Summary
Approximately one-third of our sample was diagnosed with definitive subclavian vein occlusion (SVO) in the long term after cardiac resynchronization therapy-defibrillator implantation. Even though venography is accepted as the gold standard method in detecting SVO, we demonstrated that venous Doppler ultrasonography, a useful, simple and non-invasive method, can be a valuable tool in detecting SVO, particularly under elective conditions before cardiac resynchronization therapy replacement or lead extraction procedures, allowing for preparations to be made to avoid potential errors.
Introduction
After cardiac implantable electronic device (CIED) implantation, capsule formation around the coil and inflammation and fibrosis around the leads may occur [1, 2]. Since three leads are used in cardiac resynchronization therapy (CRT) implantation, patients undergoing CRT implantation experience three times more remodeling than patients undergoing other CIED implantations. Venous leads are larger in diameter, especially when a cardiac resynchronization therapy-defibrillator (CRT-D) is used.
Ipsilateral subclavian vein patency is crucial during CIED replacement. The European Lead Extraction Registry study reported that the presence of occlusion or thrombus in the ipsilateral venous pathway during CIED replacement was an independent predictor of major vascular complications [3, 4]. Although there are studies on the frequency and risk factors of subclavian vein occlusion (SVO) after CIED implantation [5–17], no study has been conducted to date investigating the frequency of SVO in the isolated CRT-D patient group. The incidence of SVO after biventricular device implantation (ICD only 47%) has been reported to be 12%. Patients planned for CRT implantation are at higher risk for venous stenosis compared to patients planned for other CIED implantations [7–10, 12]. Moreover, CRT implantation is a more complex procedure – requiring at least 3 venous punctures and leads – compared to other CIED implantations.
In all studies on CIED-related venous stenosis or occlusion, venous stenosis and occlusion were generally assessed using ipsilateral venography before the procedure [5–7, 9–17]. Venous Doppler ultrasonography (USG), an inexpensive, simple, non-invasive, and reproducible method, is recommended to be performed routinely after CIED implantation, especially in patients with complaints of upper extremity edema, while it is not performed routinely before CIED battery replacement. There is one study in the literature investigating the incidence of pacemaker lead-related venous thrombosis 3 months after CIED implantation [8], but there is no study evaluating venous stenosis and occlusion in the long term.
Aim
In view of the foregoing, we carried out this study to investigate the frequency of long-term SVO following CRT-D implantation using venous Doppler USG.
Material and methods
Study design and setting
This study was designed as a cross-sectional and single-center study. The study protocol was approved by the local ethics committee. It was conducted at the Arrhythmia Clinic of Adana City Training and Research Hospital, a tertiary referral hospital in the central district of Adana province in south Turkey, between 2018 and 2019. The study was conducted in accordance with the principles outlined in the Declaration of Helsinki. Written informed consent was obtained from all patients included in the study.
Population and sample
The study population consisted of 202 patients who underwent CRT-D implantation and elective battery replacement. A power analysis, based on 80% power and probability (p) < 0.05 in line with literature data, revealed that it would be sufficient to include 100 patients in the sample. Patients who refused to undergo the procedure, did not sign the consent form, had symptoms and signs of upper extremity venous stenosis, whose subclavian vein image could not be obtained by venous Doppler USG, had surgery in the deltopectoral region or had an infection, had a pacemaker lead problem (fracture, infection, etc.), had erosion at the pacemaker site, had ≤ 2 transvenous electrodes (atrial fibrillation), were allergic to contrast material, had serum creatinine ≥ 2 mg/dl, had venous malformations, or had ipsilateral dialysis fistula were excluded from the study. Sufficient Doppler US images could not be obtained in 9 patients. In the end, the study sample consisted of 181 patients.
Data collection procedure
All patients underwent a detailed and complete cardiac evaluation and physical examination, their medical histories were taken, smoking status and whether they had hypertension (HT), diabetes mellitus (DM), or hyperlipidemia (HLD) were noted, systolic and diastolic blood pressures and pulse rates were measured, and New York Heart Association (NYHA) stages were determined. The cause of heart failure was categorized as ischemic or non-ischemic. Patients’ medical and device histories were queried and duration of implantation was calculated. Patients underwent a comprehensive laboratory examination, 12-lead electrocardiography (ECG), thoracic X-ray, and CIED measurements, and their last calculated left ventricular ejection fraction (LVEF) value was recorded from the chart.
Venous Doppler ultrasonography procedure
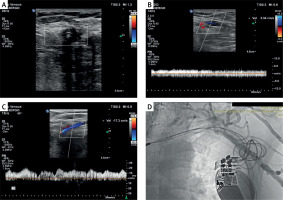
A specialist with at least 10 years of Doppler USG experience routinely performed US examinations on all patients in the supine position using a USG device (Philips EPIQ 7, Philips Health Care, Bothell, WA, USA) and a 12-5 MHz high-resolution linear probe. Both upper extremities of the patients were evaluated bilaterally with venous Doppler USG. To this end, brachial, axillary, subclavian, and jugular vein structures were firstly evaluated in B-mode. The probe was positioned to avoid venous compression, and lumen width, flow direction with color Doppler, and flow pattern with spectral Doppler were assessed. Consequently, complete loss of flow in at least three different regions of the subclavian vein was deemed to indicate a preliminary diagnosis of SVO (Figure 1 A). Antegrade continuous flow pattern in the ipsilateral and contralateral axillary veins, presence of respirophasic flow pattern, and axillary vein flow velocity were evaluated (Figures 1 B, C, respectively). All venous Doppler USG images were acquired using the device software. Images were evaluated by two different radiologists, and if consensus was not reached, a third radiologist’s opinion was obtained.
Figure 1
A – Doppler USG image of flow loss in the subclavian vein due to luminal obstruction with 3 pacemaker leads. B – Doppler USG image pertaining to the ipsilateral axillary vein featuring subclavian vein occlusion: i) disappearance of respirophasic flow, ii) decreased flow (3.34 cm/s) iii) antegrade continuous flow. C – Normal Doppler USG image pertaining to the contralateral axillary vein of a patient with left subclavian vein occlusion; i) antegrade respirophasic flow pattern, ii) normal flow velocity (17.3 cm/s). D – Verification of the occlusion by Doppler USG in the left subclavian vein of the same patient with venography image
Venography procedure
Definitive diagnosis of SVO was made with venography on the day of CRT-D battery replacement in patients with a preliminary diagnosis of SVO by venous Doppler USG. To this end, a 1.3-gauge venous cannula was inserted into the ipsilateral antecubital vein and 20–30 ml of non-ionic, water-soluble contrast agent was prepared. A tourniquet was applied on the venous pathway before contrast agent injection. After contrast agent injection, the tourniquet was removed and then 20 ml saline was infused. Subsequently, complete cineangiography of the axillary vein, subclavian vein, and brachiocephalic trunk was performed in the anterior posterior projection. The images were evaluated by an experienced electrophysiologist. Abrupt cessation of contrast flow in the subclavian vein and visualization of the collateral system were deemed to indicate a definitive diagnosis of SVO (Figure 1 D).
Statistical analysis
Statistical analyses of the collected data were performed using SPSS version 23.0 (IBM Corp., Armonk, NY, USA, 2015) for Windows. The results of the statistical analyses were expressed using descriptive statistics, i.e., mean ± standard deviation values in the case of continuous variables determined to conform to the normal distribution, median values with interquartile ranges in the case of continuous variables determined not to conform to the normal distribution, and numbers (n) and percentage (%) values in the case of categorical variables. The normal distribution characteristics of continuous variables were analyzed using the Kolmogorov-Smirnov test. In comparing numerical variables between any two groups, Student’s t-test and the Mann-Whitney U test were used for normally and non-normally distributed variables, respectively. The χ2 test was used to compare categorical variables between the groups. The κ coefficient was used to examine the interobserver and intraobserver variabilities of venous Doppler USG and venography parameters. Sensitivity, specificity, and positive and negative predictive values of venous Doppler in predicting SVO as identified by venography were calculated. T1-weighted, fat-saturated axial magnetic resonance imaging (MRI) was the reference standard. Variables found to be significantly associated with SVO in univariate analysis were further evaluated using multivariate logistic regression analysis. Values of p < 0.05 were deemed to indicate statistical significance.
Results
The mean age of the study sample, consisting of 110 males and 71 females, was 63.9 ±8.1 years. A definitive SVO diagnosis characterized by complete loss of flow and absence of a respirophasic flow pattern was made in 59 (33%) patients. Patients with SVO were significantly older, were more likely to have HT and DM, and had longer duration of CRT implantation than patients without SVO. There was no significant difference between the groups in other demographic and clinical characteristics (Table I). Laboratory analyses revealed significantly higher glucose and total cholesterol levels in patients with SVO than in those without SVO. There was no significant difference between the groups in other laboratory and LVEF parameters (Table I).
Table I
Distribution of patients’ demographic, clinical, and laboratory characteristics by study group
Analysis of Doppler USG data revealed no reciprocal flow pattern in the ipsilateral axillary vein in all patients with SVO compared to only 2 patients without SVO (Table II). There was no significant difference in axillary vein diameter between the groups (Table II). Except for 2 cases where SVO originated from the mid 1/3 of the subclavian vein, in all cases with SVO, it originated from the distal 1/3 of the subclavian vein. Sensitivity, specificity, and positive and negative predictive value of venous Doppler USG in predicting SVO as identified by venography were calculated (Table III). Cohen’s κ coefficients calculated for venography and venous Doppler USG measurements revealed intra- and inter-observer variability values above 90%.
Table II
Distribution of patients’ venous Doppler USG results by study group
Table III
Diagnostic accuracy of venous Doppler USG in detecting SVO based on the diagnosis of SVO established by venography
Variables found to be significantly associated with SVO in univariate analysis were further evaluated using multivariate logistic regression analysis. Consequently, DM and advanced age were found to be independent variables significantly predicting SVO (p < 0.05 for each case) (Table IV).
Discussion
Our study is the first to date to evaluate SVO with venous Doppler USG before CRT replacement. We concluded that venous Doppler USG is a useful method in detecting long-term CVE after CRT-D implantation. We also observed that approximately one-third of patients who underwent CRT-D implantation developed SVO. There are no clear data in the literature regarding the incidence of SVO specifically in CRT-D patients. Early detection of SVO in CRT-D patients who may require lead extraction or a fourth lead due to lead dysfunction is very important in order to be prepared for potential errors in advance of the procedure. Another important finding of our study is that, in line with literature data, DM and advanced age were independently associated with the development of SVO.
CEID implantation initially involved a simple single-chamber pacemaker, then a 2-chamber pacemaker, 3-chamber CRT (without ICD lead), 3-chamber CRT-D (with ICD lead), and finally a 4-chamber left bundle branch-optimized CRT (LOT-CRT). Patency of venous structures, i.e., axillary and subclavian veins, is essential for these procedures to be performed properly. As with all CIED devices, battery replacement is necessary after CRT implantation. In addition to elective battery replacement, procedures such as new lead requirement, i.e., CRT upgrade, lead revision and lead extraction, especially in cases such as lead fracture, microfracture, isolation problems (lead impedance changes), and infection, may also be required [3]. The incidence of SVO in lead extraction studies has been reported to be approximately one in four (25–26%), and special devices and materials may be required before the procedure in this patient group [10, 11]. Ipsilateral subclavian vein patency, generally assessed by ipsilateral venography, is particularly important in cases where a new lead is needed due to lead problems that may occur during the replacement procedure. In studies on subclavian vein stenosis or occlusion, venography is preferred for diagnostic evaluation [5–7, 9–17]. Major vascular complications are more common in cases with occlusion in the ipsilateral venous pathway. In this context, early detection of SVO allows for preparing for procedures and complications that may occur during and after CRT replacement [3, 4].
Subclavian vein stenosis is a common complication of transvenous lead implantation in patients with CIEDs, the incidence and severity of which vary according to device, lead, and patient characteristics [5–17]. While the incidence of venous stenosis has been reported to be between 20% and 50%, the incidence of total occlusion has been reported to be between 8.2% and 26% [5–17]. The primary pathophysiological factors leading to subclavian vein stenosis after pacemaker implantation are reportedly procedure-related venous endothelial trauma and secondary inflammation [17]. Considering that 91% of cases with venous thrombus are asymptomatic, another pathophysiology that is likely to lead to occlusion in the future is acute and subacute thrombi occurring in the lead site [8]. However, studies have shown that post-procedure anti-platelet and anti-coagulant therapy does not affect the occurrence of venous stenosis [7, 15]. It has been reported in the literature that advanced age, DM, HT, and smoking are associated with the development of SVO [6, 7]. In parallel, we found DM and advanced age to be independent risk factors for the development of SVO. These findings suggest that especially older patients and those with DM should be closely monitored for the development of SVO.
A recent retrospective study reported the incidence of subtotal SVO, subtotal SVO after CIED implantation, and total occlusion as 70% to 99%, 12.3%, and 8.2%, respectively [6]. Another study conducted with 212 patients reported the total occlusion rate as 26% and concluded that the primary determinants of the development of venous stenosis were the number of leads and the total implanted lead diameter [7]. They found that the most significant predictors of SVO were advanced age and having three leads [7]. In comparison, we detected SVO in 1 out of every 3 CRT patients, which is a higher rate than the SVO incidences reported in the literature. While several other studies have found a significant relationship between lead number and stenosis [8–10], a number of others have not [11, 15, 16, 18].
LOT-CRT, a feasible and safe method that provides greater electrical resynchronization compared to traditional CRT, can be a good alternative to traditional CRT, especially in cases where traditional CRT cannot provide sufficient electrical resynchronization [19]. We have not evaluated patients who underwent LOT-CRT, as their elective replacement was not due. Then again, the lead used for left bundle branch area pacing during LOT-CRT implantation was thin, with a thickness of 4.1F, suggesting that the incidence of SVO would not be significantly affected. In addition, considering that CRT has more leads than other CIEDs and that each lead is exposed to structural and functional problems in the long term and therefore more revisions will be needed in patients undergoing CRT, ipsilateral lead patency becomes even more important in patients undergoing CRT. To the best of our knowledge, there is no study investigating the incidence of venous occlusion in patients undergoing only the CRT-D procedure. In a study with a 1.7-year follow-up period, the incidence of SVO in CRT patients was reported as 12% [9]. The fact that we found the incidence of SVO much higher in our sample compared to the incidence of SVO reported in the said study may be attributed to our much longer follow-up period and to the fact that only 47% of the patients included in the said study had an ICD lead, which has been shown to be an independent predictor of SVO in the literature [10, 12].
Venous Doppler USG examination is not routinely performed before or after CIED implantation or CRT battery replacement, except in patients with signs and symptoms of upper extremity venous thrombosis. Compared to venography, which is a relatively invasive procedure that involves the use of 10–20 ml of contrast medium and is performed on the operating table immediately before the replacement procedure, thus requiring hospitalization, venous Doppler USG is a non-invasive, reproducible, inexpensive procedure that does not involve the use of opaque material and is performed under elective conditions before replacement, thus not requiring hospitalization. In sum, ipsilateral venous Doppler USG examination is a valuable method in diagnosing venous occlusion, which occurs in one in every 4 patients, before pacemaker battery replacement, and in ensuring that the necessary preparations are made based on this diagnosis.
There were several limitations to our study. First, its cross-sectional and single-center design may have led to less significant results compared to the randomized, prospective, and multicenter design. Secondly, since only patients with CRT-D were included in the study, venous Doppler USG examination was not performed according to the type of CIED. Thirdly, the fact that all patients included in the study were asymptomatic and were due for elective replacement, and that patients with symptoms of upper extremity venous stenosis were excluded may have impacted the incidence of venous stenosis in the sample. Fourthly, patients who could not undergo venous Doppler USG due to insufficient patency in relation to subclavian vein anatomy had to be excluded from the study. Lastly, considering that CRT has a higher risk of causing venous stenosis compared to other CIEDs, we evaluated only venous occlusion and did not evaluate the various degrees of venous stenosis detected by venography, as we wanted to identify patients with complete occlusion, i.e., SVO, using venous Doppler USG. If we had evaluated venous stenosis, we might have obtained different results.
Conclusions
Approximately one-third of our sample was diagnosed with definitive SVO in the long term after CRT-D implantation. Even though venography is accepted as the gold standard method in detecting SVO, we demonstrated that venous Doppler USG, a useful, simple and non-invasive method, can be a valuable tool in detecting SVO, particularly under elective conditions before CRT replacement or lead extraction procedures, allowing for preparations to be made to avoid potential errors. We also found that DM and advanced age were independent risk factors for SVO, suggesting that especially elderly patients and patients with DM should be closely monitored for the development of SVO and smaller-diameter transvenous leads should be used in their procedures. Future larger-scale, multicenter, randomized studies are needed to corroborate and expand on our findings.







