Purpose
Prostate cancer (PCA) is the second most common cause of new cancer diagnoses, and the fifth most common cause of cancer-specific deaths among men worldwide [1]. Similar results have been reported in Japanese men [2].
Iodine-125 (125I) low-dose-rate brachytherapy (LDB) is a well-established treatment for clinically localized PCA. LDB can be performed with a short hospitalization period, and is convenient for early return to daily life and social activities. LDB is an effective treatment for localized PCA not only in low-risk, but also in intermediate- and high-risk patients [3, 4]. Oncological outcomes, adverse events, and treatment-related changes in quality of life (QoL) are important factors in treatment decision-making. Many investigators have reported that clinically localized PCA treated with LDB showed favorable oncological outcomes [4, 5], less severe toxicity [6], and less negative impact on long-term QoL [7, 8].
Our institution, a core hospital for regional medical care, provides a wide range of treatments for urological malignancies, and performs LDB in about 50 cases per year, one of the highest numbers in Japan. We have performed LDB with or without external beam radiotherapy (EBRT) for about 400 patients with localized PCA since March 2007. Clinical outcomes of LDB are not widely reported in Asia, including Japan. Therefore, it is meaningful to report the clinical outcomes at our institution, which has a relatively large number of patients. Previously, we reported on QoL after LDB for localized PCA [9].
The purpose of this study was to retrospectively evaluate the oncological outcomes and genitourinary (GU) and gastrointestinal (GI) adverse events in acute and late phases of LDB with and without EBRT for localized PCA in a single Japanese institution.
Material and methods
Patients
Three hundred and fifty-two patients were treated with LDB for clinically localized PCA between March 2007 and August 2018 at the authors’ University Hospital. In this retrospective study, patients with a follow-up period of less than 24 months and those whose longitudinal prostate-specific antigen (PSA) levels or adverse events could not be assessed were excluded. Based on the exclusion criteria, 18 patients (5.1%) were disqualified, and 334 patients (94.9%) were eligible for this study.
Treatments
Patients were classified into risk groups according to the National Comprehensive Cancer Network (NCCN) risk criteria [10]: low-risk was defined as clinical T-stage T1-2a, PSA level < 10 ng/ml, and a Gleason score (GS) of 6 (GS6); intermediate-risk was described as clinical T-stage T2b-c, PSA level 10-20 ng/ml, and GS7; and high-risk was defined as clinical T-stage T3a, PSA > 20 ng/ml, and GS8-10. Patients in low- and intermediate-risk groups with a GS of 3 + 4 and a biopsy-positive core rate < 33% received LDB monotherapy. In contrast, patients in the remaining intermediate-risk group obtained additional doses of EBRT. Patients in the high-risk group received LDB, EBRT, neoadjuvant hormone therapy (NAHT), and adjuvant hormone therapy (AHT) for nine months from pre-treatment to post-treatment. These patients received androgen deprivation therapy and/or anti-androgens, such as bicalutamide, as hormone therapy.
Pathological diagnoses were established by a qualified pathologist in our institution. We developed a treatment plan three weeks prior to LDB to confirm prostate volume and determine number of seeds to be implanted. NAHT was administered for 3 months in patients with a prostate volume > 40 ml, or trimodality or at the discretion of the attending urologist.
All implantations were performed using 125I loose seeds and a Mick applicator (Mick Radio-Nuclear Instruments Inc., NY, USA), and were based on interactive planning and modified peripheral loading methods. Treatment plans for the initial 184 patients were generated using an interplant software (CMS, St. Louis, MI, USA), whereas that for the latter 150 patients, VariSeed was used (Varian Medical Systems, Palo Alto, CA, USA). Prescribed dose was 145 Gy for LDB monotherapy and 110 Gy for LDB and EBRT combination therapy, followed by an additional EBRT of 45 Gy. Dose-volume histograms for the prostate, urethra, and rectum were constructed to determine minimal dose irradiating 90% of the prostate volume (D90), volume of the prostate receiving 100% (V100) and 150% (V150) of the prescribed dose, minimal dose received by 5% (UD5) and 30% (UD30) of the urethra, and percentage of rectal volume that received 100% of the prescribed dose (RV100).
Intra-operative dosimetric parameters for both LDB monotherapy and LDB and EBRT combination therapy were as follows: prostate V100 > 95%, prostate D90 > 100%, and < 130% of the prescribed dose, prostate V150 < 60%, and rectal V100 < 1.0 cc. UD30 was set at < 220 Gy and < 160 Gy for LDB monotherapy and LDB and EBRT combination therapy, respectively. UD5 was set at < 240 Gy for LDB monotherapy, but not for LDB and EBRT combination therapy. Post-implant dosimetric analysis was performed using computed tomography and magnetic resonance imaging conducted 4-5 weeks after LDB.
Patients were generally discharged two days after implantation. Most patients were prescribed α-blockers (e.g., tamsulosin, silodosin, or naftopidil) or a phosphodiesterase-5 inhibitor (tadalafil), whereas some patients did not receive any medication. Both α-blockers and phosphodiesterase-5 inhibitor were continued for a minimum of approximately one month until post-implant dosimetric analysis, after which they were continued, modified, or discontinued depending on urinary symptoms. EBRT was performed at 6-8 weeks after implantation using intensity-modulated radiation therapy (IMRT), with a total dose of 45 Gy/25 fractions. IMRT radiation field covered the prostate and seminal vesicles.
Follow-up and outcome measurements
Baseline patient characteristics, treatment-related factors, and dosimetry factors were collected from medical records. Post-treatment follow-up was done every three months for the first two years, every six months for the next five years, and every year for the next ten years. Follow-up assessments were performed with blood tests, including PSA, physical examination, QoL assessment, and adverse event assessment. Patient comorbidities were assessed using age-adjusted Charlson comorbidity index (ACCI) [11].
Incidences of acute and late genitourinary (GU) and gastrointestinal (GI) adverse events were determined using National Cancer Institute common terminology criteria for adverse events, version 5 (NCI CTCAE v.5.0) [12]. GU adverse events included urinary frequency, painful urination, hematuria, and urinary retention. GI adverse events consisted of anal pain and rectal bleeding. Acute and late-phase adverse events were defined as symptoms occurring within three months and after 12 months after LDB, respectively. International prostate symptom score (IPSS) was used to assess lower urinary tract symptoms. In addition to the total IPSS, voiding score, storage score, and QoL index were also evaluated [13]. Disease-specific health-related QoL was assessed with University of California Los Angeles prostate cancer index (UCLA-PCI), which consists of 20 questions assessing urinary function, urinary bother, bowel function, bowel bother, sexual function, and sexual bother [14].
Statistical analysis
Baseline characteristics, treatment parameters, and dosimetry factors were compared between the two groups (monotherapy and EBRT combination therapy) using Mann-Whitney U test, χ2 test, and Fisher exact test, as appropriate. Shapiro-Wilk test was applied to evaluate the normality of distribution of continuous variables. All data were expressed as median (range). Kaplan-Meier method was used to evaluate the bio-chemical relapse-free survival (bRFS), cause-specific survival (CSS), and overall survival (OS). bRFS was defined according to Phoenix definition [15]. Differences between Kaplan-Meier curves were examined using log-rank test. Univariate and multivariate logistic regression analyses were performed to examine independent predictors associated with grade ≥ 1 GU and GI adverse events in acute and late phases. For multivariate analysis, age, EBRT, NAHT, AHT, and factors with a p-value ≤ 0.1 in univariate analysis were selected as variables. All statistical analyses were performed using JMP version 15 (SAS Institute Inc., Cary, NC, USA). All tests were two-sided, and a p < 0.05 was considered statistically significant.
This retrospective study was approved by the authors’ affiliated institution. Study protocol conformed to provisions of Declaration of Helsinki (as revised in Fortaleza, Brazil, October 2013). Informed consent was obtained from all patients involved in this study.
Results
Table 1 shows baseline characteristics and treatment parameters of the 334 patients. The median age of the overall cohort was 68 (50-83) years. The low-, intermediate-, and high-risk groups consisted of 133 (39.8%), 163 (48.8%), and 38 (11.3%) patients, respectively. The median follow-up period was 71 (range, 24-156) months. There was no significant difference in baseline IPSS severity and UCLA-PCI urinary function between the LDB monotherapy and the EBRT combination therapy groups. In contrast, the monotherapy group had a significantly higher UCLA-PCI bowel function than the EBRT group. The EBRT combination group was significantly older and had a higher ACCI than the LDB monotherapy group.
Table 1
Patients’ characteristics at baseline
[i] LDB – low-dose-rate brachytherapy, BMI – body mass index, PSA – prostate-specific antigen, NCCN – National Comprehensive Cancer Network, EBRT – external beam radiotherapy, IPSS – international prostate symptom score, UCLA-PCI – University of California Los Angeles prostate cancer index, PDE5i – phosphodiesterase- 5-inhibitor, ACCI – age-adjusted Charlson comorbidity index
Regarding treatment parameters, neoadjuvant and adjuvant hormone therapy was administered to 145 (43.4%) and 38 (11.4%) patients, respectively. All adjuvant hormone therapies were administered in the EBRT combination group. The monotherapy group had a significantly higher use of α1-blockers (71.7% vs. 41.5%, p < 0.0001) than the EBRT group.
Post-implant dosimetric data at 4-5 weeks after LDB are shown in Table 2. Regarding dosimetry factors, the median V100, V150, and D90 were 96.0%, 69.9%, and 164.1 Gy, respectively. The median UD5 and UD30 were 225.8 Gy and 203.1 Gy, respectively. The median RV100 was 0.15 ml. The monotherapy group had significantly higher V100 (96.4% vs. 94.9%, p < 0.0001), V150 (71.6% vs. 66.8%, p = 0.0001), D90 (171.6 Gy vs. 125.4 Gy, p < 0.0001), UD5 (237.5 Gy vs. 186.9 Gy, p < 0.0001), and UD30 (217.1 Gy vs. 159.8 Gy, p < 0.0001) than the EBRT group. Meanwhile, there was no significant difference in RV100 between the two treatment groups.
Table 2
Statistics for dosimetric data
[i] V100 – prostate volume receiving 100% of prescribed minimal dose, V150 – prostate volume receiving 150% of prescribed minimal dose, D90 – minimal dose received by 90% of prostate, UD5 – minimal dose received by 5% of urethra, UD30 – minimal dose received by 30% of urethra, RV100 – volume of rectum receiving 100% of prescribed dose
Survival analysis
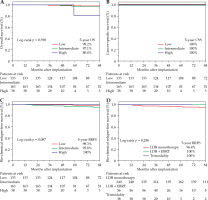
The 5-year OS rates for the low-, intermediate-, and high-risk groups were 99.2%, 97.1%, and 80.0%, respectively (p = 0.398, Fig. 1A). The cause of all deaths was other than PCA. The 5-year CSS rate for all risk groups was 100% (Fig. 1B). The 5-year bRFS rates for the low-, intermediate-, and high-risk groups were 98.3%, 95.8%, and 100%, respectively (p = 0.087, Fig. 1C). The 5-year bRFS rates for the LDB monotherapy, EBRT combination, and trimodality groups were 96.4%, 100%, and 100%, respectively (p = 0.262, Fig. 1D).
Fig. 1
Oncologic outcomes. Kaplan-Meier curves for A) OS, B) CSS, C) bRFS by NCCN risk, and D) treatment type. Percentage survival estimates (number at risk) at 5 years after implantation. Differences between Kaplan-Meier curves were tested using log-rank test
OS – overall survival, CSS – cause-specific survival, bRFS – bio-chemical relapse-free survival, NCCN – National Comprehensive Cancer Network, LDB – iodine-125 low-dose-rate brachytherapy, EBRT – external beam radiotherapy
GU adverse events
Regarding acute GU adverse events in all patients, most patients had G1 and G2 GU adverse events: 159 (47.6%) and 63 (18.9%) patients, respectively. Only one patient (0.3%) had a ≥ G3 adverse event. Urinary frequency was the predominant acute GU adverse event. The EBRT combination therapy group had a significantly higher number of acute G1 GU adverse events than the LDB monotherapy group. With regards to frequency, the results were different between the two groups. The acute G1 frequency was significantly higher in the EBRT combination therapy group than in the LDB monotherapy group. In contrast, the acute G2 frequency was significantly higher in the LDB group. Regarding late GU adverse events, frequency and hematuria were predominant. There was no significant difference in late GU adverse events between the LDB monotherapy and the EBRT combination therapy groups (Table 3).
Table 3
Acute and late genitourinary toxicities
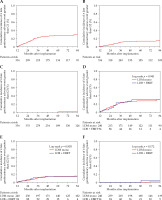
For late GU adverse events in all patients, the 5-year cumulative ≥ G1 and ≥ G2 adverse event rates were 27.9% and 14.4%, respectively (Fig. 2A, B). The incidence of ≥ G3 adverse events was only 0.5% (Fig. 2C). There was no significant difference in the 5-year cumulative ≥ G1, ≥ G2, and ≥ G3 late GU adverse events between the LDB monotherapy and the EBRT combination therapy groups (Fig. 2D-F).
Fig. 2
Cumulative incidence of late genitourinary adverse events greater than A, D) G1, B, E) G2, and C, F) G3
Multivariate logistic regression analysis identified high pre-treatment IPSS and non-use of α1-blockers as factors associated with ≥ G1 acute GU adverse events. No factor was associated with ≥ G1 late GU adverse events in multivariate logistic regression analysis (Table 4).
Table 4
Univariate and multivariate analysis of predictors associated with acute and late genitourinary toxicity greater than G1
[i] BMI – body mass index, PSA – prostate-specific antigen, NAHT – neoadjuvant hormone therapy, AHT – adjuvant hormone therapy, V100 – prostate volume receiving 100% of prescribed minimal dose, V150 – prostate volume receiving 150% of prescribed minimal dose, D90 – minimal dose received by 90% of prostate, UD5 – minimal dose received by 5% of urethra, UD30 – minimal dose received by 30% of urethra, EBRT – external beam radiotherapy, IPSS – international prostate symptom score, UCLA-PCI – University of California Los Angeles prostate cancer index, PDE5i – phosphodiesterase-5-inhibitor, ACCI – age-adjusted Charlson comorbidity index
GI adverse events
Regarding acute GI adverse events in all patients, most patients had G1 and G2 GI adverse events, including 11 (3.3%) and 5 (1.5%) patients, respectively. No patient had a ≥ G3 adverse event. The EBRT combination therapy group had a significantly higher rate of acute G1 and G2 GI adverse events compared to the LDB monotherapy group. The rates of acute GI adverse events, such as anal pain and rectal bleeding, were equal. The acute G1 and G2 anal pain were significantly higher in the EBRT combination therapy group than in the LDB monotherapy group.
With regards to late GI adverse events, rectal bleeding was predominant. There was no significant difference in late GU adverse events between the LDB monotherapy and the EBRT combination therapy groups (Table 5).
Table 5
Acute and late gastrointestinal adverse events
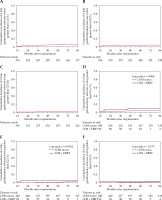
The 5-year cumulative ≥ G1, ≥ G2, and ≥ G3 late GI adverse event rates in all patients were 3.1%, 1.5%, and 0.5%, respectively (Fig. 3A-C). Rectal bleeding was the most frequent late GI adverse event. The EBRT combination therapy group had a significantly higher number of ≥ G2 late GI adverse events than the LDB monotherapy group. In contrast, there was no significant difference in the 5-year cumulative ≥ G1 and ≥ G3 late GI adverse events between the LDB monotherapy and the EBRT combination therapy groups (Fig. 3D-F).
Fig. 3
Cumulative incidence of late gastrointestinal adverse events greater than A, D) G1, B, E) G2, and C, F) G3
No factor was associated with ≥ G1 acute and late GI adverse events in multivariate logistic regression analysis (data not shown).
Discussion
In this study, the oncological outcomes of LDB in our institution were very good. Both GU and GI adverse event rates were acceptable, and the incidence of serious adverse events (≥ G3) was extremely low. A simple comparison of the present study with previous studies is difficult because of inconsistencies in the LDB procedure, hormone therapy, and adverse event evaluation periods as well as criteria, such as CTCAE and Radiation Therapy Oncology Group criteria. However, considering these factors, the results of this study are comparable to those of previous studies.
Several reports have reported favorable oncologic outcomes of LDB. The 5-7-year bRFS rates for low-, intermediate-, and high-risk groups have been reported to be 90.2-98.0%, 83.9-99.1%, and 70.3-88.2%, respectively [4, 5, 16, 17]. In this study, the oncological outcomes of LBD were comparable to these reports. Additionally, the bRFS by NCCN risk and by treatment were not significantly different according to log-rank test. Considering previous ABS recommendations [18], the results of dosimetric factors at our institution were favorable. In our institution, the quality of treatment was well-maintained through the use of recommended techniques and high-quality collaboration between the urologist and radiologist. Additionally, appropriate treatment options were selected for each patient.
In this study, both acute and late GU adverse events were nearly all cases of G1 and G2, suggesting that LDB could be performed safely. In previous studies, ≥ G1, ≥ G2, and ≥ G3 acute GU adverse event rates were 66.5-86.2%, 6.0-55.9%, and 0.1-4.3%, respectively [19-23]. These results are comparable to ours. Moreover, ≥ G1, ≥ G2, and ≥ G3 late GU adverse event rates were 49.5-66.3%, 12.4-30.3%, and 0.9-3.2%, respectively [20-23]. These results are also comparable to those of the present study.
Whether LDB monotherapy or EBRT combination therapy results in a higher incidence of adverse events is still controversial [6, 24]. In this study, no consistent results were obtained when comparing adverse events in the LDB monotherapy and the EBRT groups regarding GU. Other factors in addition to EBRT could influenced the GU events after LDB.
In this study, a higher pre-treatment IPSS and non-use of α1-blockers were identified as factors associated with ≥ G1 acute GU adverse events. A higher pre-treatment IPSS [19, 23], NAHT [23], greater number of needles [23], and higher prostate V100 [19] have been reported as factors associated with acute GU adverse events. Additionally, prophylactic efficacy of α1-blockers has been validated [25]. These reports seem to support our results. There are three α1 adrenoceptor sub-types, including α1A, α1B, and α1D [26]. Naftopidil, tamsulosin, and silodosin are α1A/α1D adrenoceptor antagonists, with different affinities for the α1 adrenoceptor sub-types. Previous reports investigating the comparative effects of naftopidil, tamsulosin, and silodosin against post-LDB GU suggested the benefits of silodosin [27]. However, other studies found no significant differences in efficacy between silodosin and naftopidil [28]. Therefore, further studies are required to determine a more effective α1 blocker and optimal duration of its’ administration.
No predictors of late GU adverse events were identified in this study. A higher pre-treatment IPSS [19, 23], EBRT [22], NAHT [23], a higher prostate V100 [19], a higher prostate V150 [23], and a prior acute GU adverse event [22, 23] have been reported as the predictors of late GU adverse events.
In this study, the incidences of both acute and late GI adverse events were extremely low. In previous studies, the ≥ G1, ≥ G2, and ≥ G3 acute GI adverse event rates were 7.8-27.7%, 0.5-9.6%, and 0.0%, respectively [20, 22]. Moreover, the ≥ G1, ≥ G2, and ≥ G3 late GI adverse event rates were 17.2-21.1%, 2.8-5.8%, and 0.0%, respectively [20, 22]. In comparison to these reports, our results were favorable regarding both acute and late GI adverse events. The ABS recommendation is that RV100 should ideally be < 1 cc to avoid GI adverse events. Other reports have also shown that RV100 is associated with the occurrence of GI adverse events [22]. Also, EBRT combination therapy has also been reported to be associated with GI adverse events [29]. In this study, the comparison between the LDB monotherapy and the EBRT combination therapy groups also suggested that EBRT may be associated with GI adverse events after LDB. Additionally, IMRT has been reported to result in fewer GI adverse events than three-dimensional conformal radiation therapy, when used in combination with brachytherapy [30]. In this study, the very low number of GI adverse events could be attributed to the fact that RV100 was maintained at a low level and that all EBRT cases were treated with IMRT. Additionally, because only a few patients developed ≥ G1 acute and late GI adverse events, no factor was associated with these adverse events.
This study has some limitations. First, this was a single-center retrospective study that included a limited patient number and a heterogeneous patients’ population. Second, the focus of this study was on identifying factors associated with ≥ G1 GU and GI adverse events. In contrast, most previous reports focused on ≥ G2 adverse events. Although CTCAE is a well-recognized physician assessment, patient-reported adverse events can be worse than physician-reported symptoms [31]. Therefore, with respect to prediction and prevention of adverse events, it is reasonable to focus on ≥ G1 adverse events. Third, the median follow-up duration was insufficient. PCA patients are expected to have long-term survival; therefore, further follow-up of oncological outcomes and treatment-related adverse events is required. Despite these limitations, we believe that our study supports the utility of LDB and may lead to a better understanding of the course of treatment by medical staff and patients.
Conclusions
In summary, the present study demonstrated that LDB results in favorable oncological outcomes. Both GU and GI adverse event rates were acceptable, and the incidence of serious adverse events (≥ G3) was extremely low. Our study suggests that pre-treatment urinary function and the use of α1-blockers may be useful in predicting and preventing acute GU adverse events.










