Summary
Ultrasound-guided thrombin injection (UGTI) is nowadays a very effective and safe method of iatrogenic pseudoaneurysm treatment. In the period 2015–2020 in our center we observed a total of 113 cases of iatrogenic pseudoaneurysms associated with the previous cardiovascular invasive procedures. Two different localizations of psA were encountered: femoral artery (102) and radial artery (11 cases), respectively. The vast majority of that cohort (in total 106 individuals) was successfully treated with the UGTI procedure. In high-volume centers the risk of an unsuccessful measure is negligibly low. Indispensable for the positive procedure outcome are adjusted equipment and optimal injection technique.
Introduction
The radial artery (RA) has nowadays emerged as the preferable access site for a majority of cardiovascular procedures due the fact that it has been associated with lower incidence of local complications in comparison to the femoral artery (FA) approach [1, 2], even though transfemoral access remains widely used especially when larger sheaths are required [3]. Evident examples of the above are: inability to apply transradial access for coronary artery catherization, intra-aortic balloon pump positioning as well as some types of electrophysiology procedures. Arterial cannulation is inherently associated with a risk of vascular complications including pseudoaneurysm (psA) that are encountered in both radial and femoral artery access cases [1–5]. Pseudoaneurysm is caused by a perforation in the arterial wall leading to blood flow into perivascular tissue, forming a sac that is surrounded by adventitia [3]. Among the various methods of treatment of psA the most popular are: watchful waiting for a spontaneous resolution, ultrasound-guided compression therapy (UGCT), ultrasound-guided thrombin injection (UGTI) as well as surgical treatment [1, 3]. Non-surgical methods remain effective in most cases, so they appear to be preferable methods for dealing with pseudoaneurysms.
Aim
The aim of the study was to evaluate the incidence, characteristics and treatment scheme of iatrogenic psA in a single high-volume cardiovascular interventions center. We attempted to reveal the main disparities both in clinical characteristics and efficacy of non-surgical therapy of RA and FA pseudoaneurysms.
Material and methods
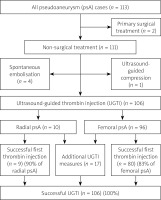
The study is a prospective analysis of femoral artery and radial artery pseudoaneurysm cases in a 6-year period (2015–2020) in Świętokrzyskie Cardiology Centre in Kielce, Poland. Among a wide range of cardiovascular procedures coronary artery catheterizations predominated with a total number of 7268. Transfemoral access was applied in 3689 (50.8%) interventions whereas transradial access was applied in 3589 (49.2%). Additionally, 2974 electrophysiology procedures as well as 1187 neurosurgical (concerning intracranial arteries) interventions were performed. All the above were pursued via femoral artery puncture. In the whole cohort 113 cases of pseudoaneurysm were diagnosed altogether. A routine ultrasound assessment of the puncture site was performed in a vast majority of patients after the interventional procedure during the index hospitalization. Nevertheless, 10 (8.8%) cases of psA were confirmed in ultrasonography after the discharge. The preferred method of pseudoaneurysm treatment in our center UGTI, which was applied in 106 (95.5% of all psA) cases.
Results
In the 2015–2020 period in our center we observed a total of 113 cases of iatrogenic pseudoaneurysms associated with the previous cardiovascular invasive procedures. Two different locations of psA were encountered: the femoral artery in 102 and the radial artery in 11 cases. The former related to: interventional treatment of coronary artery disease (81 cases of psA among 3789 invasive procedures: coronary angiography (CA) – 32, percutaneous coronary intervention (PCI) – 47 and intra-aortic balloon pump (IABP) placement – 2), electrophysiology procedures (15 cases of psA among 2974) and endovascular procedures in neurosurgery (6 cases of psA among 1187). The latter was solely encountered in coronary artery disease procedures (11 cases of psA among 3589; 4 cases after CA and 7 after PCI). Global prevalence of femoral artery pseudoaneurysm (FPA) was 1.29% (0.66% after CA, 2.22% after PCI and 3.77% after IABP) and radial artery pseudoaneurysm (RPA) was 0.30% (0.17% after CA and 0.56% after PCI).
The clinical characteristics of FPA and RPA cohorts are presented in Table I. Patients with radial artery psA were older and more often suffered from diabetes. There were differences in indications for invasive procedures between these two groups. In addition, the average sheath size was smaller in radial artery pseudoaneurysm cases (Table I).
Table I
Baseline characteristics of radial artery and femoral artery pseudoaneurysm (psA) cohorts
When comparing medical treatment in both groups there were no important disparities (Table II). Clinical manifestations of psA were similar in FPA and RPA except for pulsation that was more common in radial artery psA (Table III). Conversely, volume of psA and psA neck length were higher in the FPA group (Table III).
Table II
Medical treatment of radial artery and femoral artery pseudoaneurysm (psA) cohorts
Table III
Clinical manifestation and ultrasonographic characteristics of artery and femoral artery pseudoaneurysms (psA)
Among RPA a vast majority of patients underwent UGTI whereas in 1 case a spontaneous embolization of psA occurred. In the FPA cohort 2 patients were primarily qualified for surgical treatment due to large size of the pseudoaneurysm. Remaining patients underwent successful nonsurgical therapy with predominance of the UGTI procedure (Table IV). Effectiveness of the first thrombin injection was 90% in RPA and 85% in FPA, whereas all of the consecutive attempts were successful (Figure 1). Total amount of injected thrombin is presented in Table IV.
Table IV
Methods of treatment of radial artery and femoral artery pseudoaneurysms (psA)
Among individuals in whom a hemoglobin drop was observed after the procedure higher blood loss was encountered in femoral psA. In addition, only in the FPA group were there cases where blood transfusion was administered (Table IV).
Discussion
In the last years the percentage of the transradial approach in invasive procedures has overtaken the transfemoral access in our center, in accordance with current guidelines [6, 7]. The radial artery has become a preferable access for the majority of invasive cardiovascular procedures due to the fact that it reduces adverse clinical events, major bleeding as well as all-cause mortality [8, 9]. The MATRIX study revealed that the radial approach was associated with a significantly lower rate of surgical access site repair [8]. Moreover, RIVAL study investigators confirmed that radial artery puncture is correlated with 63% relative risk reduction of all major vascular complications and 70% relative risk reduction of pseudoaneurysm needing closure when compared to femoral artery access [9]. Our analysis revealed that the risk of iatrogenic pseudoaneurysm occurrence was over 4-fold higher in transfemoral procedures. Importantly, in the whole cohort only 2 patients with pseudoaneurysm were transferred directly for surgical treatment, which is contrary to numerous previous reports [3, 10–13]. In the past surgical procedures were the prevalent method of therapy of pseudoaneurysms, especially applied in large femoral psA when rapid expansion of the puncture site occurred and either concomitant distal ischemia or neurologic deficit due to local pressure was observed [12]. The precise engaging criteria for an operative procedure remain unknown. Nevertheless, psA diameter greater than 3 cm is a common indication for surgical management [13]. The most important finding in our study is that none of the patients qualified for conservative treatment crossed over to the operative arm. Non-surgical treatment was successful in all 111 consecutive patients. The prevalent method of pseudoaneurysm therapy in our center was UGTI, applied in 106 (93.8%) individuals. What is important, the percentage of the first thrombin injection success was as high as 90% in radial psA and 85% in femoral psA. Ultrasonographic assessment can reveal the predictors of pseudoaneurysm recurrence after UGTI such as the late to early velocity index [14].
In a few cases other methods such as compression or watchful waiting appeared to be sufficient. Based on previous studies and case reports, among non-surgical methods of pseudoaneurysm management one of the most popular is ultrasound guided compression therapy (USCT). It is a widely available, safe and effective technique that ensures psA repair at a satisfactory level of 74% [10] and 69% [11]. Failure of UGCT is attributed to factors such as puncture site artery, interventional procedure, size of aneurysm, neck diameter as well as use of anticoagulants [11]. Apart from UGTI also compressive technique is applicable in radial psA as well with acceptable results that were confirmed by Burzotta et al. with a positive result in 10 out of 12 cases [15]. On the other hand there are some limitations of UGCT: it is time consuming and a very painful procedure for the patients. When comparing UGCT and UGTI effectiveness it seems that the latter has become the preferable method of treatment of iatrogenic pseudoaneurysms; the latest reports confirmed their efficacy as 94.8% and 98.9%, respectively [16, 17].
What is more, radial pseudoaneurysms have numerous disparities from femoral psA – they are smaller but usually more painful, with no asymptomatic cases, often encountered in women. There is no risk of serious bleeding complications in radial psA or risk of limb ischemia. During the radial psA UGTI the most preferable equipment is a linear US transducer with frequency of 7–10 MHz along with a short-needle low-diameter syringe (e.g. 0.40 × 13 mm/27G × 1/2’’) for precise dosing of the thrombin.
Study limitations: Our study has several limitations. It is a single-center observational study. Despite the fact that the number of examined patients after a cardiovascular invasive procedure was high, our cohort of pseudoaneurysm cases is limited. What is more, some potentially important parameters might not have been included in the analysis. Finally, this is a single-center study; therefore, the results should be interpreted with caution.
Conclusions
Ultrasound-guided thrombin injection (UGTI) is nowadays a very effective and safe method of iatrogenic pseudoaneurysm treatment. In high-volume centers the risk of an unsuccessful measure is negligibly low. Indispensable for the positive procedure outcome are adjusted equipment and optimal injection technique.