Purpose
Uveal melanoma is the most frequent intraocular tumour in adults [1,2]. It originates from pigment cells of the iris, the ciliary body and choroid undergoing tumour transformation [3]. The prevalence of uveal melanoma depends on race, sex and latitude. It is estimated that each year in the USA 4.3 cases per 1,000,000 inhabitants are diagnosed, whilst in Europe from 2 to 8 are diagnosed [4,5]. There are no specific data concerning the Polish population, yet it is anticipated that each year 200-300 new cases of uveal melanoma are diagnosed [6].
Intraocular melanomas develop usually unilaterally in the choroid tissue (85%). The incidence of this tumour in the ciliary body (9%) and the iris (6%) is much lower [7,8].
The five-year mortality in the case of large uveal melanomas is 53%, medium-sized 32%, whilst in the case of small ones it is 16% [9]. The risk factors which decrease the chances of survival are a larger size of tumour, vascularisation forming a characteristic loop, tumour location (at the optic disc and/or macula, involvement of the ciliary body) as well as intraocular proliferation [10]. A relapse of the tumour is also a bad prognostic factor. A relapse is defined as recurrent, local tumour growth after remission obtained as a result of treatment.
Radiotherapy is the most frequently applied method of conservative treatment for uveal melanomas. Local treatment renders positive results with external beam radiation therapy (proton radiotherapy, Gamma knife) or brachytherapy applied at a higher frequency. This latter method, which has been used with good results for more than 50 years, consists in suturing, onto the surface of the sclera, applicators containing radioactive isotopes, e.g. iodine-125 (125I), ruthenium-106 (106Ru), iridium-192 (192Ir), palladium-103 (103Pd). Suturing the applicators with a radioactive element onto the surface of the sclera at the tumour base results in the supply of an appropriate dose of irradiation into the tumour tissue, whilst the irradiation of the surrounding healthy tissues is reduced.
Our Clinical Department of Ophthalmology and Ophthalmologic Oncology currently uses applicators containing 106Ru and 125I [11].
Radioactive ruthenium (106Ru) is used for the treatment of melanomas with a thickness not greater than 5 mm, whilst iodine (125I) is applied to larger tumours, whose thickness exceeds 5 mm. According to Shields and Shields in tumours with a thickness larger than 12 mm, and base diameter 15 mm, enucleation of the eyeball is indicated and conservative treatment is undertaken only when the patient does not consent to enucleation [2].
The purpose of this study was to perform a retrospective analysis of the factors which might affect the occurrence of a relapse of uveal melanoma after 125I brachytherapy treatment in our own patients.
Material and methods
The retrospective analysis concerned 343 patients treated for uveal melanoma with 125I brachytherapy at the our Clinical Department of Ophthalmology and Ocular Oncology in the years 2001-2012.
All patients under 18 years old were included in the study, in which indirect ophthalmoscopy and ultrasound examination were performed. As a result of it, uveal melanoma was recognized, which qualified them for 125I brachytherapy. For all the patients involved, the treatment with 125I was of the first choice. Patients with a follow-up shorter than 3 months and for whom this treatment was a second or a third therapy were excluded from the analysis.
The majority of the patients qualifying for this kind of treatment had the tumour thickness greater than 5 mm but no more than 12 mm.
The tumour apex dose was between 80 to 120 Gy and the application time varied from 24.5 to 193.6 hours.
Ophthalmologists skilled in plaque placement surgery, the radiation oncologist and medical physicists were involved in the treatment procedure, making up a multidisciplinary ocular tumour team. The course of the treatment and follow-up were very similar to that described by Tagliaferri et al. [12].
The differences consisted in the application of ready-made radioactive plaques purchased from Bebig (including 14 seeds of 125I) and without applying dummy plaques and intraoperative ultrasound examinations.
Due to the organizational and economic circumstances there was no possibility to prepare patient dedicated applicators.
The clinical evaluation included the sex and age of the patient, tumour size (T stage in the TNM classification – version 7), its shape, pigmentation, location, the presence of orange pigment or petechiae on the tumour surface, retinal detachment, blood or dispersed pigment in the vitreous body [13].
The above clinical characteristics were read from specially prepared questionnaires, which each time are filled in for all the patients with intraocular tumour. The questionnaires have been used in our clinic since 1998.
According to the questionnaire, the tumour colour can be amelanotic, medium-pigmented or brown (dark-pigmented). The presence of orange pigment or petechiae on the tumour surface, retinal detachment, blood or dispersed pigment in the vitreous body are evaluated in two categories: PRESENT or ABSENT. The assessments are made in every case by an experienced ocular ophthalmologist.
Additionally, the physical properties of the brachytherapy were analysed, such as total dose, the irradiation dose rate applied to the tumour top and base as well as irradiation time. The perioperative location of the tumour base was also evaluated in a diaphanoscopic examination (transillumination) before suturing the applicator.
Two groups of patients were distinguished: those with and without a relapse of the uveal melanoma. The diagnostic criterion for the relapse was growth of the tumour base or tumour height by 0.5 mm.
Statistical analysis was performed using Statistica v.10 StatSoft Polska software. In all calculations, the adopted level of statistical significance was α = 0.05. In the research, for quantitative variables, the basic statistics were calculated such as the mean, standard deviation, median, interquartile range, minimum, and maximum values. For verification of correlations between studied variables, the chi-square test or the accurate Fisher test were performed. Local treatment failure and mortality were calculated by the Kaplan-Meier actuarial method.
The authors declare that this investigation was carried out following the rules of the Declaration of Helsinki from 1975 (revised in 2008).
Results
Sample characteristics
The study group comprised 176 women (51.3%) and 167 men (48.7%), aged from 24 to 89 years (mean age 60.4, SD = 13.6). The observation period ranged from 3 to 144 months (Me = 45, Q1 = 25, Q3 = 76.75).
Among the patients, a dome-shaped melanoma was most prevalent in 215 patients (62.7%), and a mushroom-like one was found in 128 subjects (37.3%), whereas no cases with a flat tumour were observed. The most frequent type of pigmentation was a medium one, in 174 subjects (50.7%), and brown melanomas were found in 91 patients (26.5%), whilst amelanotic ones were found in 78 patients (22.7%).
In the group under analysis, tumours qualified as T3 were dominant (n = 235; 68.5%), T2 tumours occurred much more rarely (n = 86; 25.0%), with only sporadic incidence of T4 tumours (n = 19; 5.5%) and T1 (n = 3; 0.9%).
The average irradiation dose applied to the tumour top was 99.8 Gy (SD = 10.6; min = 80, max = 120), whilst the average dose applied to the tumour base (to the sclera) was 345.0 Gy (SD = 116.1; min = 83.4, max = 701.2). The average irradiation dose rate applied to the tumour top was 144.1 cGy/h (SD = 61.4; min = 41.3, max = 437.4), and to the base: 483.3 cGy/h (SD = 218.7; min = 83.8, max = 1047.0). The exposure duration varied from 24.5 to 193.6 hours with the mean exposure time being 82.0 hours (SD = 34.4).
In all patients an intraoperative diaphanoscopic examination (transillumination) allowed the tumour base to be located.
Other data concerning the study group are presented in Table 1.
Table 1
Characteristics of study group and relapse group
The relapse of uveal melanoma
A relapse occurred in 29 subjects (8.5%), including 13 women (7.3% of studied women) and in 16 men (9.6% of studied men) treated for uvea melanoma with 125I brachytherapy (Figure 1). No differences in local recurrence with regards to sex were found (p = 0.465).
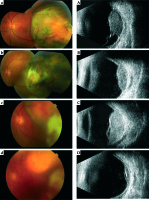
Fig. 1
A patient with local relapse: condition before treatment (a – fundus, A – ultrasound), positive result after 12 months (b – fundus, B – ultrasound), relapse after 23 months (c – fundus, C – ultrasound) and uveal scar after second treatment (d – fundus, D – ultrasound)
The mean age of patients with relapse was 58.5 years (SD = 15.5), with the youngest patient being 24 years old and the oldest 82 years old. In the group of patients without recurrence, the mean age was 60.6 years (SD = 13.4), with the youngest patient being 25 years old and the oldest 89 years old. The analysis revealed that the age of the patients did not affect the increase in the probability of relapse (p = 0.441).
The mean time to relapse was 29.6 months (SD = 4.2; min = 21.4, max = 37.8).
The most common increase in tumour height was noticed, and very rarely an extraocular extension was observed (Table 2).
Table 2
Recurrence type (N = 29)
| Recurrence type | n (%) |
|---|---|
| Margin | 3 (10.35) |
| Thickness | 16 (55.17) |
| Margin + Thickness | 0 (0.00) |
| Extraocular extension | 10 (34.48) |
In the patients with a dome-shaped tumour, recurrence was more frequent than in the case of the patients with a mushroom-like shape of tumour seen in the ultrasound: n = 23 (10.7%) vs. n = 6 (4.7%) respectively (p = 0.053).
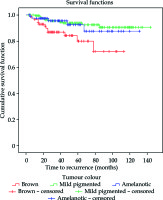
An analysis of the pigmentation showed a tendency for a more frequent relapse in brown melanomas (n = 13; 14.3%) in comparison with the tumours with moderate pigmentation (n = 11; 6.3%) or amelanotic ones (n = 5; 6.4%).
In patients with a relapse, the most frequent tumour stage was T3 (n = 22; 75.9%), followed by T2 (n = 5; 17.2%), T4 (n = 1; 3.4%), and T1 (n = 1; 3.4%) (p = 0.276). An analysis of the height and size of the tumour base as a risk factor for recurrence showed that in tumours with a height exceeding 10 mm or the bases larger than 15 mm, recurrences are more frequent (p = 0.082 and p = 0.232 respectively).
The mean radiation dose applied to the tumour top in the patients with relapse was 101.16 Gy (SD = 9.61 Gy), whilst in the group without relapse it was 99.70 Gy (SD = 10.74 Gy) (p = 0.481).
The mean radiation dose applied to the tumour base, i.e. the sclera, in the patients with relapse was 381.31 Gy (SD = 131.94), and in the group without relapse it was 341.66 Gy (SD = 114.15 Gy) (p = 0.090).
The mean radiation dose rate applied to the tumour top in the patients with relapse was 123.95 Gy/h (SD = 51.33 Gy/h), whilst in the patients without relapse it was 146.02 Gy/h (SD = 61.99 Gy/h) (p = 0.058).
The mean radiation dose rate applied to the tumour base in the patients with relapse was 457.92 cGy/h (SD = 216.26 cGy/h), whilst in the patients without relapse it was 485.63 cGy/h (SD = 219; 11 cGy/h) (p = 0.564).
The irradiation exposure time in patients with a relapse of the proliferative process was 95.63 h on average (SD = 39.11 h), whereas in the patients from the comparison group it was shorter: 80.75 hours (SD = 33.69 h) (p = 0.05).
The mean activity of plaque in the patients with relapse was 115.87 MCi (SD = 59.17 MCi), and in the patients without relapse it was 126.59 MCi (SD = 56; 40 MCi) (p = 0.392).
A relapse of uveal melanoma occurred significantly more frequently (p < 0.001) when the anterior edge of the tumour involved the ciliary body in comparison with all other locations. In the patient group with relapse 69.0% (n = 20) of tumours involved the ciliary body, in comparison with the control group, in which this percentage was much lower, 32.5% (n = 102).
The analysis of the presence of orange pigment at the moment of diagnosis showed a lack of statistically significant difference between the two groups – with relapse and without relapse.
Other factors examined in the study group, such as the presence of exudative retinal detachment, petechiae on the surface of the tumour or bleeding and dispersed pigment in the vitreous body did not have any effect on the increase of risk of local relapse (Table 3).
Table 3
Comparison of study group and relapse group
An increased frequency of distant metastases was not observed in the patients with a local relapse (p = 0.714).
Survival and relapse
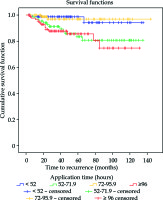
In the group under analysis, survival depending on the time of relapse was statistically significant for application time (p = 0.004) and tumour pigmentation (p = 0.010). Deaths among patients with a local relapse were least frequent when the duration of the brachytherapy was between 72 and 95.9 hours (Figure 2), whereas they were most frequent in cases of brown tumour pigmentation (Figure 3). The characteristics of clinical features and radiation parameters for the group with the duration of treatment between 72 and 95.9 hours are presented in Table 4.
Table 4
Characteristics of the group with duration of brachytherapy between 72 and 95.9 hours
Other factors such as the sex of the patient (p = 0.299), age (p = 0.667), the shape of the tumour observed in ultrasound (p = 0.061), the size of the tumour – T stage (p = 0.575), the irradiation dose applied to the tumour top (p = 0.380) and to the sclera (p = 0.223), the dose rate applied to the top of the tumour (p = 0.057) and sclera (p = 0.940) and plaque activity (p = 0.811) did not have any effect on survival depending on the occurrence of relapse.
Discussion
A positive local result of the treatment of intraocular melanoma predicts a better prognosis. The occurrence of a local relapse means a higher probability of systemic dissemination and death. According to the Ophthalmic Oncology Task Force, there is a significant difference in 5-year and 10-year survival after treatment of uveal melanoma between groups of patients with relapse and without relapse (p < 0.001), making up 71% and 62% as opposed to 87% and 82% respectively [14].
Therefore, it is extremely important to find the factors which might contribute to the occurrence of a local relapse. In 2002, a report prepared by the Collaborative Ocular Melanoma Study (COMS) evaluated the frequency of local relapse after treatment with 125I to be 10.3% [15]. In the studies carried out by other authors, the frequency of relapse after 125I brachytherapy was 4.5% according to Bellerive et al., 9% according to Perez et al., 4.7% according to Vonk et al. and 5.1% according to Sánchez-Tabernero et al. [16,17,18,19]. A meta-analysis of 49 papers (a group of 12,524 patients) showed that the risk of local recurrence of uveal melanoma treated with 125I varies between 4.0% and 9.6% [20].
In our analysis local relapse was found in 29 patients, i.e. in 8.5% cases. This result was consistent with data presented by other researchers.
We did not observe differences between the groups with relapse and without relapse with regards to the sex (p = 0.465) or age of the patients (p = 0.441). In none of the above quoted papers was a significant effect of the sex and age of the patients on the occurrence of local relapse found with the exception of the work by Bellerive et al. in which an older age predisposed to a local relapse of the intraocular tumour [16].
In the patients with dome-shaped tumours the relapse was more frequent than in those in whose case a mushroom-like shape was found in the ultrasound (p = 0.053). We are not aware of papers which confirm an increased risk of local recurrence depending on the shape of the tumour after the use of 125I brachytherapy. The work by Miguel et al. evaluating 20-year results of the treatment of uveal melanoma observed that the necessity of enucleation of an eyeball occurs significantly more frequently in the case of dome-shaped tumours than in the case of a mushroom-like shape (p = 0.002). These authors however did not find any correlation between the shape of the tumour and an increased risk of local relapse [21].
No significant correlation was found between the size of the tumour, defined as its T stage according to the American Joint Committee on Cancer (AJCC), and the predisposition to develop a relapse, a fact which is consistent with other reports [14,19].
A local relapse may occur in various periods after the completion of treatment – in the research of the Ophthalmic Oncology Task Force this period varied between 1 month and 9.8 years [14]. In our study, the relapse was observed on average after 29.6 months with the shortest period between the treatment and the relapse being 21.4 months (SD = 4.2; min = 21.4, max = 37.8).
In our study group, no increased frequency of metastases was found in patients in whose case a local relapse occurred (p = 0.714). In spite of this, many authors stress a significant increase in the tendency to develop metastases in cases of local relapse of uveal melanoma [14,19].
This difference might be due to the comparatively low number of patients in our case (n = 29) in contrast with groups analysed by other authors [14,19].
The mean dose applied to the top of the tumour did not have any effect on the occurrence of local relapse in our patients. Some similar observations were made by Echegaray et al. in a meta-analysis of 15 reports in which they observed a tendency of local relapse to decrease by 0.14% with a dose increase by 1 Gy to the top of the tumour, yet this result was not statistically significant (p = 0.336) [22].
Wagner et al. did not find any correlation either between the dose applied to the top and base of the tumour and the probability of the occurrence of relapse. They also observed that these two parameters affect the occurrence of complications after 125I treatment [23].
One of the factors which, in our study, had an important effect on the occurrence of a local relapse (p < 0.001) was the involvement of the ciliary body. Kaliki and Shields confirm that melanomas which develop in the ciliary body or with secondary involvement of the ciliary body are more aggressive in their course than those which extend to the iris or choroid [1]. Perhaps this observation is related to the fact that on account of their location, the melanomas of the ciliary body are diagnosed later and therefore have larger dimensions of the base and top, which, in turn increases the risk of a local recurrence [1].
Miguel et al. noted that persistent or recurrent retinal detachment after radiotherapy can be a sign of disease activity and even constitutes a prognostic factor for failure of local control and enucleation [24]. In our study we did not observe any connection between a retinal detachment and a tumour relapse.
An analysis of the survival of patients with a relapse allowed us to observe that an application time ranging between 3 (72 h) and 4 days (95.9 h) decreased the risk of death (p = 0.004), as opposed to brown pigmentation of the tumour (p = 0.010), which meant larger mortality.
The time of irradiation of uveal melanoma using 125I, as recommended by the Ophthalmic Oncology Task Force, is 5-7 days and for 106Ru: 3-7 days [25]. However, Quivey et al. in their analysis of the influence of the time and dose on local failure of 125I brachytherapy, observed that a longer application time indicates a significantly higher probability of relapse (p ≤ 0.0001), with a mean irradiation time of 124.7 h (5.2 days) within a range of 28 to 333 hours [25]. In our study, the mean treatment time was 82.0 h within a range from 24.5 to 193.6 hours. In the case of the patients with a relapse, it was longer (95.63 h) than in the group without relapse (80.75 h) (p = 0.05), a fact which might confirm the observations made by Quivey et al. Quivey et al. noted that the mean dose rate at the tumour apex was 98 cGy per hour for the nonrecurrent group while it was 76 cGy per hour for the recurrent patients; the overall treatment time was 4.8 days and 6.4 days in these groups, respectively [26]. We observed the same dependency: the mean radiation dose rate applied to the tumour top in the patients with relapse was lower (123.95 Gy/h), whilst in the patients without relapse it was higher (146.02 Gy/h) (p = 0.058). The impact of the application time on relapse occurrence requires further research. The best option, in our opinion, is conducting a study with animals models or cell cultures, where the only changing factor will be the application time.
Several reports have indicated that in uveal melanoma, the risk of metastases and death increases with increased tumour pigmentation [27,28]. Another current reports suggests a better prognosis in the case of uveal melanomas with less pigmentation, including amelanotic ones [29,30]. The theory which might explain this could be a reverse correlation between the melanin and vitamin D receptor (VDR) levels in uveal melanoma. In predominantly amelanotic tumours, the levels of VDR were the highest, then VDR decreased in medium-pigmented and dark brown uveal melanomas [31]. Similar observations were made in the studies on cutaneous melanoma, where the level of VDR decreased together with an increase of melanin content and poorer prognosis [32,33].
In the above context, the loss of the protective function of vitamin D could be secondary to the reduction of the VDR expression by increased melanogenesis.
Important functions of the active form of vitamin D are its anti-tumour effects, antimutagenic properties (e.g. protection against the effect of free radicals) and the regulation of tumour proliferation, apoptosis, and angiogenesis [31,34,35].
It was shown that vitamin D deficiency or dysregulated vitamin D signaling can play an important role in oncogenesis, clinical advancement and prognosis in such neoplasms as cutaneous melanomas, bladder, breast, lung, ovarian, pancreatic, thyroid, prostate and colorectal cancers [31].
A local recurrence may be related to such factors as the displacement of an applicator, tumour location on the optic disc of the 2nd nerve or close to it, resistance to irradiation, melanoma histopathological type or the genotype of its cells. Therefore, it is necessary to improve methods for the location and confirmation of the correct suturing of an applicator such as perioperative ultrasound, and, with the locations in the area of the disc, completion of the therapy with transpupillary thermotherapy (TTT) [21,24,36,37].
There are ongoing studies concerning the genetic and epigenetic background of uveal melanoma which might allow for the introduction of new factors sensitizing the tumour cells to irradiation.
The treatment of uveal melanomas with applicators containing 125I allows for a high rate of positive local results and in the majority of cases constitutes the treatment of first choice in the case of tumours with a height ranging from 5 to 12 mm and with the largest base up to 15 mm.
Conclusions
The treatment of uveal melanomas with applicators containing 125I allows for a high rate of positive local results. However, in the process of treatment we should always take into consideration the possibility of a local relapse. In our study, the predisposing factors for a local relapse, which might also increase the probability of death in the case of recurrence, comprised the involvement of the ciliary body. The survival depending on the time of relapse could be related to application time and tumour pigmentation. Deaths among patients with a local relapse could be least frequent when the duration of the brachytherapy is between 72 and 95.9 hours, whereas they could be most frequent in cases of brown tumour pigmentation.
We recommend conducting further studies to evaluate the influence of the tumour pigmentation and the application time on local recurrence of uveal melanoma after 125I brachytherapy. A randomized study would be the best option for such a purpose.