipid droplets. Pemafibrate can reduce the size of lipid droplets without changing the total amount of hepatic fat [18]. Recently, lipid droplets have attracted considerable attention as metabolically active organelles. The modulation of lipid droplet size may alter physiological conditions of the liver, including inflammation and fibrosis.
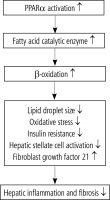
Recent evidence suggests that the beneficial effects of pemafibrate in MASLD extend beyond the modulation of lipid droplet size. As a selective PPARα modulator, pemafibrate activates a broad array of transcriptional programs that promote fatty acid β-oxidation in mitochondria, peroxisomes, and microsomes, reduce lipotoxicity, and suppress inflammatory signaling via transrepression mechanisms. In addition, PPARα activation has been shown to inhibit hepatic stellate cell activation and fibrogenesis in preclinical models, contributing to its antifibrotic potential. These pleiotropic effects of PPARα activation collectively improve hepatic inflammation, oxidative stress, and fibrosis progression in MASLD [20, 21]. A schematic overview of these mechanisms is provided in Figure 3.
Fig. 3
Mechanisms of PPAR-α activation in metabolic dysfunction-associated steatotic liver disease (MASLD)
There are some limitations to this study. This is a retrospective observational study without a control group, and the number of patients was small. Furthermore, pemafibrate treatment was combined with a statin in 26 patients (65%) and this could have enhanced the improvement of MASLD and affected the final results. In these 26 patients, long-term statin therapy had already normalized LDL cholesterol levels before starting pemafibrate therapy. Statin use can reduce the development and fibrosis of MASLD regardless of the presence of diabetes [22]. Lastly, histopathological assessment for hepatic fibrosis and size of lipid droplets was not performed. Furthermore, only a small number of patients had advanced fibrosis at baseline, which may limit the generalization of the results to more severe MASLD populations. However, this study has some advantages. Firstly, the observation period is the longest (three years) in the literature [15–17, 23]. Additionally, no diabetic subjects were included, because glucose metabolism and insulin resistance strongly influence the development and aggravation of MASLD. In particular, combination use of SGLT2 inhibitor and/or pioglitazone can affect the parameters regarding MASLD. Finally, unlike treatment with an SGLT2 inhibitor for patients with MASLD, no weight reduction was observed, ruling out an effect of body weight change.
Conclusions
Three-year pemafibrate therapy was safe, even when used in combination with statins. It appeared to improve the biomarkers of hepatic inflammation and fibrosis within one year, and these beneficial effects were maintained throughout the three years. The improvement of hepatic fibrosis markers may indicate potential regression of fibrosis. Long-term control of transaminases was associated with these favorable changes. Long-term pemafibrate therapy has the potential to prevent progression of MASLD.






