Purpose
Intracavitary brachytherapy (BRT) has a significant role in the treatment of gynecologic malignancies [1,2,3,4,5,6,7]. It offers the major advantage of rapid dose fall off that provides better dose sparing of the surrounding tissues and critical organs adjacent to the target [8,9]. The recent National Cancer Database analysis showed that consolidation BRT was a critical component for locally advanced cervical cancer [5]. Additionally, vaginal BRT, alone or in combination with external beam radiotherapy (EBRT), is used in a selected group of patients with cervical, vaginal or vulvar cancer [10]. Recently there is growing interest in the use of vaginal BRT alone as adjuvant treatment after surgery in endometrial carcinoma (EC) [3].
The vaginal cuff is the main location of local relapses after surgery in EC and it was shown that adjuvant vaginal BRT alone produced similar local control and survival rates compared to external pelvic radiotherapy in patients with intermediate or high-intermediate risk factors [11]. The most commonly used applicator for intracavitary BRT in EC is a centrally located single channel (CSC) vaginal cylinder and the dose is generally prescribed to either the surface of the cylinder or 0.5 cm in depth. These applicators can also be used for primary vaginal cancer or recurrent vaginal tumors either alone or combined with EBRT. However, these CSC applicators offer limited possibilities in the planning optimization due to their radial symmetry of dose distribution and can be used only in tumor volumes having thickness or depths less than 5 mm from the applicator surface. To improve the quality of treatment in deeper and asymmetric tumors, several multichannel (MC) applicators have been developed. In these commercial MC applicators, additional peripheral channels added to the central axis provide more conformal dose distributions up to 1 cm depth. Even with these MC applicators, vaginal lesions thicker or deeper than 1 cm cannot be covered effectively without an additional unwanted dose to the surrounding normal tissues such as bladder, rectum or sigmoid colon. Therefore standard MC applicators lose their advantage in these cases and interstitial BRT is frequently required [12,13,14,15], which needs expertise in the field, general anesthesia and hospitalization.
In the present study we aimed to design a novel high-dose-rate (HDR) intensity-modulated brachytherapy (IMBRT) applicator as an alternative to interstitial BRT in vaginal tumors thicker than 1 cm and in cervical tumors with gross residual disease after EBRT or with difficult anatomy in order to cover the tumor adequately while decreasing the dose to the surrounding normal tissues. In addition to the novel design, this study investigates the adaptation of current three-dimensional (3D) printer technology to medical applications.
When the novel applicator meets all requirements for clinical use (e.g. safety, sterilization, health and environmental protection), the workflow for the use of a novel applicator in the treatment can be divided into five steps: 1) making a preliminary patient-specific computed tomography (CT) simulation and preplan to determine the type, length and position of the compensator materials using an automated contouring template, 2) scanning treatment planning CT with defined preplan conditions including type, length and position of the materials using an extended CT scale and metal artifact reduction algorithm, 3) transferring planned dwell time and position of the radioactive source from preplan CT to new planning CT, 4) controlling transferred treatment plan parameters and performing necessary arrangements to improve the quality of treatment planning, 5) approval of the treatment plan and transfer to the treatment machine.
Material and methods
This novel IMBRT applicator was designed by a senior radiation oncologist (H.F.A.) and a medical physicist (F.B.). All of the treatment planning simulations and quality control (QC) tests were performed using Varian BrachyVision v.8.9 (Varian Medical Systems, Palo Alto, CA) treatment planning system (TPS) with TG-43-based formalism and a Varian GammaMed iX Plus HDR 192Ir BRT unit (Varian Medical Systems, Palo Alto, CA), respectively. To evaluate the feasibility of this novel IMBRT vaginal applicator, investigations were carried out following 4 main steps: 1) material selection for applicator, phantom, auxiliary fixation tools and compensators, 2) modeling and printout of the products, 3) evaluating printing accuracy of the 3D printer, 4) performing mechanical and dosimetric QC tests.
Material selection
In the production of the applicator, auxiliary fixation tools and QC phantom polylactic acid (PLA) filament with a density of 1.25 g/cm3 was used as the printing material. PLA is suitable for medical applications since it is biocompatible and it can be sterilized with ethylene oxide (EtO) and gas plasma. Aluminum (density: 2.5 g/cm3), stainless steel (density: 8.0 g/cm3) and Cerrobend alloy (density: 9.4 g/cm3) were selected as compensator materials according to their radiation attenuation characteristics. The main purpose of the directional compensator is not to provide complete shielding of critical organs such as the rectum and bladder near the target volumes but to modulate dose distribution to reduce the critical organ dose and increase target coverage.
Modeling and 3D printing
All of the products were modeled in SketchUp Pro 2017 (Trimble Inc., Sunnyvale, CA) 3D modeling program. Additionally, Meshmixer software (Autodesk, San Rafael, USA) was used to make the necessary arrangements and to save the file in .stl format for 3D printing.
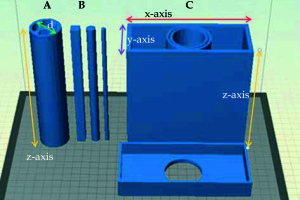
The IMBRT applicator was modeled in two different diameters of 30 and 35 mm. The total length of the applicator was 190 mm. The designed applicator had five removable parts, i.e. the external body, internal body, source transfer channels, compensator materials and auxiliary fixation tools, as illustrated in Figures 1 and 2. The applicator had four chamber cavities allowing placement of source transfer channels or compensator materials according to the target and critical organs location and one central channel for tandem insertion in the internal body. In the peripheral channels the source was encapsulated within a 5-French plastic catheter. The special design of the applicator allows different combinations of the source transfer channels and desired lengths of compensator materials during application and treatment. Fixation tools (Figures 1E and 2B) compatible with the BRT treatment couch were also designed to fix the removable part of the applicator. Additionally, the specific QC phantom and applicator were modeled (Figure 3) and printed out to perform all QC tests in the same measurement setup. The output of the products except for compensator materials was taken using PLA filament in a MakerBot Replicator Z18 3D printer (MakerBot Industries, Brooklyn, USA) and modeled applicators were printed by choosing 0.2 mm print resolution, 2 layer support material and 100% infill percentage.
Fig. 1
Novel IMBRT applicator modeling A) internal body, B) external body, C) source transfer channels, D) compensator materials and E) fixation tools

Printing accuracy of 3D printer
Calibration of the 3D printer was performed before each printing process to improve the quality of the printout. Printing accuracy of the product was also evaluated through physical measurements using a Vernier caliper with a resolution of 0.1 mm. Physical measurements were performed at multiple positions along the prints and all measurements were compared with the designed dimensions of the product in modeling software. Additionally, total printing time was evaluated for all parts of the modeled applicator.
Mechanical QC tests
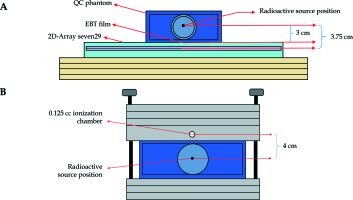
Mechanical QC tests were performed in three steps: 1) catheter and source dwell positioning accuracy, 2) reproducibility of catheter and source position for different step sizes, and 3) linearity of relative dose with changing dwell time and compensator materials. In the first step, 5-French catheters were inserted into the peripheral transfer channel of the designed applicator and a source dwell positioning accuracy test was performed with Kodak EDR2 radiographic film (Eastman Kodak Company, Rochester, NY, USA). To verify the dwell positioning accuracy of the radioactive source, x-ray markers were placed into the catheter and reference radiography was acquired to image the reference x-ray marker position. Next, an autoradiograph was performed with radioactive source and irradiated dwell positions were compared with the reference dummy marker position. The tolerance value for source dwell positioning accuracy was less than 2 mm. Additionally, the actual catheter insertion position obtained with autoradiography was compared with the reconstructed catheter position in the TPS. In the second step, reproducibility of the catheter and source position for three different step sizes of 1, 0.5 and 0.1 cm was tested using the 2D-Array seven29 (PTW-Freiburg, Germany) two dimensional (2D) ionization chamber system (Figure 4A). All measurements were repeated 3 times for all step sizes and in every measurement the catheter was removed and re-inserted into the transfer channel to evaluate the reproducibility of the insertion and source position. In the analysis, the gamma evaluation method was used under the criteria of 1 mm distance to agreement (DTA) and 1% dose difference (DD) of local dose. As software PTW VeriSoft v.5.1 (PTW-Freiburg, Germany) was used and relative dose distributions were compared during the analysis. In the third step, linearity of the relative dose with changing time was tested in order to evaluate the linearity of the HDR BRT unit for different compensator materials. Linearity test measurements were taken using PTW 31010 semiflex ionization chambers (PTW-Freiburg, Germany) at two different stages. In the first stage the radioactive source was sent to a certain position within the novel IMBRT applicator and irradiations were carried out in 6 different periods of 5, 6, 10, 15, 25 and 35 seconds. In the second stage aluminum, stainless steel and Cerrobend alloy were placed into the modeled QC applicator instead of PLA and similar measurements were repeated for different compensator materials. Distance between the source and ionization chamber was fixed to 4 cm as shown in Figure 4B. Transit time corrections were applied for all measurements by subtracting 5 seconds from the reading in all measurements and the net reading was obtained. Then, all of the measurements were normalized to 10 seconds net reading to evaluate the linearity of the system for defined conditions. The tolerance value for the linearity was less than 2%.
Dosimetric QC tests
In the first step of dosimetric QC tests, measurements were performed in homogeneous medium. The IMBRT applicator was placed in the designed QC phantom and the CT of the QC phantom was scanned with 2 mm slice thickness using a Toshiba Aquilion LB CT (Toshiba Medical Systems, Otowara, Japan) having 100 kVp tube voltage and 350 mAs current value. CT images were then transferred to BrachyVision TPS via a digital imaging and communications in medicine (DICOM) connection. For each designed applicator two different treatment plans including the central CSC and MC in homogeneous medium were created and irradiated in the treatment device. The measurement setup for the 2D dose was set as shown in Figure 4A. 2D dose maps calculated at defined depths were then compared with 2D-Array ionization chamber system and Gafchromic EBT3 film (Ashland Specialty Ingredients, NJ, USA) measurements using gamma analysis method for the local dose. As software PTW VeriSoft v.5.1 was used and relative dose distributions were compared during the analysis. Additionally, point dose measurements were performed with the PTW 31010 semiflex ionization chamber at defined setup conditions as shown in Figure 4B and the measured point dose was compared with the calculated dose in homogeneous medium. In point dose measurement cross-calibration was performed during the use of the PTW 31010 semiflex ionization chamber.
In the second step, the effects of compensator materials on dose attenuation were measured with the QC applicator using the 2D-Array seven29 ionization chamber system. However, the measured dose distribution could not be compared with calculated data in TPS since TG-43 formalism presumes homogeneity of the medium and so the heterogeneity was not taken into account during dose calculation. Therefore compensator materials including aluminum (density: 2.5 g/cm3), stainless steel (density: 8.0 g/cm3) and Cerrobend alloy (density: 9.4 g/cm3) were placed into the anterior region of the applicator and the dose line profile obtained with compensator materials was compared with the PLA-based measurements to evaluate the attenuation properties of the selected materials for a designed compensator geometry.
Results
Printing accuracy of 3D printer
The measured differences between the modeled and the printed dimensions of all the products were within 0.5 mm (±0.1 mm resolution of the Vernier caliper). Printed dimensions of the external body and internal body were on average 0.4 mm (range: 0.3-0.5 mm) greater than modeled and dimensions of the transfer channels were on average 0.2 mm (range: 0-0.3 mm) smaller than the designed ones. However, a snug fit of the 5-French catheters in the printed channels was ensured. In addition to printing accuracy, total printing time was evaluated for defined conditions (printing resolution: 0.2 mm, infill percentage: 100%, etc.) and it was almost 20 hours for all parts of the applicator.
Mechanical QC tests
Source dwell positions measured by EDR2 films were found to be in good agreement with reference x-ray marker position and the positional differences were smaller than 0.5 mm. Additionally, the actual catheter insertion position obtained with autoradiography was well matched (≤ 1 mm along the axis) with the reconstructed catheter position in the TPS. The reproducibility of the catheter and source positions for different step sizes was analyzed and 11 of the 12 measurements had a greater than 99% passing rate for 1 mm DTA and 1% DD criteria (Table 1). The lowest gamma passing rate was 97.7% for 0.1 mm step size when a comparison was made between 3 consecutive measurements. The linearity values for different applicators and selected compensators were found to be less than 1.5% (Table 2).
Table 1
Reproducibility of the catheter and source position for different step sizes
Table 2
Linearity value for novel IMBRT applicator and different compensator materials
| Time (s) | Linearity | Linearity | Linearity | Linearity | Linearity | Linearity | Linearity |
|---|---|---|---|---|---|---|---|
| 30 mm applicator | 35 mm applicator | QC applicator | QC applicator | QC applicator | QC applicator | QC applicator | |
| PLA | Free Air | Aluminum | Stainless steel | Cerrobend | |||
| 5 | NA | NA | NA | NA | NA | NA | NA |
| 6 | 1.0036 | 1.0118 | 1.0041 | 0.993 | 0.9853 | 0.9885 | 0.9892 |
| 10 | 1.0043 | 0.9881 | 1.0099 | 0.9946 | 1.0064 | 0.9987 | 1.0000 |
| 15 * | 1.0000 | 1.0000 | 1.0000 | 1.0000 | 1.0000 | 1.0000 | 1.0000 |
| 25 | 1.0007 | 0.9985 | 1.0008 | 1.0015 | 1.0000 | 1.0025 | 0.9928 |
| 35 | 1.0029 | 0.9919 | 1.0058 | 0.993 | 1.0018 | 1.0000 | 0.9964 |
Dosimetric QC tests
Point doses measured in homogeneous medium were found to be compatible with calculated doses in the TPS. The maximum difference was 3.8%, as presented in Table 3. This difference was first thought to be due to deviation in the catheter and radioactive source position. In fact even 1 mm deviation from the real position could create dose differences up to 10% depending on the dose gradient in treatment planning. To minimize this problem a QC phantom was designed and measurements were performed in a fixed geometry. However, there was still positional uncertainty due to the source dwell positioning accuracy, which was lower than 0.5 mm as measured in the mechanical QC test. The second possibility for this difference was that although the phantom used in the QC tests was produced from water equivalent material, the water equivalent distance (WED) might differ slightly from the physical distance (PD) due to the minimal density differences. PD was arranged as 4 cm in the QC phantom in the measurements but measured WED was found to be 3.7 cm in the TPS. Therefore as presented in Table 3 measured point doses were found to be somewhat higher than TPS values for all treatment plans since TG-43 formalism calculated dose according to the PD of 4 cm.
Table 3
Point dose measurements in homogeneous medium
| Point dose | 30 mm diameter applicator | 35 mm diameter applicator | ||
|---|---|---|---|---|
| CSC | MC | CSC | MC | |
| TPS (cGy) | 208.5 | 215.2 | 219.6 | 220.3 |
| Measurements (cGy) | 213.6 | 223.4 | 222.4 | 228.1 |
| Dose differences (%) | 2.4 | 3.8 | 1.3 | 3.5 |
In 2D dose analysis measurements were taken using the 2D-Array ionization chamber system and EBT3 Gafchromic film at 3.75 cm and 3 cm distance from the source axis, respectively (Figure 4A). The 2D dose analysis results and the dose distributions obtained at the relevant depth in the TPS were compared with the gamma analysis method. As criteria of gamma analysis four different DTA and DD values (3 mm/3%, 3 mm/5%, 5 mm/3% and 5 mm/5%) were compared. The results obtained with both dosimetric systems are presented in Tables 4 and 5. All of the measurement results were above the tolerance value of 90% gamma passing rate.
Table 4
Comparison of TPS and 2D-Array measurement in homogeneous medium
Table 5
Comparison of TPS and EBT3 film measurement in homogeneous medium
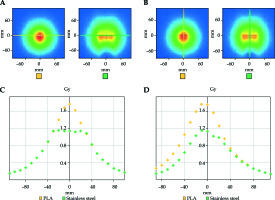
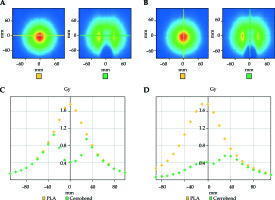
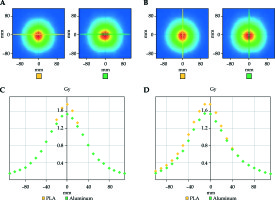
Attenuation characteristics of the compensator materials in heterogeneous medium are illustrated in Figures 5-7. In modeled geometry, aluminum, stainless steel and Cerrobend alloy attenuated the dose more than 15%, 35% and 75% with respect to PLA respectively. Attenuation ratios of the materials were found to be associated with density of the compensator as expected.
Fig. 5
Measured dose map A) in x axis, B) in y axis and line dose graphic C) in x axis, D) in y axis for PLA (yellow) and aluminum (green)
Discussion
BRT significantly increases loco-regional control when applied alone or combined with EBRT in gynecological malignancies [6,7]. The main beneficial effect of BRT is that it provides better organ sparing of the surrounding tissues due to the rapid dose fall off with distance from the target volume [8,9].
Vaginal BRT alone or combined with EBRT in EC is being widely used in the adjuvant setting and the technical developments in the computer era led to replacement of 2D radiography-based treatment planning by CT-based treatment modalities, with improvements in imaging quality and availability. In conventional techniques it was mostly recommended that dose prescription should be made at a depth of 5 mm from the applicator surface or to the surface [2]. Uncertainties due to patient anatomy and tumor size have been neglected, which may lead to overdoses or underdoses in the target and toxic doses to the critical organs in the vicinity. With the advent of three dimensional brachytherapy (3DBRT), several studies have been carried out to eliminate the uncertainties in gynecological BRT. In a study performed by Kim et al. [16] it was reported that the use of 3DBRT modalities in vaginal cuff irradiation could significantly reduce the rectum and bladder doses without sacrificing the target dose. In a similar study by Onal et al. [17], it was found that 43% of the patients treated with a cylinder applicator had air gaps between the applicator surface and vaginal mucosa that caused dose uncertainty. It was also reported that 6.3% of these patients did not take the prescribed dose when the dose prescription was made to the depth of 5 mm from the applicator surface.
In the last two decades the developments in imaging, pathology, surgery, radiotherapy and systemic treatment have given rise to early diagnosis and better delineation of the disease, which led to effective treatment with high cure rates in patients with gynecological cancer. It is becoming increasingly important to reduce the treatment-related side effects in these patients with long survival expectancy. Rectum, bladder and sigmoid are the major dose-limiting organs in the treatment of gynecologic malignancies [2,12,15,18], particularly in patients with locally advanced disease when there is a need for combining intracavitary BRT with EBRT due to the large tumor volume. In order to overcome the problems in gynecological BRT, MC applicators with different designs and trademarks, such as Capri, which is a trademark of Varian Medical Systems, Inc. have been introduced by the manufacturers. Iftimia et al. [12], Demanes et al. [13] and Tanderup et al. [14] reported that MC applicators had dosimetric advantages in terms of target coverage and critical organ doses compared with CSC applicators. Additionally, Iftimia et al. [12], Mendez et al. [19] and Shin et al. [20] reported that MC applicators may be used instead of interstitial BRT in small lesions up to 1 cm in depth. However, in the case of increased tumor size and depth, existing MC applicators lose their effectiveness. Although interstitial BRT has a great advantage in selected patients, the necessity of operating room conditions during application and a postoperative care unit after treatment are the major disadvantages. In this study we proposed a novel compensator-based IMBRT applicator system to eliminate these disadvantages in selected patient groups. One of the most important advantages of this applicator is that the special design of the applicator allows different combinations of the source transfer channels and desired lengths of compensator materials during application and treatment. Additionally, this novel applicator system will allow a simultaneous integrated boost dose to the tumor or irradiation of more than two different treatment plans consecutively in the same treatment fraction without changing the position of the external part of the applicator. With this aspect treatment plans created with the IMBRT applicator can be an alternative to interstitial BRT even at a depth of more than 1 cm. In addition to all these advantages the invasive procedure required by the interstitial BRT can be eliminated in a selected group of patients and treatment comfort can be increased without compromising the quality of the treatment planning.
3D printers have been widely used in the production of compensator materials for proton therapy [21,22], bolus [23,24,25,26,27], QC phantom for EBRT [28,29,30,31,32], applicators and auxiliary equipment for BRT facilities [33,34,35,36,37,38,39,40]. Ju et al. [21] and Zou et al. [22] stated that the compensators produced with PLA material had similar dosimetric and physical properties with conventional compensators used in proton therapy. In EBRT facilities on the other hand, Park et al. [22] showed that a patient-specific bolus could be produced using 3D printer technology in patients with an irregular body contour. In that way it was emphasized that daily setup uncertainty and dose differences due to the bolus placement could be minimized using a 3D printer-based patient-specific bolus. Ehler et al. [32] reported that patient-specific QC phantoms could also be printed for RT facilities. In addition, similar to our study, Harris et al. [33] and Jones et al. [34] showed that a low-cost and patient-specific mould BRT applicator could be manufactured using a 3D printer. In another study Sethi et al. [36] evaluated treatment plans of three different patients and emphasized that the limited size of commercially available applicators could be eliminated by using 3D printing technology and effectiveness of the treatment plans could be increased by producing a patient-specific applicator. Additionally, Cunha et al. [35] and Sethi et al. [36] produced a cylindrical applicator for vaginal cuff applications using 3D printer-based technology.
In 3D printers it is possible to use different filament materials such as ABS, PLA, polycarbonate-ISO (PC-ISO), nylon, PET, carbon fiber, aluminum and wood according to the printer model and the area of use. For medical purposes printing material can be selected depending on several characteristics of the filament including sterilization, biocompatibility, non-toxicity, flexibility and durability. In the production of the BRT applicator, materials should be tissue equivalent for TG-43 formalism, sterilized, biocompatible, non-toxic and with strength for clinical use. Cunha et al. [35] used PC-ISO filament with a density of 1.2 g/cm3 in the production of the applicator and showed that the measurements were compatible with TPS. Athanasiou et al. [41] and Rankin et al. [42] stated that PLA material is suitable for medical applications and ethylene oxide (EtO) and glutaraldehyde solution can be used for sterilization. In our study PLA was used as a filament material with a density of 1.25 g/cm3 and we also found that measurements taken in homogeneous medium were consistent with TPS for PLA material.
There are several limitations of our study. The first one is that the calculation algorithm used in the TPS was based on TG-43 formalism. We could not take into account the heterogeneity and could not analyze the effects of compensator materials on treatment planning. The second limitation is that this study was only focused on mechanical and dosimetric feasibility of the novel IMBRT applicator without any chance to test it in patients. In the next study two sets of measurements will be performed to eliminate these limitations. In the first step simulation plans with heterogeneity correction will be created in a QC phantom and verification of the treatment planning will be performed using appropriate dosimetry systems such as EBT3 Gafchromic film. In the second step simulation plans will be created based on real clinical cases and a prospective clinical study will be designed to analyze the effectiveness of the system in suitable patient groups.
Conclusions
This novel IMBRT applicator has met the necessary requirements in mechanical and dosimetric QC tests in homogeneous medium. In the next stage simulation plans with heterogeneity correction will be created in a QC phantom and a prospective trial will be planned in appropriate patient groups so that the clinical effectiveness of the applicator can be evaluated.