Purpose
Prostate cancer (PCa) is the most common type of cancer among men in Europe. Current recommendations for screening and diagnosis are based on prostate-specific antigen (PSA) levels and/or suspicious digital rectal examination (DRE) and/or imaging. Transrectal ultrasound-guided biopsy (TRUS-Bx) is now the standard of care. The contemporary random 10-12-core systematic biopsy strategy relies on sampling efficiency for cancer detection. Prostate biopsy is performed by either the transrectal or transperineal approach [1]. Although laterally directed cores within the peripheral zone increase detection, prostate cancers are frequently multifocal, small, intermingled with benign stroma, and not uniformly distributed within the gland. Consequently, clinically significant cancers frequently go undetected when employing the standard 12-core biopsy template [1]. Conventional TRUS-Bx also leads to incorrect risk stratification of tumors as low volume or low grade. These clinically insignificant cancers are often identified by chance during a systematic biopsy, contributing, in part, to the problem of over-detection and over-treatment of low-risk and indolent PCa [2].
Advances in multiparametric magnetic resonance imaging (mpMRI) have demonstrated an improvement in detection and characterization of clinically significant prostate cancer (csPCa). mpMRI is increasingly used to localize suspicious areas that could be targeted by magnetic resonance imaging-targeted biopsies (MRI-TBx) [3]. Introducing mpMRI and MRI targeted biopsy as modalities to evaluate men at risk for prostate cancer may help in determining more efficiently which men need a prostate biopsy. MRI targeted biopsy enables more accurate sample collection, thereby allowing greater detection of clinically significant disease with fewer biopsy cores, more accurate risk stratification, and avoidance of the detection of indolent disease [4,5]. MRI targeted biopsy enables a better risk stratification among men who are subjected to active surveillance.
Once the prostate cancer is diagnosed, it can be radically treated by surgery (radical prostatectomy) or by irradiation therapy according to available guidelines or patient preferences. The recent developments in the field of irradiation technology have made radiotherapy one of the main local treatment methods for the prostate cancer patients. Brachytherapy low-dose-rate (LDR) or high-dose-rate (HDR) is a standard primary treatment for low- or intermediate-risk PCa [6]. TRUS-Bx is routinely acquired for LDR prostate brachytherapy preplanning (pTRUS), although diagnostic mpMRI may serve this purpose as well [7]. Fredman et al. compared the predictive abilities of TRUS vs. MRI relative to intraoperative TRUS (iTRUS) to assess the role of mpMRI in brachytherapy preplanning. Shaaer et al. described the use of mpMRI during prostate HDR brachytherapy planning [8]. These works suggest that mpMRI carries numerous diagnostic, staging and preplanning advantages that facilitate better patient selection and delivery of novel dose escalation and targeted therapy, with no additional surgical or anesthesia time [7,8].
The group of patients who were diagnosed with the pre-biopsy mpMRI (PB-mpMRI) tool for the detection of PCa is presented in the article. The aim of this study was to assess the diagnostic value of PB-mpMRI among the group of patients referred for a primary and secondary biopsy.
Material and methods
In the period between November 2018 and May 2019, a group of 40 consecutive patients, selected in a single urological institution, who were considered for a prostate biopsy with a suspicion of PCa, was evaluated. The contrast-enhanced mpMRI test was performed to facilitate the decision to perform prostate biopsy among men with elevated PSA levels (> 4 ng/ml), abnormal DRE or family history of PCa. The men with previous negative biopsies (two or more biopsies before), among whom the suspicion of prostate cancer was still present, were qualified for another biopsy and the MRI-TBx procedure was performed. In areas of suspicion, identifying hidden diseases by the MRI-TBX procedure is advantageous. Radiology technicians performed mpMRI scans according to the standardized protocol. All men underwent 1.5 T (31 patients – 77.5%) or 3 T (9 patients – 22.5%) mpMRI. The evaluation of MRI data was carried out in accordance with the Prostate Imaging-Reporting and Data System (PI-RADS) v.2 and improved by a sectoral map. Three senior radiologists, with relevant experience in Prostate Imaging-Reporting and Data System v.2 (PI-RADS v.2) reporting, evaluated suspicious lesions using T2-weighted imaging, diffusion-weighted imaging (DWI) and dynamic contrast-enhanced MRI [9]. The radiologists had access to clinical information and assigned a PI-RADS v.2 suspicious result to each region of interest (ROI). The ROI found on the MRI scan diagram as depicted by the radiologist (suspicious lesions) was projected (cognitively) prior to the procedure. We could not evaluate inter-reader variability as two different readings were not available. PI-RADS > 3 were considered suspicious. Patients with mpMRI lesions (PI-RADS 4-5) underwent cognitive targeted (COG-Bx) plus systematic TRUS-Bx. We defined lesion-specific csPCa as Gleason score > 7 (3 + 4) [10]. The patients with PI-RADS 1-3 on mpMRI avoided immediate biopsy; they are under the standard care and supervision. The procedure was performed with prophylactic intravenous antimicrobial cover (ciprofloxacin). The patients were also instructed to continue oral antibiotics for another 4 days. We investigated the impact of introducing mpMRI on the diagnostic yield of transrectal prostate biopsy (TRUS-Bx).
Data preprocessing and statistical analyses were performed using Analyse-it for Microsoft Excel Method Validation Edition (version 2.20) and STATISTICA v.12 (StatSoft, OK, USA) software. The Mann-Whitney method was applied to analyze the statistical difference between groups of patients and Spearman rank correlation was used to analyze the data. Statistically significant differences were set at p < 0.05 using the two-sided test. Receiver operating characteristics (ROC) curves and the area under the ROC curves (AUC) were calculated to determine the potential of mpMRI and to discriminate between the patients who should undergo prostate biopsy and those who may be under constant observation (active surveillance), to reduce the false-negative rate of biopsy and avoid detection of low-grade disease. ROC curves were also assessed to determine the potential of physiological characteristics to discriminate between PCa and no-PCa (not diagnosed for prostate cancer) patients.
Results
Out of 40 consecutive men included in the study group, 55% were subjected to primary biopsy and 45% were directed to redo TRUS-Bx with the suspicion of prostate cancer. The median age of all patients was 66. The general median PSA was 7.1 ng/ml and median PSA density (PSAD), calculated on the basis of prostate volume obtained by mpMRI, was 0.181. We defined 40 suspected lesions on MRI images – 2 lesions PI-RADS 1 (5%), 7 lesions PI-RADS 2 (17.5%), 13 lesions PI-RADS 3 (32.5%), 11 lesions PI-RADS 4 (27.5%) and 7 lesions were scored PI-RADS 5 (17.5%). Within the group of patients with PCa, cancer tissue was identified only in the peripheral zone, while in the no-PCa group 56.25% and 40.6% of altered prostate tissue was localized, respectively, in transitional and peripheral zones. The patient characteristics are shown in Table 1.
Table 1
Patients’ characteristics
Two-sided Spearman’s correlation test was conducted for relationships between mpMRI, PSA and PSAD within the entire study group (40 patients) and for Gleason score, mpMRI, PSA and PSAD within the PCa group (8 patients). Obtained Spearman’s correlation coefficients (rs) values significant in the range 0.001 < p < 0.1 are presented in Table 2.
Table 2
Results of Spearman’s correlation analysis (significance levels for each correlation pair are represented by italics)
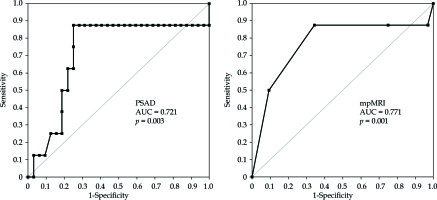
ROC analyses were performed to evaluate the capability of mpMRI to discriminate between direction of patients for prostate biopsy or active surveillance (Table 3, Figure 1). ROC values were AUC 0.721 (0.118, 95% CI: 0.490-0.952, p = 0.031) and AUC 0.771 (0.117, 95% CI: 0.542-1.001, p = 0.01), respectively for determining upon PSAD and mpMRI. The cut-off value was chosen to maximize both sensitivity and specificity, by applying the Youden index (maximum = sensitivity + specificity − 1) for each ROC analysis (Table 3).
Table 3
Results of ROC analysis for mpMRI and PSAD
| Characteristic | AUC | SE | Sensitivity | Specificity |
|---|---|---|---|---|
| mpMRI | 0.771 | 0.117 | 0.875 | 0.656 |
| PSAD | 0.721 | 0.118 | 0.875 | 0.75 |
| PSA | 0.652 | 0.122 | 0.75 | 0.594 |
Cancer detection and Gleason score differentiation via mpMRI resulted in the initiation of therapy: i) radical prostatectomy (1 patients); ii) radiotherapy treatment – brachytherapy (6 patients); and iii) active surveillance for low-risk disease (1 patient). The obtained characteristics of the prostate cancers among the majority of our patients implied the use of brachytherapy [1] as the primary treatment method. Patients’ preferences, when making a therapeutic decision and choosing the treatment method, were taken into account.
Prostate mpMRI PIRADS 1-2 reduces the need for repetitive biopsy through non-invasive serial monitoring for those under active surveillance.
Discussion
Diagnosis of clinically significant disease can be challenging even with application of state-of-art clinical methods. Biopsy remains the standard tool for PCa diagnosis. In prostate cancer diagnostics, standard TRUS-Bx is characterized by low detectability (approximately 25%), as three out of four patients undergoing biopsy are identified with no PCa [11]. As a consequence, a substantial number of patients continue to be under suspicion of cancer despite multiple benign biopsies. As a result, mpMRI is increasingly used to localize suspicious areas that could be targeted by MRI-TBx (MRI-targeted biopsy). To standardize mpMRI evaluation and decrease reporting ambiguity, the PI-RADS classification was created. The PIRADS v.2 protocol is routinely used in the assessment of focal prostatic lesions. However, variability in interpretation of prostate mpMRIs, including PI-RADS, among radiology experts and novices, remains an ongoing problem [12]. This is reflected in the creation of subsequent versions of the PIRADS and conducting scientific research in order to assess the diagnostic efficiency of mpMRI [13].
MRI-guided biopsy (MRI-TBx) can be performed using different techniques. Three techniques of MRI-guided procedures are available: i) in-bore MRI; ii) MRI-ultrasound software-assisted fusion biopsy; and iii) cognitive biopsy [11]. Importantly, the experience of the biopsy operator plays a major role in the precision of targeted biopsies. The cognitive approach is simple but mostly operator dependent. It is imperative for the operator to have a good understanding of the position of the lesion in the prostate gland to make a precise biopsy. Despite this, the advantages of MRI-targeted biopsy are twofold: improving detection of high-grade cancer by reducing the false-negative rate of biopsy, and avoiding detection of low-grade disease by selectively targeting tumor foci which are more likely to be clinically significant [14]. Stratification of risk for PCa patients is of great importance, as the mortality of PCa greatly varies according to various factors and in particular Gleason score. Over the last decade, PCa screening (PSA, systematic biopsies) has reduced the incidence of advanced PCa and PCa mortality; however, it leads to over-diagnosis and over-treatment of low-risk and indolent PCA [11,15].
mpMRI performed in our study group allowed 60% of patients to avoid undergoing invasive biopsies and experiencing potential minor complications (hematuria, hematospermia, rectal bleeding, mild to moderate degrees of vasovagal episodes, genitourinary tract infection) and major complications (urosepsis, rectal bleeding requiring intervention, acute urinary retention, Fournier’s gangrene and myocardial infarction) [16,17]. TRUS-Bx is associated with significant pain and discomfort in a proportion of men and it has psychological effects, including anxiety and depression [17]. As the mortality of PCa greatly varies according to several factors and in particular the Gleason score, it is essential to distinguish types of PCa and adapt proper treatment. These patients are under constant observation with active monitoring.
MRI-TBx is shown to detect a significantly higher rate of International Society of Urological Pathology (ISUP) grade > 2 cancers [18] and ISUP grade > 3 [19,20] in comparison with standard TRUS-Bx. Among the men with prior negative biopsies, the advantage of MRI-targeted biopsy application in identifying areas of suspicion within the prostate, which as several recent series have demonstrated might have been overlooked during consecutive systematic sampling, is indisputable. It has been observed that targeted biopsy detected more clinically significant cancers and fewer clinically insignificant cancers than systematic biopsy [21]. In our study, eight out of ten performed TRUS-Bx biopsies confirmed PCa with Gleason score ≥ 6 disease. Also Mendhiratta et al. found MRI fusion targeted biopsy to detect more Gleason score ≥ 7 disease [22]. In the light of evidence suggesting a clinical benefit of targeted biopsy for men with prior negative biopsies, the National Comprehensive Cancer Network (NCCN) guidelines for prostate cancer detection have suggested that men with prior negative biopsies (two or more) who are subjected to repeat biopsy should undergo pre-biopsy mpMRI to identify the areas of occult disease [23]. csPCa diagnosis constantly evolves to develop the most effective methods, and the 2019 edition of the European Association of Urology (EAU), European Association of Nuclear Medicine (EANM), European Society for Radiotherapy and Oncology (ESTRO), European Society of Urogenital Radiology (ESUR), International Society of Geriatric Oncology (SIOG) guideline on prostate cancer recommends performing mpMRI before the first set of biopsies [24,25].
Analysis of Spearman’s coefficient showed the highest statistical significance of correlation between mpMRI results and Gleason score in the group of PCa patients (rs = 0.41, p = 0.0085). This indicates the dependence between PI-RADS and Gleason scores. ROC analysis shows that the PSAD parameter has a higher diagnostic value in comparison with PSA (Table 3).
The low number of participants was due to the fact that they came from a single urological office, which is undoubtedly the limitation of this study. However, statistical analysis was carried out thoroughly and statistical significance was demonstrated in presented cases. We are still collecting data as new patients are referred for prostate biopsy and more physicians start to use pre-biopsy mpMRI. Additionally, based on the obtained mpMRI results, we will evaluate the significance of the SelectMDx test as a diagnostic tool to determine whether a patient is at higher or lower risk for prostate cancer and which patient can safely avoid biopsy.
Conclusions
Introducing pre-biopsy mpMRI into our contemporary PCa diagnosis pathway increased the diagnostic yield of transrectal biopsy by increasing the prostate cancer detection. This enabled the introduction of treatment (brachytherapy and radical prostatectomy) among patients with csPCa. mpMRI application also allowed biopsy to be avoided among patients with no csPCa.