Purpose
Treatment for cervical cancer mostly combines external beam radiotherapy (EBRT) with high-dose-rate (HDR) brachytherapy (BT) and concomitant chemotherapy (CCRT) with weekly cisplatin [1-3]. In BT, clinically favorable dose distributions are achieved by adjusting source position and its associated dwell time within applicator’s catheters (intracavitary BT) and additional interstitial needles (interstitial BT). The main challenge in the process of BT treatment planning is to find the combination of dwell position, dwell time, or associated weight, to maximize target dose coverage while minimizing dose to organs at risk (OARs). Another goal of BT is to produce robust plans.
Manual optimization for treatment planning is conventionally used for BT. In EBRT, inverse optimization algorithms have been the golden standard for several years, meanwhile research is ongoing exploring the use of such algorithms in intracavitary/interstitial BT. There are two main inverse optimization methods used: hybrid inverse treatment planning and optimization (HIPO) and inverse planning simulated annealing (IPSA), both employed in the Oncentra Brachy treatment planning system (TPS) version 4.6.3 (Elekta AB, Stockholm, Sweden). These new planning methods are facilitated by the integration of 3D imaging, especially magnetic resonance imaging (MRI) for BT treatment planning [4]. With MRI, it is possible to contour volumes more accurately than with CT scan. Inverse planning methods allow dose constraint-based dwell time adjustment, increasing target coverage, and spare OARs, which are better defined with MRI [5-7]. Some studies have shown that inverse planning optimization (with HIPO or IPSA) seems to be more efficient than manual optimization for cervical cancer BT treatment [8-11]. Indeed, inverse planning optimization is not operator-dependent, and should result in a more reproducible and faster planning process. According to these studies, HIPO planning provides slightly better results than IPSA for cervical cancer BT.
Recent research on cervical cancer BT, such as EMBRACE protocols, allowed to define precise constraints for treatment planning in terms of dose-volume indices (DVI), including planning aims (soft constraints) and limits for prescribed dose (hard constraints) [12]. These limits are calculated during inverse optimization using the TG-43 formalism from the American Association of Physicists in Medicine (AAPM) [13], and then compared with the EMBRACE recommendations.
The purpose of this study was to compare dosimetric metrics for cervical cancer BT from manual and inverse treatment planning optimization methods, with manual planning using Oncentra Brachy and with inverse planning using Oncentra Brachy HIPO and RayStation Brachy (RSB), version 2024B (RaySearch Laboratories AB, Stockholm, Sweden).
Material and methods
Thirty-one consecutive patients with cervical cancer, treated between 2022 and 2024, were included in this study. Patients underwent CCRT (1.8-2.0 Gy per fraction, 5 times a week) with a median total dose of 45 Gy, followed by four fractions with two implants (two fractions each) of 8 Gy to D90 high-risk clinical target volume (HR-CTV) using high-dose-rate brachytherapy (HDR-BT) [14, 15], delivered for about 1 week. For this study, patients requiring combined intracavitary and interstitial BT and those with at least 4 interstitial needles were selected. Indeed, inverse planning is more efficient with a bigger number of interstitial needles [9]. Depending on tumor anatomy, two different applicator models from Elekta were applied, i.e., Geneva universal gynecological applicator or Venezia advanced gynecological applicator [16, 17]. Patient distribution according to those criteria is summarized in Table 1. All patients were treated by HDR-BT, with a remote afterloading unit MicroSelectron v. 2 (Nucletron BV, Veenendaal, The Netherlands). Treatment was delivered using an iridium-192 (192Ir) source with an initial activity of 429.4 GBq (corresponding to a reference air kerma rate of 47.24 mGy.m2.h-1) and a source step of 2.5 mm. For each patient, three different treatment plans were generated: one manual, which is commonly used clinically, and two with inverse treatment planning consisting of HIPO and RSB. Manual plans were created by two experienced medical physicists and used clinically for patients treatments. All volumes of interest were contoured by the same senior expert radiation therapists, and the same contour set was applied across all optimization methods.
Table 1
Distribution of patients by applicator type and number of interstitial needles
| Parameter | # Patients | % | |
|---|---|---|---|
| Applicator | |||
| Geneva | 22 | 71 | |
| Venezia | 9 | 29 | |
| Interstitial needles | |||
| 4 | 9 | 29 | |
| 5 | 3 | 10 | |
| 6 | 6 | 19 | |
| 7 | 2 | 6 | |
| 8 | 7 | 23 | |
| 9 | 2 | 6 | |
| 10 | 1 | 3 | |
| 11 | 1 | 3 | |
The hybrid inverse treatment planning and optimization (HIPO) algorithm is an anatomy-based optimization method developed by Karabis, Giannouli, and Baltas [18]. Its efficacy versus manual planning has already been shown for prostate cancer BT [9, 19, 20]. Dwell time gradient restriction at 0.5 in HIPO parameters were selected.
On the other hand, the RayStation Brachy module is a TPS newly developed by RayStation Laboratories. It includes an inverse planning engine that allows defining a variety of objectives and constraints for OARs, targets, and points. To verify the accuracy of dose distribution on this new TPS, quality control test was performed recommended by the Report No. 36 of the Société Française de Physique Médicale (SFPM) for HDR source projectors [21]. The results were meeting the requirements of all three geometries described in the report. The optimization problem was defined using different optimization functions and constraints on target volumes and OARs:
min. DVH with the prescribed dose to 95% volume of HR-CTV,
max. DVH with 150% of the prescribed dose to 50% volume of HR-CTV,
min. dose for GTV and intermediate risk clinical target volume (IR-CTV),
max. dose for OARs,
dose fall-off and max. DVH for “External IR-CTV” volume.
These optimization functions had patient-specific weights, ranging from 10 to 90 for OARs and from 50 to 150 for targets.
In both inverse planning methods, the same procedure was applied for each patient. First, objectives and constraints for target volumes and OARs were defined according to the EMBRACE protocols [12]. Then, inverse planning was performed, and the resulting doses were compared with those obtained from manual plans and recommended by the EMBRACE protocols. In the final step, constraints and objectives were adjusted to optimize the final treatment plan. After each inverse planning, the plan was normalized to the prescribed dose of 8 Gy for D90 HR-CTV.
Organs and targets were contoured on MRI, according to the GYN GEC-ESTRO recommendations: bladder, rectum, sigmoid, and bowel for OARs, and GTV, HR-CTV, and IR-CTV for targets [22, 23]. Points of interest (POIs), such as the recto-vaginal point, were also placed on images, according to the ICRU Report 89 [24]. The following dose-volume parameters were collected for treatment plan analysis:
V200 = volume of normal tissue irradiated by 200% of prescribed dose,
D90 HR-CTV = dose received by 90% of HR-CTV,
D98 (x) = dose received by 98% of target x,
D2cc (y) = dose received by the most irradiated 2 cm3 of OAR y,
recto-vaginal point dose,
total reference air kerma (TRAK).
In addition, dwell time percentage in intrauterine tandem, ovoids, and interstitial needles were assessed. For all patients, three dosimetric indexes were calculated: Paddick, which is used to assess conformity of irradiated volume with target volume [11, 25], COIN to measure the extent to which the prescribed dose is adapted to shape and volume of the target while minimizing irradiation to healthy tissue [11, 25], and dose homogeneity index (DHI) [27]. For all three indexes, a value of 1 indicated a good conformity or homogeneity. Finally, dwell time inhomogeneity index (DTII) [28] for interstitial needles was calculated:
Where a DTII value close to 0 indicated a good dwell time homogeneity.
During planning, all doses were calculated in equivalent dose of 2 Gray (Gy) per fraction (EQD2) according to linear-quadratic model with α/β = 3 for OARs and recto-vaginal point, and α/β = 10 for targets, by adding 4 fractions of BT plus 45 Gy of EBRT, to verify that the EMBRACE dose limits and objectives were met. Results were then analyzed using Student’s paired t-test, comparing manual vs. HIPO, HIPO versus RSB, and manual versus RSB. The difference was considered significant for a p-value lower than 0.05.
Results
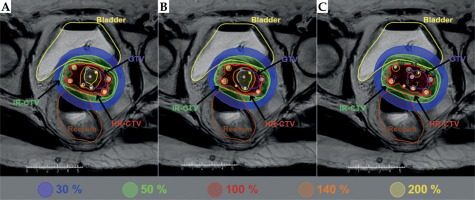
For the three planning methods, i.e., manual, HIPO, and RSB, an example of dose distribution in the axial view is shown in Figure 1. All three plans were generated for the same patient, with 8 interstitial needles. The main difference between manual and inverse planning was observed for 200% isodose. After manual planning, most of the dose was received from the intrauterine tandem, with low dwell time in additional needles, creating many hot spots. In HIPO method, target volume coverage was similar, with a reduction of hot spots through a decrease of dwell time in intrauterine tandem and interstitial needles. In RSB planning, there was an increase in the loading of interstitial needles at the expense of ovoids and intrauterine tandem, leading to a lower coverage of target volume but fewer hot spots in dose distribution.
Fig. 1
Example of dose distribution in one patient using 3 treatment planning methods: A) manual, B) HIPO, and C) RSB
The average values of dose-volume parameters for the three optimization methods are reported in Table 2, along with p-values of the three comparisons: manual vs. HIPO, HIPO vs. RSB, and manual vs. RSB. For all the patients in this study, both HIPO and RSB methods produced dosimetrically acceptable treatment plans. In all cases, the GEC-ESTRO dosimetric objectives and limits for target coverage and OARs sparing were met. In the dose to GTV, no significant difference for all three plans was observed (p > 0.05). HIPO plans were significantly better than manual plans (p < 0.05), where an increase of the dose to target volumes and a decrease of the dose to OARs were seen. Compared with manual planning, RSB demonstrated statistically different DVIs only for D2cc of the bowel, which was reduced. Moreover, RSB increased the dose to all OARs compared with HIPO, reduced the D98 to HR-CTV, and increased the D98 to IR-CTV. However, the dose to OARs remained within the limits prescribed by the GEC-ESTRO group. The recto-vaginal point dose was significantly higher in RSB plans compared with HIPO, and lower in HIPO compared with manual plans. The total volume of tissue receiving 200% of the prescribed dose was highest in manual optimization, followed by HIPO and RSB.
Table 2
Summary of DVIs (mean ± SD) and p-values of manual optimization and inverse optimization, with HIPO and RSB for a single-fraction of HDR-BT; all doses are physical doses
The average values of dosimetric indices and TRAK are presented in Table 3. Conformity of the plans, given by Paddick and COIN indices, was greater in HIPO optimization, followed by RSB and manual. For the homogeneity of dose distribution, there was no statistical difference in HIPO and manual plans (p > 0.05), but RSB plans increased significantly the homogeneity than the other two. The mean TRAK value for manual and RSB was similar, whereas for HIPO, it was significantly lower than for both other methods.
Table 3
Summary of dosimetric indexes (mean ± SD) and p-values of manual optimization and inverse optimization, with HIPO and RSB for a single-fraction of HDR-BT
Table 4 shows the average dwell time values in different parts of the applicator, including intrauterine tandem, ovoids, and interstitial needles. For each patient, the source strength was normalized to 43.64 mGy.m2.h-1. Inverse optimization methods decreased the proportion of dwell time in ovoids, and increased the loading in interstitial needles compared with manual planning. RSB also decreased the dwell time percentage in intrauterine tandem, while HIPO increased it, although the difference between manual and HIPO for intrauterine tandem was not statistically significant. For RSB plans, there was a statistical difference in manual plans for each part of the applicator, without significant difference in the total dwell time. The difference between HIPO and RSB was not significant, except the dwell time percentage in vaginal ovoids (p > 0.05). The total dwell time was significantly reduced in HIPO planning method.
Table 4
Summary of dwell times (mean ± SD) and p-values of manual optimization and inverse optimization, with HIPO and RSB for a single-fraction of HDR-BT
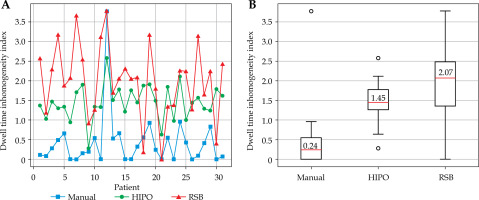
Figure 2 shows the variation in dwell time homogeneity in interstitial needles for all three methods. Each comparison was statistically significant (p < 0.05). Inverse planning methods resulted in a lower homogeneity of dwell time in needles compared with manual planning. Similarly, RSB plans were less homogeneous than those generated with HIPO.
Discussion
The dose calculation algorithms implemented in RSB and Oncentra were both based on the TG-43 formalism, and the source model used in the two TPSs was identical, namely the MicroSelectron HDR v. 3. The radial dose functions and anisotropy functions were also equivalent [29]. Consequently, when treatment conditions are strictly comparable (identical dwell positions and dwell times), the resulting dose distributions exhibit a high level of agreement between two systems. The quality assurance checks recommended by the SFPM Report No. 36 [21] were performed on each TPS and yielded similar results, further confirming the robustness of their dose calculations.
During manual plan optimization, the medical physicist adjusts the dwell time according to the dwell position and the patient’s anatomical configuration. Methodology and tools provided by both Oncentra and RSB are highly similar: the user can directly set dwell times or their relative weighting. Therefore, the results of manual optimization appear comparable between the two TPSs, with variability mainly attributed to the operator. For this reason, manual optimization has not been reproduced in RSB; instead, clinically validated plans approved by the radiation therapist and medical physicist were retained as the reference.
If the GYN GEC-ESTRO recommendations for dose to targets and OARs are the main criteria in brachytherapy, other parameters are also important. Therefore, during manual planning in cervical cancer BT, physicists aim to achieve a pear-shaped dose distribution. This typical dose distribution has been shown to result in good clinical outcomes, with high local control and low side effects [6, 30]. As in Trnková et al. [8], we also attempted to preserve this spatial dose distribution during inverse planning to avoid high-dose regions outside the cervix.
Furthermore, dwell time homogeneity is a criterion often analyzed by physicists, as it is related to the presence or not of hot spots in dose distribution. According to other studies on inverse planning in HDR-BT, manual planning often results in more uniform plans than inverse optimization methods [11, 28], especially in terms of dwell time in interstitial needles. This is in line with the current study, where DTII in interstitial needles was lower for manual planning than for inverse planning. Also, RSB plans are less homogeneous than those of HIPO, which is most likely due to a dwell time gradient restriction in HIPO parameters. This parameter, available in RSB, was not used in this study because it resulted in higher doses to all OARs, which was not desired.
However, the higher DTII observed in interstitial needles with inverse planning is an aspect that could be improved. In Chajon et al. [31], although they studied inverse planning in pulse-dose-rate BT for gynecological cancer, they also concluded that supplementary parameters are needed in inverse planning to increase the homogeneity of dwell time distribution. Such parameters are expected to be added in future versions of RSB, which are still under development.
Regardless of the dwell time homogeneity, a dosimetrically acceptable plan is characterized by a good target volume coverage and OARs sparing. The target coverage was only increased for the D98 HR-CTV in HIPO than manual. At the same time, HIPO planning resulted in a lower dose to OARs compared with the other two methods. This may be related to the two conformity indexes, i.e., Paddick and COIN, calculated in this study: both indices increased in HIPO planning compared with manual and RSB optimization. For RSB planning, although there is no variation of the dose to targets, the dose to OARs, except the bowel, increased. However, even if the dose to OARs is higher than with manual planning and HIPO, it remained within the limits prescribed by the GYN GEC-ESTRO group.
Another parameter to consider when performing HDR-BT planning is the value of TRAK to characterize the treatment intensity [24]. In our study, we showed that the TRAK value in HIPO plans increased significantly than in the other two methods. This is consistent with the fact that the total dwell time of these plans has also decreased. Moreover, we could see a significant reduction in high-dose volumes (V200) of total tissue in HIPO. Similarly, RSB plans resulted in lower V200 value for total tissue compared with manual, while achieving a similar TRAK and total dwell time. With these results, combined with the DHI value that was higher in RSB, we can assume that RSB method allows a more homogeneous dose distribution, with more efficient use of dwell positions despite a higher DTII value in interstitial needles.
Trnková et al. [8], one of the few studies on HIPO method for cervical cancer BT, showed that HIPO plans achieved better conformity than manual plans, reflected by higher Paddick and COIN indices, while resulting in a 13% reduction in total dwell time for patient with interstitial needles. This is in line with our study that reported a 10% decrease in total dwell time and an improvement in plan conformity for both inverse planning methods.
The observed dosimetric differences among the three optimization strategies can be explained by the intrinsic characteristics of inverse-planning algorithms. While HIPO is based on a stochastic method that tends to homogenize dwell times and more strictly conform to dose limits for anatomical structures [18], RSB optimization algorithm is based on iterative adjustment of plan parameters through the weight on targets and OARs [32], leading to a higher total dwell time and a lower dwell time homogeneity. However, both the inverse algorithms used BFGS method for dose distribution optimization, which justify the coherence between HIPO and RSB calculations.
One aspect that was not considered in this study nor in previous ones on inverse planning, is the time needed to create a clinically acceptable plan with inverse planning methods. The time was difficult to calculate, as it strongly depends on the individual patient case, and can vary significantly from one case to another. A more detailed study about planning time could be of interest to determine whether inverse planning methods offer a time-saving advantage for the physician compared with manual planning.
In BT, inverse planning is currently used clinically mainly for prostate cancer. However, our findings, along with recent studies, provide valuable insights into the advantage of inverse planning methods over manual optimization for other anatomical sites. Priean et al. demonstrated the superiority of HIPO for interstitial HDR cervical BT [33], while Major et al. showed that inverse planning can provide better dose distribution for interstitial HDR breast BT [34]. As emphasized by Berger et al. [35], inverse planning is a modern tool, and its efficacy compared with manual planning has been further supported by our study.
Conclusions
In HDR cervical cancer BT, inverse planning methods lead to dosimetrically acceptable plans, which improve upon manual plans in several aspects. HIPO method, implemented in the Oncentra TPS, provide better plans with lower dose to OARs while increasing the target coverage, with a similar homogeneity of dose distribution and a lower total treatment time. RSB method, implemented in the RayStation TPS, increase the received dose in all tissues, while remaining within dose limitations for OARs, but significantly increase the homogeneity of dose distribution.