Purpose
Global Cancer Data, GLOBOCAN 2018, has estimated that breast cancer is the most common cancer, representing approximately 14% of total cancer cases in India. It is the leading cause of cancer mortality, accounting for 11.1% of the total cancer-related deaths [1]. Breast conservative therapy (BCT), the standard of care for early breast cancer, involves lumpectomy with an adequate margin and axillary clearance, followed by adjuvant radiotherapy. It has been proven in multiple randomized control trials that there is no difference in survival outcomes between breast conservative treatment and total mastectomy [2, 3]. Adjuvant radiotherapy, following breast conservative surgery, involves irradiation of the whole breast as well as regional lymphatics in high-risk cases [4]. The recurrence in the vicinity of tumor bed was 50-60% of all local recurrences, which necessitated tumor bed boost irradiation [5, 6]. Whole breast irradiation to a dose of 45-50 Gy and a tumor bed boost of 10-20 Gy are recommended for adjuvant treatment following breast conservative surgery in patients with high-risk features (age < 50 years, grade 3 tumors, extensive intraductal component) [7]. The benefit of boost irradiation has been clearly shown in the European Organization for Research and Treatment of Cancer (EORTC) trial, which demonstrated a significant improvement in local control, especially among younger patients who received a tumor bed boost irradiation [8]. Various techniques used for delivering tumor bed boost include conformal teletherapy with fixed electron or photon beams, intensity-modulated radiation therapy (IMRT), volumetric-modulated arc therapy (VMAT), and interstitial brachytherapy.
High-dose-rate brachytherapy (HDR-BT), despite being invasive, is a well-studied modality for partial breast irradiation as well as for tumor bed boost after breast conservative surgery in breast cancers [9-18]. Volumetric-modulated arc therapy is a novel non-invasive radiation treatment technique that is based on volumetric-modulated rotational delivery, as compared to classic IMRT, which uses fixed gantry beams. By varying the speed of gantry rotation, multi-leaf collimator shape and continuously changing fluence (dose-rate), the VMAT delivers highly conformal IMRT plans in a short time, with less monitors units and possibly less dose to organs at risk [19]. The choice of boost technique is very important in breast cancer radiotherapy. Although dosimetric comparisons between brachytherapy and 3-dimensional conformal radiotherapy (3D-CRT) and IMRT were available [20-22], there is a lack of data on comparison of incidental doses to organs at risk (OARs) with tumor bed boost irradiation by VMAT and brachytherapy. Hence, the purpose of the study was to dosimetrically compare organs at risk between interstitial brachytherapy and VMAT-based tumor bed boost, following breast conservative surgery.
Material and methods
Treatment planning data of 50 patients with breast cancer who underwent breast conservation surgery, followed by either HDR interstitial brachytherapy (n = 25) or VMAT (n = 25) for tumor bed boost at regional cancer center, Jawaharlal Institute of Postgraduate Medical Education and Research Institute (JIPMER), were included in this retrospective study. All patients received whole breast irradiation of 46 Gy in 23 fractions. Tumor bed boost was considered in all patients < 50 years of age or those with high-risk features, such as grade 3 tumors and/or extensive intraductal component. No specific criteria were considered for choosing the technique for tumor bed boost. The organs at risk evaluated included the ipsilateral and contralateral lungs, heart, ipsilateral and contralateral breasts, skin, and ribs. All these organs at risk were contoured using available contouring atlases [23, 24]. Treatment planning for both groups were performed to meet the criteria provided by the Radiation Therapy Oncology Group (RTOG) 0413 protocol for target coverage and OARs sparing [25].
Interstitial brachytherapy
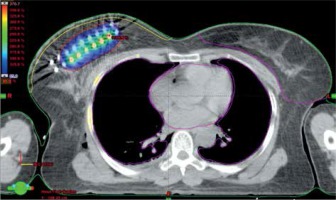
The patients in the brachytherapy group received perioperative interstitial HDR-BT of 15 Gy in 6 fractions over a period of 3 days as a boost, and whole breast irradiation was delivered after a gap of 2-3 weeks. Stainless steel or flexible catheters were implanted on operating table after lumpectomy and axillary dissection. Catheters were implanted after inspection of the tumor bed with guidance of template, so as to maintain the inter-catheter distance of 1 cm and preserve the geometry of the implant. The number of planes and number of catheters were decided intra-operatively, after examining the tumor bed and assessing the remaining breast volume. These patients were simulated after 24-48 hours in computed tomography (CT) simulator, and a 3 mm slice CT images were obtained from lower neck to upper abdomen; the images were then transferred to Oncentra treatment planning system (Elekta Instrument, AB Stockholm, Sweden). Target delineation was performed according to the GEC-ESTRO Breast Cancer Working Group recommendations [26]. The clinical target volume (CTV) volume included lumpectomy cavity plus at least 1-2 cm margin, depending on the size of the resection margin and cropped from the underlying pectoralis muscle and overlying skin by 5 mm. Skin was contoured as 5 mm thickness from the body surface. As there is no margin given for uncertainties in brachytherapy, planning target volume (PTV) became the CTV. The catheters were reconstructed and dose optimization was performed by adjusting dwell times in individual dwell positions in order to achieve the target volume coverage. AAPM TG43 formalism was used to generate dose distributions. Source position nearest to skin and ribs were placed at a distance of 10 mm to decrease high-dose regions within the ribs and skin. The dosimetric parameters evaluated included target coverage (V90, D90), high-dose volumes (V150, V200), dose homogeneity index (DHI), where DHI = (1 – V150/V100), and maximum skin isodose. It was aimed to maintain V90 > 90% of the PTV volume and D90 > 90% of the prescribed dose as well as to maintain the skin D1cc < 80-90%. Figure 1 shows dose color wash in a patient with two-plane rigid implant.
Volumetric-modulated arc therapy
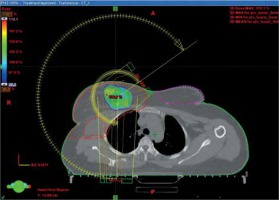
In the VMAT group, boost irradiation was applied after completion of whole breast irradiation. Usually, no interval was given between whole breast irradiation and boost irradiation. The seroma was contoured in CT planning obtained for whole breast radiation therapy (RT), with clips also contoured as a separate structure. Subsequently, CTV boost was created by adding the seroma and clips and by adding a margin of 1 cm, so that the clips were completely included in the CTV with a margin for microscopic disease. The CT planning was then registered with the pre-operative CT, and the location of previous tumor and tumor bed in the CT planning were confirmed. A margin of 5 mm was given for PTV boost from CTV boost but it was cropped to lie 5 mm below the skin. PTV boost_eval was defined by cropping PTV boost 5 mm inside the skin surface. PTV boost_eval was used for generating dose-volume histograms (DVHs) and comparative analyses. It was aimed to keep V90 > 90% of the PTV boost_eval and D90 > 90% of the prescribed dose. VMAT plans were created using Eclipse (version 10, Varian Medical Systems, USA) treatment planning system (TPS) using 6 MV photon arcs. The plans were made with one or two partial arcs, avoiding direct entrance through the contra-lateral breast by applying angulation to treatment couch. The dose schedule for VMAT was 16 Gy in 8 fractions. Figure 2 demonstrates dose distribution in a patient treated with VMAT-based tumor bed boost.
Statistical analysis
Patient and tumor characteristics were expressed as frequencies and percentages. SPSS software was utilized to perform the statistical analysis. Shapiro-Wilk’s test (p < 0.05) and visual inspection of histograms, Q-Q plots, and box plots revealed that the dosimetric data were not normally distributed for both the groups. Therefore, the data were expressed as a median with interquartile range. A non-parametric test (Mann-Whitney U) was applied for the analysis of parameters between the groups. A p-value of < 0.05 was considered significant.
Results
Characteristics
The patient and tumor characteristics are summarized in Table 1. The general characteristics of patients in each group were comparable. This study included T1, T2, and T3 tumors, which were down-staged by neoadjuvant chemotherapy. Out of the 25 patients in the brachytherapy group, 22 were rigid implants with a template and three were flexible implants. The median number of planes was 2, sixteen cases were two-plane implant, six cases were three-plane implant, and three cases were single-plane, as indicated in Table 2.
Table 1
Patient, tumor, and treatment characteristics
Target volumes
The results of brachytherapy and VMAT target volume parameters are presented in Table 3 as median and interquartile ranges (25th quartile and 75th quartile). In the brachytherapy group, the median PTV volume was 92.4 cc (range, 63.7-119.2 cc); D90 and V90 were 92.1% (range, 78.2-104.5%) of the prescribed dose and 91.3% (range, 82-97%) of the PTV volume, respectively; V100 was 84% (range, 74-93%). High-dose volumes, V150 and V200 were 44 cc (range, 18.5-70 cc) and 21 cc (range, 10.7-31 cc), respectively. The median DHI (1 – V150/V100) was 0.55 (range, 0.39-0.68). In the VMAT group, the median PTV volume was 133 cc (range, 75.1-158 cc); D90 was 99.9% (range, 97.7-110.7%) of the prescribed dose and Dmax was 108.4% (range, 106-112.3%); V90 and V100 were 99.9% (range, 99.8-100%) and 89.9% (range, 79.6-99.8%) of the PTV volume, respectively. The median conformity (CI = treated volume/PTV) and homogeneity indices (HI = D2% – D98%/D50%) were 0.94 (range, 0.81-1.1) and 0.09 (range, 0.09-0.14), respectively.
Table 3
Target volume parameters
Dosimetric results of organs at risk
Dosimetric parameters were obtained in percentages for simple interpretation. The dosimetric parameters in the brachytherapy and VMAT groups are listed in Table 4.
Table 4
Dosimetric parameters of organs at risk: volumetric-modulated arc therapy (VMAT) vs. brachytherapy
Ipsilateral lung
For ipsilateral lung, all the dosimetric parameters (Dmax, D0.1cc, D2cc, Dmean, and V10%) were significantly higher for the VMAT group, except for V5%. The D0.1cc and D2cc were almost 2-2.5 times higher in the VMAT group compared to the brachytherapy cohort.
Contralateral lung
For contralateral lung, all the dosimetric parameters were significantly lower for the brachytherapy group compared to the VMAT group. The mean lung dose in the brachytherapy group was 1.5% compared to 3.1% in the VMAT group (p-value < 0.0001). V5% and V10% were close to zero in the brachytherapy group, while it was significantly higher in the VMAT cohort (25% and 5.5%, respectively, p-value < 0.0001). These parameters demonstrated better sparing of contralateral lung with brachytherapy.
Contralateral breast
Parameters collected included D0.1cc, D2cc, Dmean, Dmax, and V5%. The D0.1cc, D2cc, and Dmax were significantly higher in the VMAT group (p-value < 0.0001). Also, the mean dose was higher in the VMAT group (1.5% vs. 1.7%), although not statistically significant. The volume receiving 5% of the prescribed dose V5% was significantly low in the brachytherapy group (0.0% vs. 8.4%, p-value < 0.0001).
Ribs
The high-dose received by ribs (Dmax, D0.1cc, D1cc, and D2cc) were significantly higher in the VMAT group (p-value < 0.0001), probably because of the fact that PTV extending into the ribs was not cropped in the VMAT group, while CTV in the brachytherapy cohort was cropped from the ribs. The mean dose received by ipsilateral ribs was higher in the VMAT group (9.4% vs. 8.1%), which was not statistically significant.
Skin
The skin was contoured as 5 mm thickness uniformly for all patients over the ipsilateral breast. Though, the maximum point dose was higher, D2cc was significantly less with brachytherapy compared to VMAT technique (p-value < 0.0001). There was no significant difference in the mean dose between the two groups.
Heart
The patients with left-sided breast cancer received higher doses to the heart when compared to right-sided cases. The dosimetric parameters, such as D0.1cc, D2cc, Dmax, V10%, and V20%, were not significantly different between the two groups. There was also no significant difference in the mean dose received by the heart for left-sided (5.9% vs. 5.5%) cases. However, a significant difference was observed in the mean dose for right-sided cases (2.8% vs. 0.8%, p-value = 0.003).
Ipsilateral non-target breast
The ipsilateral breast tissue minus PTV was contoured. The mean dose was higher in the brachytherapy group (31.2% vs. 20%, p-value < 0.0001), which was statistically significant. V100% was also higher in the brachytherapy group, and was statistically significant (2.5% vs. 0.1%, p-value < 0.0001). V75% (7% vs. 6.3%) and V50% (19.5% vs. 14%) were not statistically significant. V25% was significantly higher in the brachytherapy group (41.7% vs. 32.4%, p-value = 0.008).
Discussion
Tumor bed boost irradiation is clinically relevant and significant in breast radiotherapy after conservative surgery, especially in younger patients (age < 50 years) and those with an extensive intraductal component and grade 3 disease [8]. The two main types of radiation therapy, which could be applied to irradiate the tumor bed include external beam radiotherapy and brachytherapy. External beam radiotherapy may be delivered using fixed electron or photon beams, intensity-modulated radiation therapy, and volumetric-modulated arc therapy, while brachytherapy may be performed as interstitial procedure or as balloon brachytherapy. The present dosimetric study, to our knowledge, is the first study comparing dosimetric differences between interstitial brachytherapy and VMAT for tumor bed boost radiation in early-stage breast cancer. Even though Charaghvandi et al. [27] had dosimetrically compared interstitial brachytherapy and VMAT, their study was performed to evaluate the feasibility of single-fraction ablative radiotherapy in early-stage breast cancer, with brachytherapy plans generated virtually and with optimal placement of catheters. Our study findings revealed that interstitial brachytherapy results in significantly less dose to the contralateral breast, ipsilateral lung, contralateral lung, ribs, and skin, with similar heart-sparing in both the modalities, conforming outcomes of other studies. The reduction in most OARs doses could be explained by the fact that with interstitial brachytherapy, there is a rapid dose fall-off, whereas in VMAT, the doses were relatively higher, especially to the contralateral organs at risk due to scatter dose. In some cases, the medial entry point of VMAT beam was approaching the medial part of contralateral breast. The doses received by most of the OARs in the present study with brachytherapy were within the recommended dose-volume limits of ESTRO ACROP [28], as shown in Table 5.
Table 5
Comparison of dose-volume of organs at risk for brachytherapy with ESTRO ACROP recommendations
Weed et al. [20] performed a dosimetric comparison for accelerated partial breast irradiation delivered using 3D-CRT, interstitial brachytherapy, and MammoSite brachytherapy, and found that V20 and V10 for ipsilateral lung was lower for brachytherapy techniques compared with 3D-CRT; heart-sparing was the same with all techniques. In the present study, though VMAT was used, V10 was still significantly less with interstitial brachytherapy. Additionally, no significant differences between the two modalities with regard to most dosimetric parameters of the heart for both left-sided and right-sided cases were observed.
Terheyden et al. [21] retrospectively evaluated 136 early-stage breast cancer patients, in whom doses to OARs after tumor bed boost irradiation were delivered with either high-dose-rate brachytherapy or 3D conformal radiation therapy. Patients treated with brachytherapy received a dose of 10 Gy in single-fraction, while those managed with external beam radiotherapy received a dose of 10 Gy in 5 fractions. The results demonstrated significant sparing of organs at risk with interstitial brachytherapy compared to 3D-CRT, with no significant difference for maximum dose to the heart for left-sided cases, which is analogous to the findings of the current study. The maximum dose to the heart with HDR-BT for left-sided cases and right-sided patients were 29.8% and 6%, respectively, which is similar to the doses reported in the present study (25.5% and 7.4%). With 3D-CRT, the maximum doses for left-sided and right-sided irradiation were 29.9% and 16.7%, respectively. However, in the present study where VMAT was used, Dmax dose was lower for right-sided irradiation (7%). Even though the D max dose was higher for left-sided irradiation (39.2%), the D2cc dose was 30.2%. The maximum skin doses were higher, which may be related to the differences in skin delineation methods and location of tumors within the breast.
Lettmaier et al. [29] conducted a dosimetric study on left-sided breast cancer cases for radiation exposure to heart, lung, and skin between multicatheter interstitial brachytherapy-based accelerated partial breast irradiation and whole breast external beam radiotherapy. The EBRT technique used was 3D-CRT. When we compared OARs dosimetric parameters to those in the present study, we found that the heart, D0.1cc, and D2cc were similar in both studies (D0.1cc: 24.1% vs. 24.1%; heart D2cc: 21.4% vs. 20.5%), and the ipsilateral lung D0.1cc and D2cc were relatively less (D0.1cc: 39.2% vs. 32.5%; D2cc: 33.4% vs. 27.6%). The skin D0.1cc and D2cc were higher (D0.1cc: 70.3% vs. 96.8%; D2cc: 55.1% vs. 77.3%). Although, the depth of tumor bed was not analyzed in the present study, the higher skin doses and lower lung dosimetric parameters might be attributed to the location of tumor cavity.
Major et al. [22] performed a dosimetric comparison for accelerated partial breast irradiation between multicatheter interstitial brachytherapy and intensity-modulated radiotherapy. They reported mean doses to lung as 5.1% vs. 7.1%. Wu et al. [30] described a mean dose of 8% with intensity-modulated radiotherapy for accelerated partial breast irradiation. In our study, we found the mean lung doses as 5.9% vs. 10.5% confirming the dosimetric benefit of interstitial brachytherapy. The D0.1cc and D2cc in our study were 38.5% and 24.1% for interstitial brachytherapy, and 96.8% and 41.5% for VMAT, respectively. In the study by Major et al., the corresponding values for interstitial brachytherapy were 44.3% and 36.5%, and 62.7% and 50.2% with IMRT. The dosimetric parameters for the ribs (D0.1cc = 38.5% and D1cc = 28.5%) in the present study were also in favor of interstitial brachytherapy, which is much lesser than that reported by Major et al. (58% and 46%) [22].
Therefore, there is significantly less dose to contralateral breast, ipsilateral lung, contralateral lung, ribs, and skin with interstitial brachytherapy, which could be advantageous in terms of toxicity comparing to VMAT, especially in elderly patients, given the possibility of implantation during surgery and thereby reducing overall duration of treatment [31, 32]. Although, the present study is the first one to compare the dosimetric differences between interstitial brachytherapy and VMAT for tumor bed boost, it has several limitations. Firstly, it is retrospective in nature, which leads to inherent risk of bias. No specific inclusion and exclusion criteria were considered for choosing a particular modality of tumor bed boost for patients after whole breast radiotherapy. Electrons, a convenient modality for delivering tumor bed boost, were not included in this dosimetric analysis. Other factors, such as depth of the tumor bed, were not taken into account while selecting the boost technique. Further, this study presents only the dosimetric results, with no correlation to clinical end points, including acute and late toxicities, local control, and cosmetic outcomes.
Conclusions
High-dose-rate interstitial brachytherapy, when used as a modality for tumor bed boost irradiation with breast conservation therapy, results in significant reduction of dose to most organs at risk in comparison to VMAT. Prospective randomized controlled trial incorporating conformal fixed or intensity-modulated photon beams, electron beams, and interstitial brachytherapy, with correlation to clinical and patient-reported outcomes, are essential to identify the best treatment modality for delivering tumor bed boost.